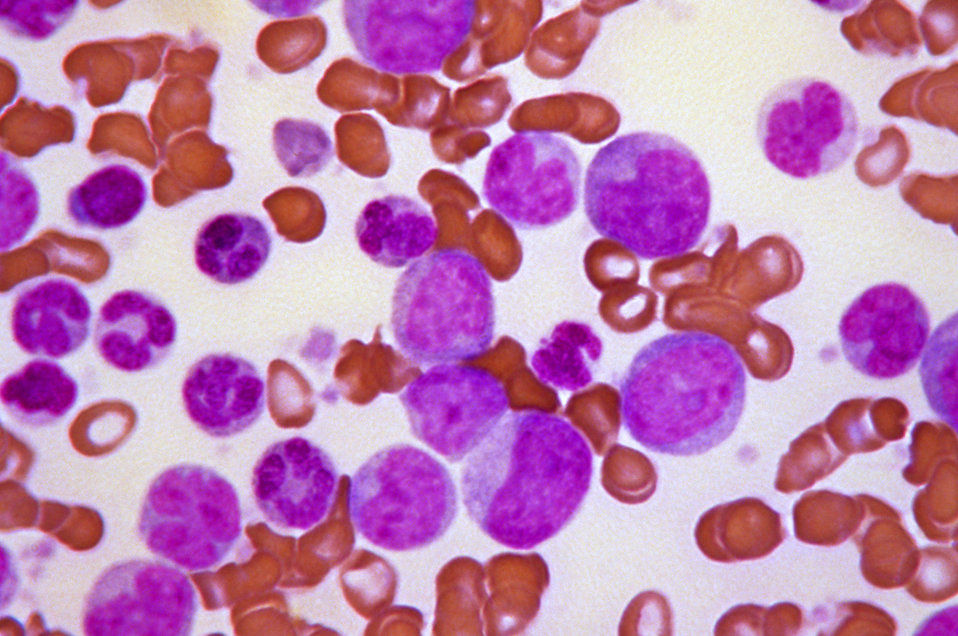
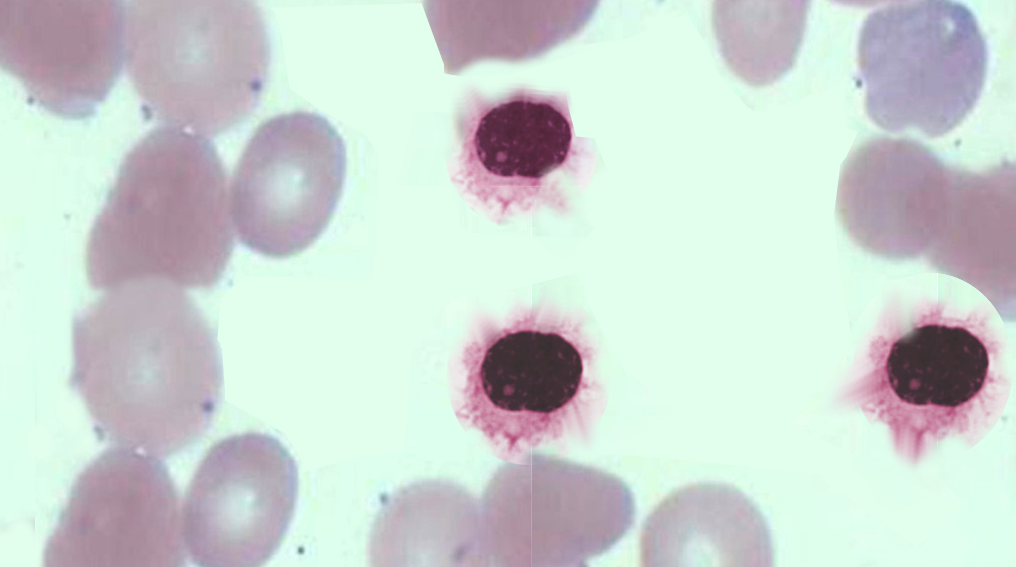
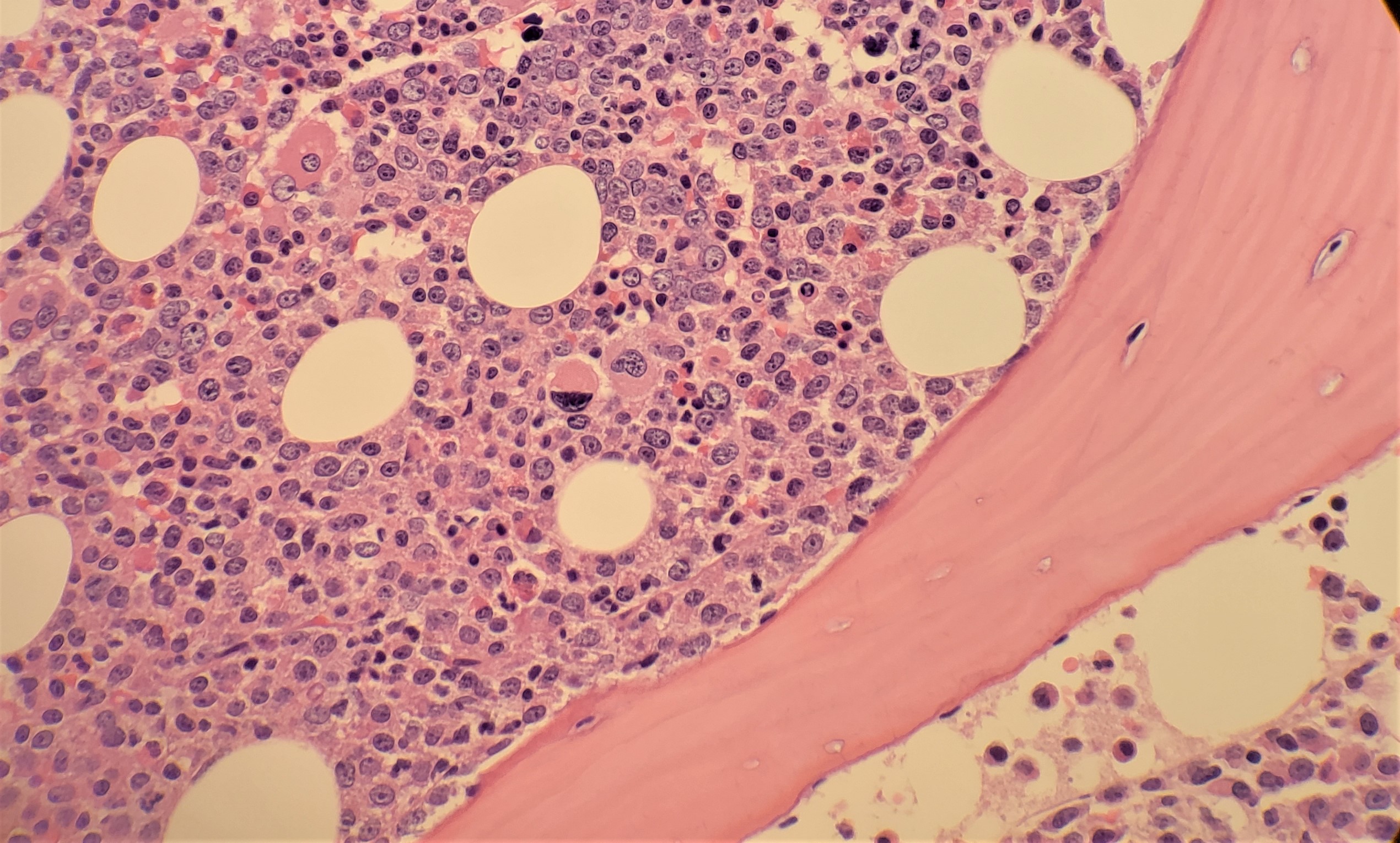
[1]
Arber DA, Orazi A, Hasserjian R, Thiele J, Borowitz MJ, Le Beau MM, Bloomfield CD, Cazzola M, Vardiman JW. The 2016 revision to the World Health Organization classification of myeloid neoplasms and acute leukemia. Blood. 2016 May 19:127(20):2391-405. doi: 10.1182/blood-2016-03-643544. Epub 2016 Apr 11
[PubMed PMID: 27069254]
[2]
. Erratum: Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: a cancer journal for clinicians. 2020 Jul:70(4):313. doi: 10.3322/caac.21609. Epub 2020 Apr 6
[PubMed PMID: 32767693]
[3]
Vardiman JW. The World Health Organization (WHO) classification of tumors of the hematopoietic and lymphoid tissues: an overview with emphasis on the myeloid neoplasms. Chemico-biological interactions. 2010 Mar 19:184(1-2):16-20. doi: 10.1016/j.cbi.2009.10.009. Epub 2009 Oct 24
[PubMed PMID: 19857474]
Level 3 (low-level) evidence
[4]
Brunning RD. Classification of acute leukemias. Seminars in diagnostic pathology. 2003 Aug:20(3):142-53
[PubMed PMID: 14552428]
[5]
Yamamoto JF, Goodman MT. Patterns of leukemia incidence in the United States by subtype and demographic characteristics, 1997-2002. Cancer causes & control : CCC. 2008 May:19(4):379-90
[PubMed PMID: 18064533]
[6]
Siegel RL, Miller KD, Jemal A. Cancer Statistics, 2017. CA: a cancer journal for clinicians. 2017 Jan:67(1):7-30. doi: 10.3322/caac.21387. Epub 2017 Jan 5
[PubMed PMID: 28055103]
[7]
Bispo JAB, Pinheiro PS, Kobetz EK. Epidemiology and Etiology of Leukemia and Lymphoma. Cold Spring Harbor perspectives in medicine. 2020 Jun 1:10(6):. doi: 10.1101/cshperspect.a034819. Epub 2020 Jun 1
[PubMed PMID: 31727680]
Level 3 (low-level) evidence
[8]
Miranda-Filho A, Piñeros M, Ferlay J, Soerjomataram I, Monnereau A, Bray F. Epidemiological patterns of leukaemia in 184 countries: a population-based study. The Lancet. Haematology. 2018 Jan:5(1):e14-e24. doi: 10.1016/S2352-3026(17)30232-6. Epub
[PubMed PMID: 29304322]
Level 2 (mid-level) evidence
[9]
Snyder R. Leukemia and benzene. International journal of environmental research and public health. 2012 Aug:9(8):2875-93. doi: 10.3390/ijerph9082875. Epub 2012 Aug 14
[PubMed PMID: 23066403]
[10]
Friedman DL,Whitton J,Leisenring W,Mertens AC,Hammond S,Stovall M,Donaldson SS,Meadows AT,Robison LL,Neglia JP, Subsequent neoplasms in 5-year survivors of childhood cancer: the Childhood Cancer Survivor Study. Journal of the National Cancer Institute. 2010 Jul 21;
[PubMed PMID: 20634481]
[11]
Davis AS, Viera AJ, Mead MD. Leukemia: an overview for primary care. American family physician. 2014 May 1:89(9):731-8
[PubMed PMID: 24784336]
Level 3 (low-level) evidence
[12]
Stieglitz E, Loh ML. Genetic predispositions to childhood leukemia. Therapeutic advances in hematology. 2013 Aug:4(4):270-90. doi: 10.1177/2040620713498161. Epub
[PubMed PMID: 23926459]
Level 3 (low-level) evidence
[13]
Leszczenko P, Borek-Dorosz A, Nowakowska AM, Adamczyk A, Kashyrskaya S, Jakubowska J, Ząbczyńska M, Pastorczak A, Ostrowska K, Baranska M, Marzec KM, Majzner K. Towards Raman-Based Screening of Acute Lymphoblastic Leukemia-Type B (B-ALL) Subtypes. Cancers. 2021 Oct 31:13(21):. doi: 10.3390/cancers13215483. Epub 2021 Oct 31
[PubMed PMID: 34771646]
[14]
Looi W,Zargari A,Dun K,Grigoriadis G,Fedele P,Gregory GP,Low MSY, Concomitant diagnosis of chronic myeloid leukaemia and myeloma. Pathology. 2021 Nov 11;
[PubMed PMID: 34776244]
[15]
Nebgen DR, Rhodes HE, Hartman C, Munsell MF, Lu KH. Abnormal Uterine Bleeding as the Presenting Symptom of Hematologic Cancer. Obstetrics and gynecology. 2016 Aug:128(2):357-363. doi: 10.1097/AOG.0000000000001529. Epub
[PubMed PMID: 27400009]
[16]
Sanz MA, Grimwade D, Tallman MS, Lowenberg B, Fenaux P, Estey EH, Naoe T, Lengfelder E, Büchner T, Döhner H, Burnett AK, Lo-Coco F. Management of acute promyelocytic leukemia: recommendations from an expert panel on behalf of the European LeukemiaNet. Blood. 2009 Feb 26:113(9):1875-91. doi: 10.1182/blood-2008-04-150250. Epub 2008 Sep 23
[PubMed PMID: 18812465]
[17]
Mantha S, Goldman DA, Devlin SM, Lee JW, Zannino D, Collins M, Douer D, Iland HJ, Litzow MR, Stein EM, Appelbaum FR, Larson RA, Stone R, Powell BL, Geyer S, Laumann K, Rowe JM, Erba H, Coutre S, Othus M, Park JH, Wiernik PH, Tallman MS. Determinants of fatal bleeding during induction therapy for acute promyelocytic leukemia in the ATRA era. Blood. 2017 Mar 30:129(13):1763-1767. doi: 10.1182/blood-2016-10-747170. Epub 2017 Jan 12
[PubMed PMID: 28082441]
[18]
Montesinos P, Bergua JM, Vellenga E, Rayón C, Parody R, de la Serna J, León A, Esteve J, Milone G, Debén G, Rivas C, González M, Tormo M, Díaz-Mediavilla J, González JD, Negri S, Amutio E, Brunet S, Lowenberg B, Sanz MA. Differentiation syndrome in patients with acute promyelocytic leukemia treated with all-trans retinoic acid and anthracycline chemotherapy: characteristics, outcome, and prognostic factors. Blood. 2009 Jan 22:113(4):775-83. doi: 10.1182/blood-2008-07-168617. Epub 2008 Oct 22
[PubMed PMID: 18945964]
[19]
Lallemand-Breitenbach V, de Thé H. Retinoic acid plus arsenic trioxide, the ultimate panacea for acute promyelocytic leukemia? Blood. 2013 Sep 19:122(12):2008-10. doi: 10.1182/blood-2013-06-505115. Epub 2013 Jul 26
[PubMed PMID: 23894153]
[20]
Iland HJ, Bradstock K, Supple SG, Catalano A, Collins M, Hertzberg M, Browett P, Grigg A, Firkin F, Hugman A, Reynolds J, Di Iulio J, Tiley C, Taylor K, Filshie R, Seldon M, Taper J, Szer J, Moore J, Bashford J, Seymour JF, Australasian Leukaemia and Lymphoma Group. All-trans-retinoic acid, idarubicin, and IV arsenic trioxide as initial therapy in acute promyelocytic leukemia (APML4). Blood. 2012 Aug 23:120(8):1570-80; quiz 1752
[PubMed PMID: 22715121]
[21]
Abaza Y,Kantarjian H,Garcia-Manero G,Estey E,Borthakur G,Jabbour E,Faderl S,O'Brien S,Wierda W,Pierce S,Brandt M,McCue D,Luthra R,Patel K,Kornblau S,Kadia T,Daver N,DiNardo C,Jain N,Verstovsek S,Ferrajoli A,Andreeff M,Konopleva M,Estrov Z,Foudray M,McCue D,Cortes J,Ravandi F, Long-term outcome of acute promyelocytic leukemia treated with all-{i}trans{/i}-retinoic acid, arsenic trioxide, and gemtuzumab. Blood. 2017 Mar 9
[PubMed PMID: 28003274]
[22]
Döhner H, Estey E, Grimwade D, Amadori S, Appelbaum FR, Büchner T, Dombret H, Ebert BL, Fenaux P, Larson RA, Levine RL, Lo-Coco F, Naoe T, Niederwieser D, Ossenkoppele GJ, Sanz M, Sierra J, Tallman MS, Tien HF, Wei AH, Löwenberg B, Bloomfield CD. Diagnosis and management of AML in adults: 2017 ELN recommendations from an international expert panel. Blood. 2017 Jan 26:129(4):424-447. doi: 10.1182/blood-2016-08-733196. Epub 2016 Nov 28
[PubMed PMID: 27895058]
[23]
Hills RK, Castaigne S, Appelbaum FR, Delaunay J, Petersdorf S, Othus M, Estey EH, Dombret H, Chevret S, Ifrah N, Cahn JY, Récher C, Chilton L, Moorman AV, Burnett AK. Addition of gemtuzumab ozogamicin to induction chemotherapy in adult patients with acute myeloid leukaemia: a meta-analysis of individual patient data from randomised controlled trials. The Lancet. Oncology. 2014 Aug:15(9):986-96. doi: 10.1016/S1470-2045(14)70281-5. Epub 2014 Jul 6
[PubMed PMID: 25008258]
Level 1 (high-level) evidence
[24]
Schlenk RF, Weber D, Fiedler W, Salih HR, Wulf G, Salwender H, Schroeder T, Kindler T, Lübbert M, Wolf D, Westermann J, Kraemer D, Götze KS, Horst HA, Krauter J, Girschikofsky M, Ringhoffer M, Südhoff T, Held G, Derigs HG, Schroers R, Greil R, Grießhammer M, Lange E, Burchardt A, Martens U, Hertenstein B, Marretta L, Heuser M, Thol F, Gaidzik VI, Herr W, Krzykalla J, Benner A, Döhner K, Ganser A, Paschka P, Döhner H, German-Austrian AML Study Group. Midostaurin added to chemotherapy and continued single-agent maintenance therapy in acute myeloid leukemia with FLT3-ITD. Blood. 2019 Feb 21:133(8):840-851. doi: 10.1182/blood-2018-08-869453. Epub 2018 Dec 18
[PubMed PMID: 30563875]
[25]
DiNardo CD, Jonas BA, Pullarkat V, Thirman MJ, Garcia JS, Wei AH, Konopleva M, Döhner H, Letai A, Fenaux P, Koller E, Havelange V, Leber B, Esteve J, Wang J, Pejsa V, Hájek R, Porkka K, Illés Á, Lavie D, Lemoli RM, Yamamoto K, Yoon SS, Jang JH, Yeh SP, Turgut M, Hong WJ, Zhou Y, Potluri J, Pratz KW. Azacitidine and Venetoclax in Previously Untreated Acute Myeloid Leukemia. The New England journal of medicine. 2020 Aug 13:383(7):617-629. doi: 10.1056/NEJMoa2012971. Epub
[PubMed PMID: 32786187]
[26]
Kolitz JE, Strickland SA, Cortes JE, Hogge D, Lancet JE, Goldberg SL, Villa KF, Ryan RJ, Chiarella M, Louie AC, Ritchie EK, Stuart RK. Consolidation outcomes in CPX-351 versus cytarabine/daunorubicin-treated older patients with high-risk/secondary acute myeloid leukemia. Leukemia & lymphoma. 2020 Mar:61(3):631-640. doi: 10.1080/10428194.2019.1688320. Epub 2019 Nov 25
[PubMed PMID: 31760835]
[27]
Slayton WB, Schultz KR, Kairalla JA, Devidas M, Mi X, Pulsipher MA, Chang BH, Mullighan C, Iacobucci I, Silverman LB, Borowitz MJ, Carroll AJ, Heerema NA, Gastier-Foster JM, Wood BL, Mizrahy SL, Merchant T, Brown VI, Sieger L, Siegel MJ, Raetz EA, Winick NJ, Loh ML, Carroll WL, Hunger SP. Dasatinib Plus Intensive Chemotherapy in Children, Adolescents, and Young Adults With Philadelphia Chromosome-Positive Acute Lymphoblastic Leukemia: Results of Children's Oncology Group Trial AALL0622. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 2018 Aug 1:36(22):2306-2314. doi: 10.1200/JCO.2017.76.7228. Epub 2018 May 29
[PubMed PMID: 29812996]
[28]
Jabbour E, Short NJ, Ravandi F, Huang X, Daver N, DiNardo CD, Konopleva M, Pemmaraju N, Wierda W, Garcia-Manero G, Sasaki K, Cortes J, Garris R, Khoury JD, Jorgensen J, Jain N, Alvarez J, O'Brien S, Kantarjian H. Combination of hyper-CVAD with ponatinib as first-line therapy for patients with Philadelphia chromosome-positive acute lymphoblastic leukaemia: long-term follow-up of a single-centre, phase 2 study. The Lancet. Haematology. 2018 Dec:5(12):e618-e627. doi: 10.1016/S2352-3026(18)30176-5. Epub
[PubMed PMID: 30501869]
[29]
Foà R, Bassan R, Vitale A, Elia L, Piciocchi A, Puzzolo MC, Canichella M, Viero P, Ferrara F, Lunghi M, Fabbiano F, Bonifacio M, Fracchiolla N, Di Bartolomeo P, Mancino A, De Propris MS, Vignetti M, Guarini A, Rambaldi A, Chiaretti S, GIMEMA Investigators. Dasatinib-Blinatumomab for Ph-Positive Acute Lymphoblastic Leukemia in Adults. The New England journal of medicine. 2020 Oct 22:383(17):1613-1623. doi: 10.1056/NEJMoa2016272. Epub
[PubMed PMID: 33085860]
[30]
Rousselot P, Coudé MM, Gokbuget N, Gambacorti Passerini C, Hayette S, Cayuela JM, Huguet F, Leguay T, Chevallier P, Salanoubat C, Bonmati C, Alexis M, Hunault M, Glaisner S, Agape P, Berthou C, Jourdan E, Fernandes J, Sutton L, Banos A, Reman O, Lioure B, Thomas X, Ifrah N, Lafage-Pochitaloff M, Bornand A, Morisset L, Robin V, Pfeifer H, Delannoy A, Ribera J, Bassan R, Delord M, Hoelzer D, Dombret H, Ottmann OG, European Working Group on Adult ALL (EWALL) group. Dasatinib and low-intensity chemotherapy in elderly patients with Philadelphia chromosome-positive ALL. Blood. 2016 Aug 11:128(6):774-82. doi: 10.1182/blood-2016-02-700153. Epub 2016 Apr 27
[PubMed PMID: 27121472]
[31]
Stock W, Luger SM, Advani AS, Yin J, Harvey RC, Mullighan CG, Willman CL, Fulton N, Laumann KM, Malnassy G, Paietta E, Parker E, Geyer S, Mrózek K, Bloomfield CD, Sanford B, Marcucci G, Liedtke M, Claxton DF, Foster MC, Bogart JA, Grecula JC, Appelbaum FR, Erba H, Litzow MR, Tallman MS, Stone RM, Larson RA. A pediatric regimen for older adolescents and young adults with acute lymphoblastic leukemia: results of CALGB 10403. Blood. 2019 Apr 4:133(14):1548-1559. doi: 10.1182/blood-2018-10-881961. Epub 2019 Jan 18
[PubMed PMID: 30658992]
[32]
Dunsmore KP, Winter SS, Devidas M, Wood BL, Esiashvili N, Chen Z, Eisenberg N, Briegel N, Hayashi RJ, Gastier-Foster JM, Carroll AJ, Heerema NA, Asselin BL, Rabin KR, Zweidler-Mckay PA, Raetz EA, Loh ML, Schultz KR, Winick NJ, Carroll WL, Hunger SP. Children's Oncology Group AALL0434: A Phase III Randomized Clinical Trial Testing Nelarabine in Newly Diagnosed T-Cell Acute Lymphoblastic Leukemia. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 2020 Oct 1:38(28):3282-3293. doi: 10.1200/JCO.20.00256. Epub 2020 Aug 19
[PubMed PMID: 32813610]
Level 1 (high-level) evidence
[33]
DeAngelo DJ, Stevenson KE, Dahlberg SE, Silverman LB, Couban S, Supko JG, Amrein PC, Ballen KK, Seftel MD, Turner AR, Leber B, Howson-Jan K, Kelly K, Cohen S, Matthews JH, Savoie L, Wadleigh M, Sirulnik LA, Galinsky I, Neuberg DS, Sallan SE, Stone RM. Long-term outcome of a pediatric-inspired regimen used for adults aged 18-50 years with newly diagnosed acute lymphoblastic leukemia. Leukemia. 2015 Mar:29(3):526-34. doi: 10.1038/leu.2014.229. Epub 2014 Jul 31
[PubMed PMID: 25079173]
[34]
Levato L, Molica S. Rituximab in the management of acute lymphoblastic leukemia. Expert opinion on biological therapy. 2018 Feb:18(2):221-226. doi: 10.1080/14712598.2018.1425389. Epub 2018 Jan 17
[PubMed PMID: 29310477]
Level 3 (low-level) evidence
[35]
Berry DH, Pullen J, George S, Vietti TJ, Sullivan MP, Fernbach D. Comparison of prednisolone, vincristine, methotrexate, and 6-mercaptopurine vs. vincristine and prednisone induction therapy in childhood acute leukemia. Cancer. 1975 Jul:36(1):98-102
[PubMed PMID: 1203854]
[36]
Sokal JE, Cox EB, Baccarani M, Tura S, Gomez GA, Robertson JE, Tso CY, Braun TJ, Clarkson BD, Cervantes F. Prognostic discrimination in "good-risk" chronic granulocytic leukemia. Blood. 1984 Apr:63(4):789-99
[PubMed PMID: 6584184]
[37]
Hasford J, Baccarani M, Hoffmann V, Guilhot J, Saussele S, Rosti G, Guilhot F, Porkka K, Ossenkoppele G, Lindoerfer D, Simonsson B, Pfirrmann M, Hehlmann R. Predicting complete cytogenetic response and subsequent progression-free survival in 2060 patients with CML on imatinib treatment: the EUTOS score. Blood. 2011 Jul 21:118(3):686-92. doi: 10.1182/blood-2010-12-319038. Epub 2011 May 2
[PubMed PMID: 21536864]
[38]
Pfirrmann M, Baccarani M, Saussele S, Guilhot J, Cervantes F, Ossenkoppele G, Hoffmann VS, Castagnetti F, Hasford J, Hehlmann R, Simonsson B. Prognosis of long-term survival considering disease-specific death in patients with chronic myeloid leukemia. Leukemia. 2016 Jan:30(1):48-56. doi: 10.1038/leu.2015.261. Epub 2015 Sep 29
[PubMed PMID: 26416462]
[39]
Yun S, Vincelette ND, Segar JM, Dong Y, Shen Y, Kim DW, Abraham I. Comparative Effectiveness of Newer Tyrosine Kinase Inhibitors Versus Imatinib in the First-Line Treatment of Chronic-Phase Chronic Myeloid Leukemia Across Risk Groups: A Systematic Review and Meta-Analysis of Eight Randomized Trials. Clinical lymphoma, myeloma & leukemia. 2016 Jun:16(6):e85-94. doi: 10.1016/j.clml.2016.03.003. Epub 2016 Mar 30
[PubMed PMID: 27101984]
Level 2 (mid-level) evidence
[40]
Cortes JE, Kim DW, Pinilla-Ibarz J, le Coutre PD, Paquette R, Chuah C, Nicolini FE, Apperley JF, Khoury HJ, Talpaz M, DeAngelo DJ, Abruzzese E, Rea D, Baccarani M, Müller MC, Gambacorti-Passerini C, Lustgarten S, Rivera VM, Haluska FG, Guilhot F, Deininger MW, Hochhaus A, Hughes TP, Shah NP, Kantarjian HM. Ponatinib efficacy and safety in Philadelphia chromosome-positive leukemia: final 5-year results of the phase 2 PACE trial. Blood. 2018 Jul 26:132(4):393-404. doi: 10.1182/blood-2016-09-739086. Epub 2018 Mar 22
[PubMed PMID: 29567798]
[41]
Réa D, Mauro MJ, Boquimpani C, Minami Y, Lomaia E, Voloshin S, Turkina A, Kim DW, Apperley JF, Abdo A, Fogliatto LM, Kim DDH, le Coutre P, Saussele S, Annunziata M, Hughes TP, Chaudhri N, Sasaki K, Chee L, García-Gutiérrez V, Cortes JE, Aimone P, Allepuz A, Quenet S, Bédoucha V, Hochhaus A. A phase 3, open-label, randomized study of asciminib, a STAMP inhibitor, vs bosutinib in CML after 2 or more prior TKIs. Blood. 2021 Nov 25:138(21):2031-2041. doi: 10.1182/blood.2020009984. Epub
[PubMed PMID: 34407542]
Level 1 (high-level) evidence
[42]
Costello J, Kang M, Banerji V. Frontline Treatment of the Young, Fit Patient with CLL: A Canadian Perspective. Current oncology (Toronto, Ont.). 2021 Sep 30:28(5):3825-3835. doi: 10.3390/curroncol28050326. Epub 2021 Sep 30
[PubMed PMID: 34677244]
Level 3 (low-level) evidence
[43]
Eichhorst B, Fink AM, Bahlo J, Busch R, Kovacs G, Maurer C, Lange E, Köppler H, Kiehl M, Sökler M, Schlag R, Vehling-Kaiser U, Köchling G, Plöger C, Gregor M, Plesner T, Trneny M, Fischer K, Döhner H, Kneba M, Wendtner CM, Klapper W, Kreuzer KA, Stilgenbauer S, Böttcher S, Hallek M, international group of investigators, German CLL Study Group (GCLLSG). First-line chemoimmunotherapy with bendamustine and rituximab versus fludarabine, cyclophosphamide, and rituximab in patients with advanced chronic lymphocytic leukaemia (CLL10): an international, open-label, randomised, phase 3, non-inferiority trial. The Lancet. Oncology. 2016 Jul:17(7):928-942. doi: 10.1016/S1470-2045(16)30051-1. Epub 2016 May 20
[PubMed PMID: 27216274]
Level 1 (high-level) evidence
[44]
Sharman JP, Egyed M, Jurczak W, Skarbnik A, Pagel JM, Flinn IW, Kamdar M, Munir T, Walewska R, Corbett G, Fogliatto LM, Herishanu Y, Banerji V, Coutre S, Follows G, Walker P, Karlsson K, Ghia P, Janssens A, Cymbalista F, Woyach JA, Salles G, Wierda WG, Izumi R, Munugalavadla V, Patel P, Wang MH, Wong S, Byrd JC. Acalabrutinib with or without obinutuzumab versus chlorambucil and obinutuzmab for treatment-naive chronic lymphocytic leukaemia (ELEVATE TN): a randomised, controlled, phase 3 trial. Lancet (London, England). 2020 Apr 18:395(10232):1278-1291. doi: 10.1016/S0140-6736(20)30262-2. Epub
[PubMed PMID: 32305093]
Level 1 (high-level) evidence
[45]
Al-Sawaf O, Zhang C, Tandon M, Sinha A, Fink AM, Robrecht S, Samoylova O, Liberati AM, Pinilla-Ibarz J, Opat S, Sivcheva L, Le Dû K, Fogliatto LM, Niemann CU, Weinkove R, Robinson S, Kipps TJ, Tausch E, Schary W, Ritgen M, Wendtner CM, Kreuzer KA, Eichhorst B, Stilgenbauer S, Hallek M, Fischer K. Venetoclax plus obinutuzumab versus chlorambucil plus obinutuzumab for previously untreated chronic lymphocytic leukaemia (CLL14): follow-up results from a multicentre, open-label, randomised, phase 3 trial. The Lancet. Oncology. 2020 Sep:21(9):1188-1200. doi: 10.1016/S1470-2045(20)30443-5. Epub
[PubMed PMID: 32888452]
Level 1 (high-level) evidence
[46]
Moreno C, Greil R, Demirkan F, Tedeschi A, Anz B, Larratt L, Simkovic M, Samoilova O, Novak J, Ben-Yehuda D, Strugov V, Gill D, Gribben JG, Hsu E, Lih CJ, Zhou C, Clow F, James DF, Styles L, Flinn IW. Ibrutinib plus obinutuzumab versus chlorambucil plus obinutuzumab in first-line treatment of chronic lymphocytic leukaemia (iLLUMINATE): a multicentre, randomised, open-label, phase 3 trial. The Lancet. Oncology. 2019 Jan:20(1):43-56. doi: 10.1016/S1470-2045(18)30788-5. Epub 2018 Dec 3
[PubMed PMID: 30522969]
Level 1 (high-level) evidence
[47]
Cheung MC, Mittmann N, Owen C, Abdel-Samad N, Fraser GAM, Lam S, Crump M, Sperlich C, van der Jagt R, Prica A, Couban S, Woyach JA, Ruppert AS, Booth AM, Mandrekar SJ, McDonald G, Shepherd LE, Yen H, Chen BE, Hay AE. A Prospective Economic Analysis of Early Outcome Data From the Alliance A041202/ CCTG CLC.2 Randomized Phase III Trial Of Bendamustine-Rituximab Compared With Ibrutinib-Based Regimens in Untreated Older Patients With Chronic Lymphocytic Leukemia. Clinical lymphoma, myeloma & leukemia. 2021 Nov:21(11):766-774. doi: 10.1016/j.clml.2021.06.011. Epub 2021 Jul 3
[PubMed PMID: 34334330]
Level 1 (high-level) evidence
[48]
Howard SC, Trifilio S, Gregory TK, Baxter N, McBride A. Tumor lysis syndrome in the era of novel and targeted agents in patients with hematologic malignancies: a systematic review. Annals of hematology. 2016 Mar:95(4):563-73. doi: 10.1007/s00277-015-2585-7. Epub 2016 Jan 12
[PubMed PMID: 26758269]
Level 1 (high-level) evidence
[49]
Freifeld AG, Bow EJ, Sepkowitz KA, Boeckh MJ, Ito JI, Mullen CA, Raad II, Rolston KV, Young JA, Wingard JR, Infectious Diseases Society of Americaa. Clinical practice guideline for the use of antimicrobial agents in neutropenic patients with cancer: 2010 Update by the Infectious Diseases Society of America. Clinical infectious diseases : an official publication of the Infectious Diseases Society of America. 2011 Feb 15:52(4):427-31. doi: 10.1093/cid/ciq147. Epub 2011 Jan 4
[PubMed PMID: 21205990]
Level 1 (high-level) evidence