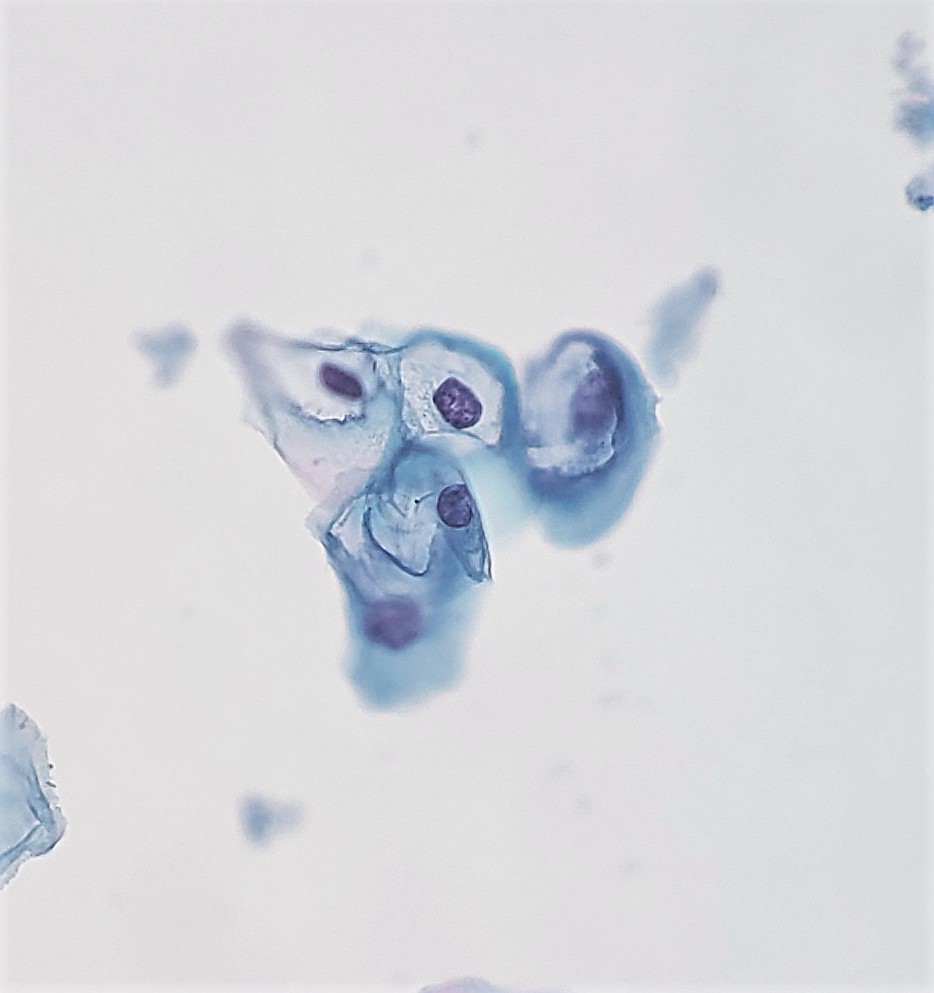
[1]
Koss LG. The 57th birthday of koilocytes. Cancer cytopathology. 2012 Dec 25:120(6):421. doi: 10.1002/cncy.21231. Epub 2012 Aug 23
[PubMed PMID: 22927183]
[2]
Zhu Y, Wang Y, Hirschhorn J, Welsh KJ, Zhao Z, Davis MR, Feldman S. Human Papillomavirus and Its Testing Assays, Cervical Cancer Screening, and Vaccination. Advances in clinical chemistry. 2017:81():135-192. doi: 10.1016/bs.acc.2017.01.004. Epub 2017 Mar 18
[PubMed PMID: 28629588]
Level 3 (low-level) evidence
[3]
Krawczyk E, Suprynowicz FA, Liu X, Dai Y, Hartmann DP, Hanover J, Schlegel R. Koilocytosis: a cooperative interaction between the human papillomavirus E5 and E6 oncoproteins. The American journal of pathology. 2008 Sep:173(3):682-8. doi: 10.2353/ajpath.2008.080280. Epub 2008 Aug 7
[PubMed PMID: 18688031]
[4]
Lawson JS, Glenn WK, Heng B, Ye Y, Tran B, Lutze-Mann L, Whitaker NJ. Koilocytes indicate a role for human papilloma virus in breast cancer. British journal of cancer. 2009 Oct 20:101(8):1351-6. doi: 10.1038/sj.bjc.6605328. Epub 2009 Sep 22
[PubMed PMID: 19773762]
[5]
Syrjänen KJ. Two Landmark Studies Published in 1976/1977 Paved the Way for the Recognition of Human Papillomavirus as the Major Cause of the Global Cancer Burden. Acta cytologica. 2017:61(4-5):316-337. doi: 10.1159/000477372. Epub 2017 Jul 11
[PubMed PMID: 28693008]
[6]
Orita Y, Gion Y, Tachibana T, Ikegami K, Marunaka H, Makihara S, Yamashita Y, Miki K, Makino T, Akisada N, Akagi Y, Kimura M, Yoshino T, Nishizaki K, Sato Y. Laryngeal squamous cell papilloma is highly associated with human papillomavirus. Japanese journal of clinical oncology. 2018 Apr 1:48(4):350-355. doi: 10.1093/jjco/hyy009. Epub
[PubMed PMID: 29447361]
[7]
Groves IJ, Coleman N. Pathogenesis of human papillomavirus-associated mucosal disease. The Journal of pathology. 2015 Mar:235(4):527-38. doi: 10.1002/path.4496. Epub 2015 Jan 20
[PubMed PMID: 25604863]
[8]
Syrjänen S, Rautava J, Syrjänen K. HPV in Head and Neck Cancer-30 Years of History. Recent results in cancer research. Fortschritte der Krebsforschung. Progres dans les recherches sur le cancer. 2017:206():3-25
[PubMed PMID: 27699526]
[9]
Bean SM, Chhieng DC. Anal-rectal cytology: a review. Diagnostic cytopathology. 2010 Jul:38(7):538-46. doi: 10.1002/dc.21242. Epub
[PubMed PMID: 19941374]
[10]
Aggarwal S, Arora VK, Gupta S, Singh N, Bhatia A. Koilocytosis: correlations with high-risk HPV and its comparison on tissue sections and cytology, urothelial carcinoma. Diagnostic cytopathology. 2009 Mar:37(3):174-7. doi: 10.1002/dc.20978. Epub
[PubMed PMID: 19170170]
[11]
Meisels A, Fortin R. Condylomatous lesions of the cervix and vagina. I. Cytologic patterns. Acta cytologica. 1976 Nov-Dec:20(6):505-9
[PubMed PMID: 1069445]
[12]
Fletcher S. Histopathology of papilloma virus infection of the cervix uteri: the history, taxonomy, nomenclature and reporting of koilocytic dysplasias. Journal of clinical pathology. 1983 Jun:36(6):616-24
[PubMed PMID: 6304149]
[13]
Purola E, Savia E. Cytology of gynecologic condyloma acuminatum. Acta cytologica. 1977 Jan-Feb:21(1):26-31
[PubMed PMID: 264754]
[14]
Pinto AP, Crum CP, Hirsch MS. MOLECULAR MARKERS OF EARLY CERVICAL NEOPLASIA. Diagnostic histopathology (Oxford, England). 2010 Oct 1:16(10):445-454
[PubMed PMID: 21076641]
[15]
Sato S, Chiba H, Shikano K, Horiguchi M, Wada Y, Yajima A, Okagaki T. [Ultrastructural observation of human papillomavirus particles in the uterine cervix intraepithelial neoplasia]. Gan no rinsho. Japan journal of cancer clinics. 1988 Jul:34(8):993-1000
[PubMed PMID: 2841511]
[16]
Toki T, Oikawa N, Tase T, Sato S, Wada Y, Yajima A, Higashiiwai H. Immunohistochemical and electron microscopic demonstration of human papillomavirus in dysplasia of the uterine cervix. The Tohoku journal of experimental medicine. 1986 Jun:149(2):163-7
[PubMed PMID: 3018963]
[17]
Saslow D, Solomon D, Lawson HW, Killackey M, Kulasingam SL, Cain J, Garcia FA, Moriarty AT, Waxman AG, Wilbur DC, Wentzensen N, Downs LS Jr, Spitzer M, Moscicki AB, Franco EL, Stoler MH, Schiffman M, Castle PE, Myers ER, ACS-ASCCP-ASCP Cervical Cancer Guideline Committee. American Cancer Society, American Society for Colposcopy and Cervical Pathology, and American Society for Clinical Pathology screening guidelines for the prevention and early detection of cervical cancer. CA: a cancer journal for clinicians. 2012 May-Jun:62(3):147-72. doi: 10.3322/caac.21139. Epub 2012 Mar 14
[PubMed PMID: 22422631]
[18]
Abreu AL, Souza RP, Gimenes F, Consolaro ME. A review of methods for detect human Papillomavirus infection. Virology journal. 2012 Nov 6:9():262. doi: 10.1186/1743-422X-9-262. Epub 2012 Nov 6
[PubMed PMID: 23131123]
[19]
Kruse AJ, Baak JP, Helliesen T, Kjellevold KH, Robboy SJ. Prognostic value and reproducibility of koilocytosis in cervical intraepithelial neoplasia. International journal of gynecological pathology : official journal of the International Society of Gynecological Pathologists. 2003 Jul:22(3):236-9
[PubMed PMID: 12819389]
Level 2 (mid-level) evidence
[20]
Jeronimo J, Massad LS, Schiffman M, National Institutes of Health/American Society for Colposcopy and Cervical Pathology (NIH/ASCCP) Research Group. Visual appearance of the uterine cervix: correlation with human papillomavirus detection and type. American journal of obstetrics and gynecology. 2007 Jul:197(1):47.e1-8
[PubMed PMID: 17618753]
[21]
Rintala MA, Grénman SE, Puranen MH, Isolauri E, Ekblad U, Kero PO, Syrjänen SM. Transmission of high-risk human papillomavirus (HPV) between parents and infant: a prospective study of HPV in families in Finland. Journal of clinical microbiology. 2005 Jan:43(1):376-81
[PubMed PMID: 15634997]
[22]
Sandstrom RE. Regarding: Koilocytes indicate a role for human papilloma virus in breast cancer. British journal of cancer. 2010 Feb 16:102(4):786-7; author reply 788. doi: 10.1038/sj.bjc.6605549. Epub 2010 Feb 2
[PubMed PMID: 20125157]
[23]
Nascimento ADMTD, Pinho JD, Júnior AALT, Larges JS, Soares FM, Calixto JRR, Coelho RWP, Belfort MRC, Nogueira LR, da Cunha IW, Silva GEB. Angiolymphatic invasion and absence of koilocytosis predict lymph node metastasis in penile cancer patients and might justify prophylactic lymphadenectomy. Medicine. 2020 Feb:99(9):e19128. doi: 10.1097/MD.0000000000019128. Epub
[PubMed PMID: 32118716]
[24]
Takamoto D, Kawahara T, Kasuga J, Sasaki T, Yao M, Yumura Y, Uemura H. The analysis of human papillomavirus DNA in penile cancer tissue by in situ hybridization. Oncology letters. 2018 May:15(5):8102-8106. doi: 10.3892/ol.2018.8351. Epub 2018 Mar 26
[PubMed PMID: 29731917]
Level 3 (low-level) evidence