Continuing Education Activity
The anterolateral thigh (ALT) flap is a versatile flap with broad indications, and it can cover almost any defect in the body from head to foot. It has become a workhorse flap in reconstructive plastic surgery and has a very high success rate of more than 95% when performed by an experienced team. This activity outlines the use of ALT flaps in reconstructive microsurgery and highlights the role of the interprofessional team in managing patients with difficult and chronic non-healing wounds.
Objectives:
- Review the anatomy of the anterolateral thigh flap and outline its indications for use in reconstructive microsurgery.
- Describe the equipment, personnel, preparation, and technique in the harvest of the anterolateral thigh flap.
- Summarize the potential complications and clinical significance of the anterolateral thigh flap.
- Outline interprofessional team strategies for improving care coordination and communication in the management of difficult and chronic wounds, and improve patient outcomes following anterolateral thigh flap reconstructive surgery.
Introduction
The anterolateral thigh (ALT) flap, which was first described by Song (1984), is now well-established in reconstructive microsurgery as a workhorse flap. It is a versatile flap with many excellent attributes that include long vascular pedicle with adequate vessel diameter, ability to harvest large areas of skin without added donor site morbidity, adaptability for use as a sensate flap, use as a flow-through flap for vascular gaps in the extremities, flexibility of the flap to be folded for use as a double skin paddle, and use as a chimeric flap to reconstruct composite soft tissue defects almost anywhere in the body. A two-team approach permits simultaneous flap harvest and resection/debridement at the recipient area. The ALT flap can also be used for breast reconstruction if lower abdominal skin is unavailable due to previous scars or surgeries.[1][2]
The ALT flap is relatively easy to harvest if the principles of perforator dissection are followed. The vascularity of the flap is reliable even though there is some variability in its perforator anatomy.[3][4][5]
Anatomy and Physiology
Anterolateral Thigh Flap
The anterolateral thigh flap is classically described as a fasciocutaneous perforator flap based on the septocutaneous perforator vessels, or more predomintantly, the musculocutaneous perforator vessels that arise from the descending branch of the lateral circumflex femoral artery (LCFA), the largest branch of the profunda femoris system in the thigh. Early anatomical dissections described the ALT pedicle as predominantly consisting of septocutaneous perforators; however, recent research suggests the contrary. Now, it is well accepted that musculocutaneous perforators comprise the predominant blood supply (87%) to this flap.[3][4][5][6]
The vascular territory of the ALT flap extends from the anterior superior iliac spine superiorly to the lateral femoral condyle inferiorly and from the medial edge of the rectus femoris muscle anteriorly to the iliopubic tract posteriorly.
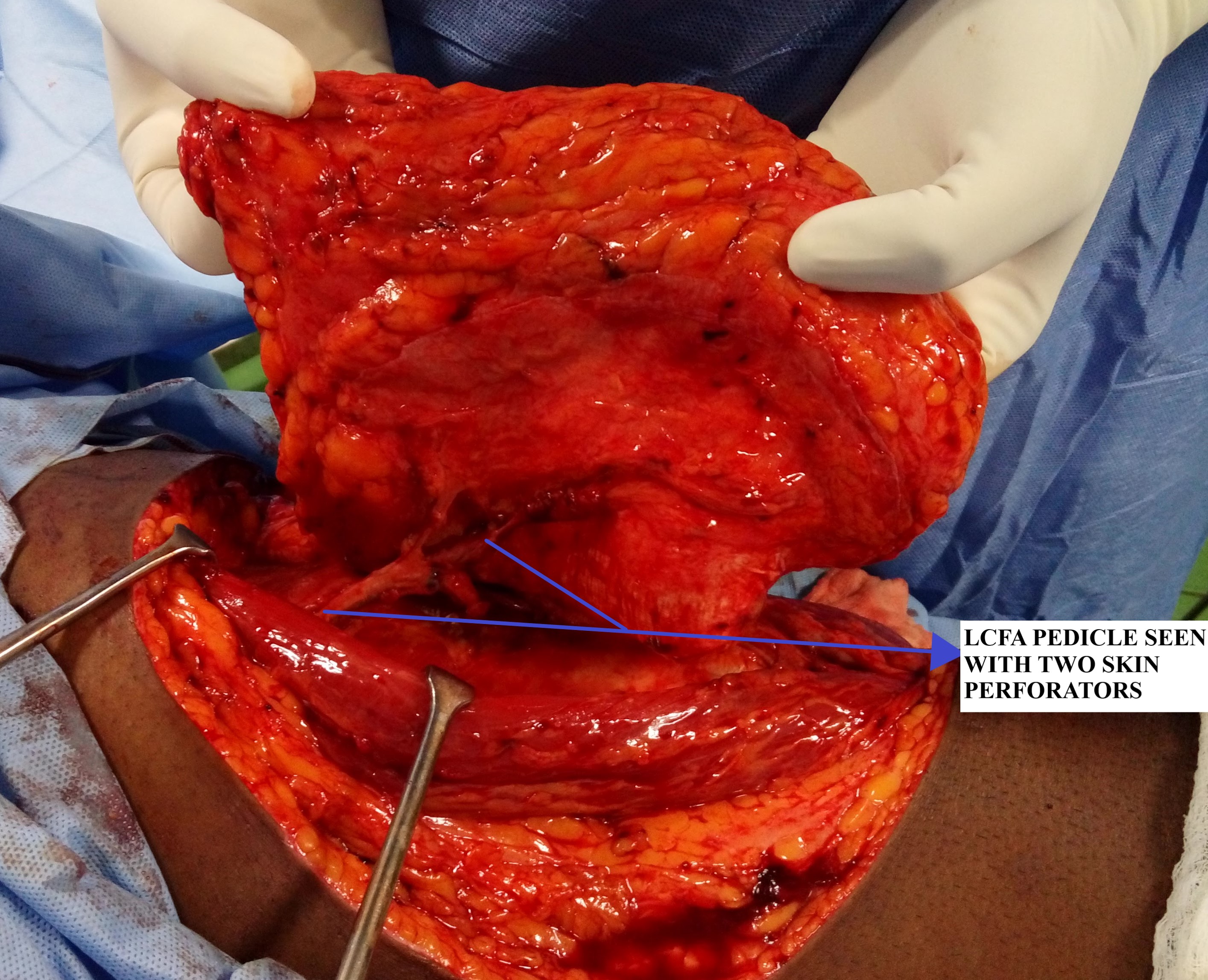
The LCFA, after arising from the profunda femoris artery, travels deep to the rectus femoris and divides into three branches: ascending, transverse, and descending. The descending branch of the LCFA travels along the medial edge of vastus lateralis muscle in the intermuscular septum, giving off perforators that supply the anterolateral thigh skin. The ALT flap provides a long pedicle (8 to 16 cm) with adequate vessel lumen diameter (2 to 2.5 mm). The flap drains via two venae comitantes that accompany the arterial pedicle. The ALT flap can also be harvested as a sensate flap by including the lateral femoral cutaneous nerve in the flap design, or even as a functional muscle flap by including the branch of the femoral nerve that innervates the vastus lateralis.[1][3][4][5][6]
Lateral Thigh Flap
The lateral thigh flap is perfused by a pedicle based on the LCFA, mainly the ascending and transverse branches (tensor fasciae latae perforator vessels). This flap is classically employed for the reconstruction of ischial and trochanteric pressure sores. It can also be based on the ALT perforator vessels (descending branch of LCFA) or applied to perineal and abdominal wall reconstruction as a pedicled flap.
Indications
The ALT flap is versatile and has a wide range of indications, including:
1. Head and Neck Reconstruction: following resection of head and neck tumors, the ALT flap can be used to resurface the defect either as a cover or as double skin paddle with cover and lining. The ALT flap is an alternative to the radial forearm free flap in patients with low body mass index, but in larger patients, the flap is bulky due to subcutaneous adipose tissue and can be used to fill deeper defects. An unique application of the ALT flap is in esophageal reconstruction, wherein it is folded onto itself (skin inside to form a lumen) as a tubed flap.[1][7][8][9]
2. Secondary Burn Reconstruction: following sequelae of burn injuries like extensive scar contractures, the ALT flap can be used after the release of extensive post-burn contractures to help prevent recurrence.[10]
3. Breast Reconstruction: it can also be used for breast reconstruction if lower abdominal skin is unavailable due to previous scars or surgeries (abdominoplasties, laparotomy) or as a salvage option following failure of primary breast reconstruction.[11][12]
4. Abdominal Wall Reconstruction: it can be used as a pedicled flap for lower abdominal reconstruction, wherein it is harvested as a myocutaneous flap or a composite flap with fascia (tensor fasciae latae). For more significant and extensive defects of the abdominal wall, free ALT transfer is preferred. These flaps can include multiple tissue components, such as muscle and/or fascia (fascia lata), using the chimeric flap principle.[13][14]
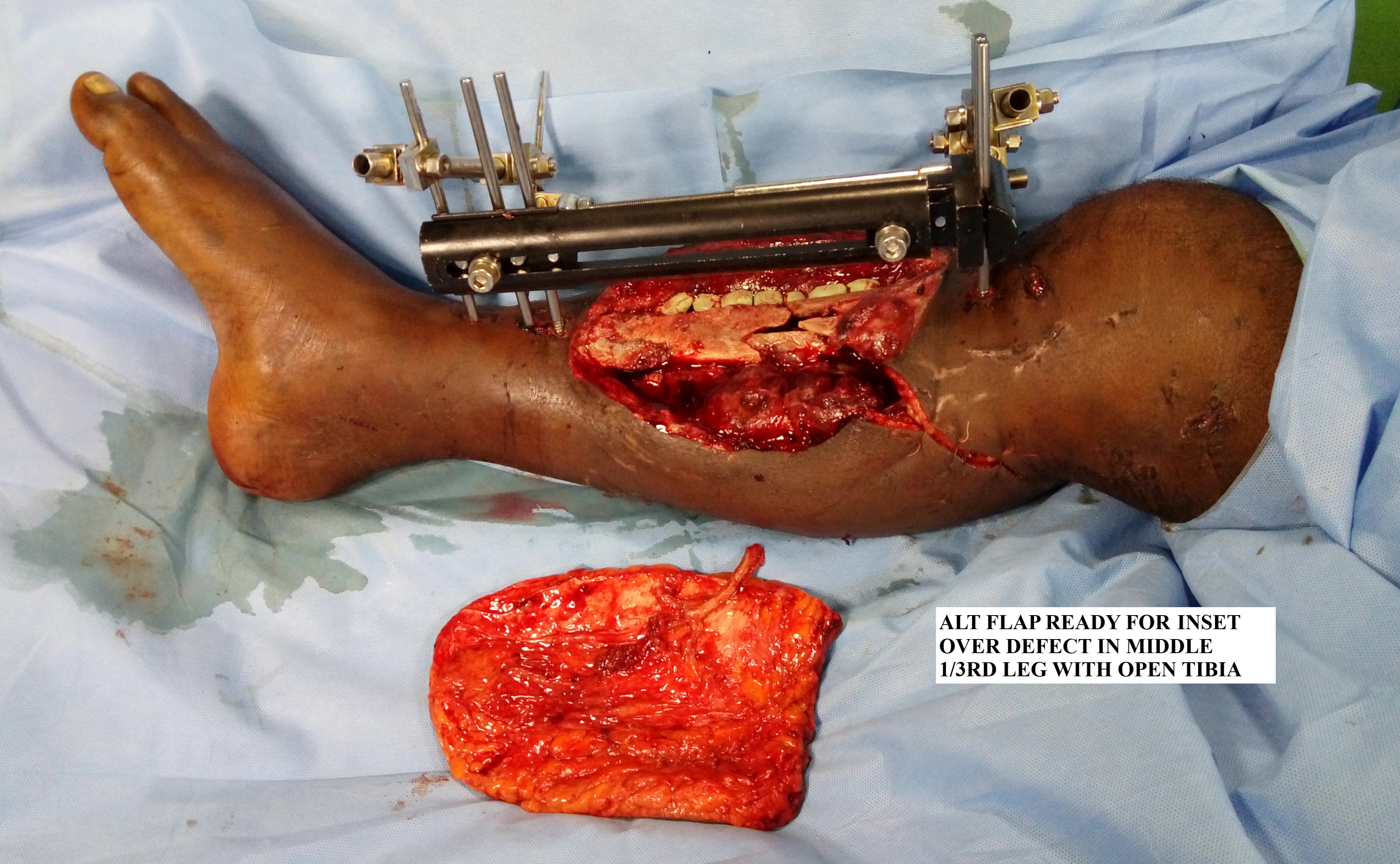
5. Upper and Lower Extremity Reconstruction: posttraumatic defects around the knee can be resurfaced using the distally-based ALT flap. For defects involving the lower one-third of the leg and the foot, a thinned ALT flap may be used. It can also be used as a flow-through flap for resurfacing defects in the lower limb while maintaining blood supply in an ischaemic leg. Similarly, it can be used for extensive defects in the upper extremity. When tendon gliding is paramount, the ALT can be harvested as a composite flap by including fascia lata to facilitate this.[15][16][17]
6. Perineum and penile reconstruction: a proximally-based ALT flap can be used for perineal reconstruction. The free ALT flap has also been used in gender reassignment surgery (female to male) for phalloplasty.[18][19]
Contraindications
There are no absolute contraindications to the use of the anterolateral flap. The relative contraindications to its use include:
- Morbid obesity
- A severe peripheral occlusive arterial disease that makes the circulation to the flap unreliable
- Previous trauma or scarring of the anterolateral thigh skin
- Extensive medical co-morbidities (patient is unfit for long surgeries)[20]
Equipment
The equipment required for flap harvest is the same as that for any free flap microsurgical case. Pre-operatively: sterile plastic sheet/drape to make a template of the defect, hand Doppler, and skin markers for marking of the perforators and the flap. Intra-operatively, surgeons will require loupes for flap dissection and an operating microscope with good optics for microsurgical anastomosis.
Instruments for flap harvest include standard plastic surgical instruments like skin hooks, self-retaining retractors, Adson forceps, Debakey forceps, fine tenotomy scissors, vessel clips, skin dermatome, and mesher. Microsurgical instruments: jeweler forceps, Castroviejo needle holder, vessel dilator forceps, microscissors (both curved and straight), microvascular clamps (both arterial and venous clamps), and micro sutures (8-0, 9-0, and 10-0 nylon sutures on tapered needles).
Heparin saline solution, 1% or 2% plain lidocaine solution, and papaverine solution are required for intraoperative irrigation of vessels to prevent vasospasm. Post-operative dressing materials required include plaster of Paris (POP) slabs, burn gauze, soft bandages and wool, elastic-crepe bandages, and sterile/antiseptic dressings.
Personnel
A two-team approach is usually feasible during the harvest of ALT flaps, depending on the case. If used in lower extremity reconstruction, the primary surgeon can harvest the flap; simultaneously, the junior surgeon or colleague can prepare the recipient bed, including exposure of the vessels for microvascular anastomosis. This approach requires two scrub nurses and one circulating nurse to monitor flap 'off and on' times and tourniquet times, if a tourniquet is used. An anesthetist and an anesthesia technician are also required. Postoperatively, the patient will require monitoring of the flap for at least 5-7 days by ward nurses trained in free flap monitoring.
Preparation
The flap harvest can be performed under general anesthesia with the patient in a supine position. The thickness of the thigh skin is assessed preoperatively by doing a skin pinch test, which gives an idea of whether the donor site will close primarily or require a split thickness skin graft from the contralateral thigh. In patients with peripheral vascular disease, a CT angiogram can be used to assess the vascular flow to the limb and the availability of perforators.
Before starting the procedure, the hair of both thighs is clipped. A critical point to note is that the foot and leg should be in a neutral position with both pointing towards the ceiling; this is mandatory for flap harvest, and rotation should be avoided at all costs.
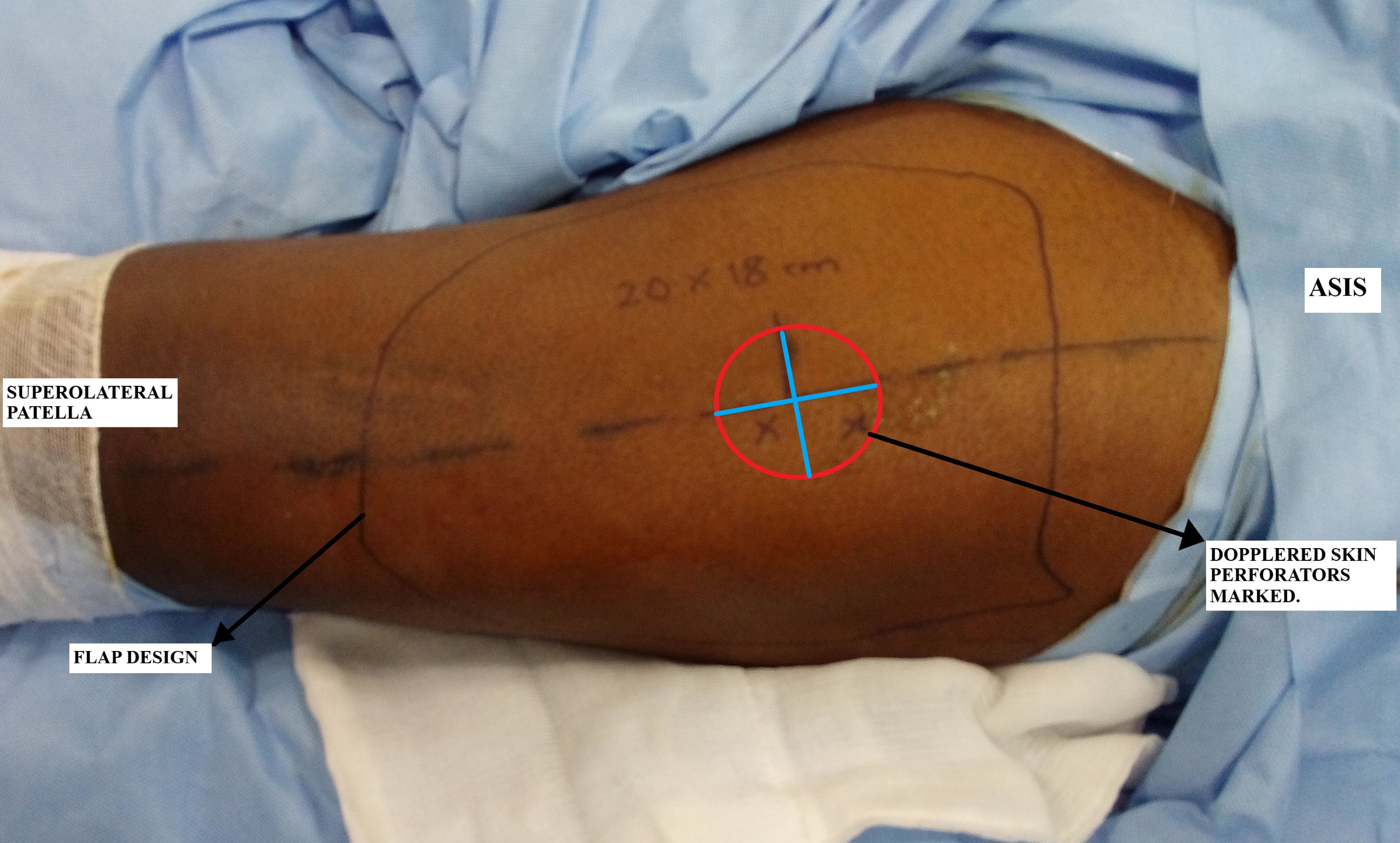
Preoperative flap marking is done by drawing a line from the anterior superior iliac spine (ASIS) to the superolateral border of the patella, which represents the septum between the rectus femoris and vastus lateralis muscles, and serves as the longitudinal axis of the flap. The midway point on this line is marked, and a circle with a 3 cm radius is drawn around it. Most perforators will be found in the outer, lower quadrant of this circle. Using a handheld Doppler, the dominant cutaneous perforators are identified and marked, and the flap is designed according to the defect size. The flap can be planned with perforators in a central position or in an eccentric position; the latter yields a greater pedicle length. Finally, prepping and draping is accomplished to facilitate a two-team approach to simultaneous flap harvest and recipient site preparation.
The flap is designed and marked to facilitate longitudinal closure of the donor site, but if the recipient defect is large, flaps up to 35 cm x 25 cm can be harvested based on a single perforator; these larger donor site defects are more likely to require skin grafting for closure.[1][2][21]
Technique or Treatment
The ALT flap can be harvested as a skin flap (skin and subcutaneous tissue) using a subfascial dissection technique to yield thin or ultrathin flaps, if necessary. It can also be harvested as a fasciocutaneous flap, which is the classically described technique and also the most commonly performed; it can be harvested as a musculocutaneous flap by including the vastus lateralis or even as a fascia only flap. It can also be harvested as a chimeric flap by adding various other tissues, each with its independent vascular supply (including rectus femoris, tensor fasciae latae, or anteromedial thigh skin along with ALT skin).[1][2][6][21]
Step 1 - Flap and perforator dissection: Flap dissection can be performed either in a suprafacial or subfascial plane, depending on the defect. Generally, the subfascial dissection technique is a more straightforward way to identify the cutaneous perforators supplying the flap; it is more commonly performed, and allows the surgeon to map the perforator anatomy and tailor the flap accordingly.
The incision is made on the medial border of the flap and carried down to the fascia. Once the subfascial plane is entered, the septum between the rectus femoris and vastus lateralis is identified as a yellow fat stripe. It represents the site where septocutaneous perforators may emerge. If no septocutaneous perforator vessels are encountered, then dissection is carried out laterally in the subfascial plane to look for musculocutaneous perforators arising from the vastus lateralis. All potential perforators are identified, and one or two vessels with larger calibers and strong pulses are chosen; they will become the primary supply to the flap.
Step 2 - Pedicle dissection and isolation: Once the perforator anatomy is confirmed, the rectus femoris is retracted medially to expose the septum entirely and trace the course of LCFA. The next step involves either a straightforward dissection, in the case of septocutaneous perforators, or a tedious dissection wherein the musculocutaneous perforators are traced proximally by painstakingly deroofing the vastus lateralis. During the intramuscular perforator dissection, numerous small muscular branches will be encountered, especially on the lateral and posterior sides of the perforator, and these vessels will have to be ligated. Dissection proceeds until the selected perforators' take-off from the descending branch of LCFA is reached. Alternatively, some muscle can be included with the flap, thereby avoiding this time-consuming and challenging step of perforator isolation. The motor nerve to vastus lateralis accompanies the descending branch of LCFA and must be preserved, although branches entering any muscle harvested may be included. Further proximal dissection of the LCFA is carried out depending on the length of the pedicle required for anastomosis with the recipient vessels. Another critical point to note in the proximal dissection is that the lateral femoral cutaneous nerve is usually found proximally along a line connecting the ASIS to the superolateral patellar border. This nerve must be preserved if not harvesting a sensory flap.
Step 3 - Flap modification/thining: When thin flaps are required for resurfacing certain areas like the dorsum of the foot, hand defects, and neck defects, primary defatting can be performed. The flap pedicle must remain intact during the defatting to ensure the flap remains viable throughout the process. The defatting must progress uniformly from the deeper larger fat globule layer to the superficial smaller fat globule layer, preserving an adequate cuff of tissue around the pedicle (at least 2 cm thickness) to prevent vascular injury and flap compromise. Flap thinning is predicated on preservation of the subdermal plexus, which is perfused by perforators supplying the flap, and in turn perfuses the skin.
Step 4 - Pedicle division & microvascular anastomosis: After adequate thinning of the flap, the next step is flap pedicle division, and the time is noted ("flap off" or "ischemia" time). Then the flap is transferred to the defect and temporarily inset with a few stay sutures in order to permit proper orientation of the flap pedicle and to prevent twisting of the vessels should the flap shift. The microvascular anastomosis is sewn between the flap vessels and the recipient vessels using 8-0, 9-0 or 10-0 nylon suture under the operating microscope. After completion of vessel anastomosis ('flap on' time noted), the adequacy of flow and any anastomotic site leaks should be evaluated. Flap perfusion and viability should also be confirmed by observing the color, warmth, and bleeding from edges of the flap.
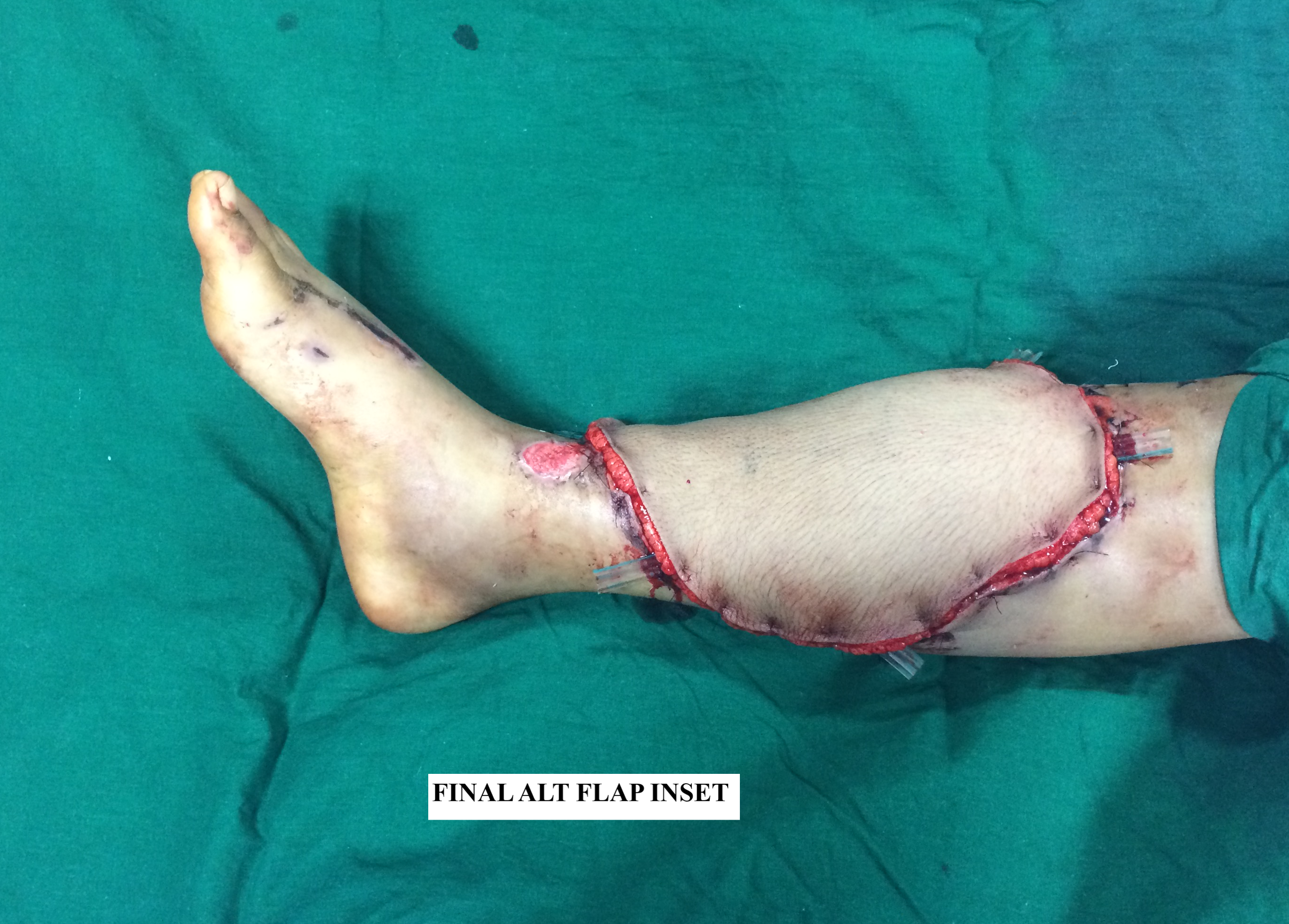
Step 5 - Flap inset: After confirmation of vessel flow and flap perfusion, the final inset of the flap is performed. Suction drains or corrugated rubber drains are inserted beneath the flap. The site of the anastomosis is marked on the flap for postoperative monitoring. Dressings and a POP slab are applied with a 'flap window' for postoperative flap monitoring. Plaster of Paris is not required for head and neck applications of the ALT flap. Tight compressive dressings are avoided in order to prevent flap pedicle compromise.
Step 6 - Donor site closure: The donor site can be closed primarily, or if the donor defect is significant and primary closure would require excessive tension, a split thickness skin graft (STSG) can be applied. A vacuum-assisted closure (VAC) dressing can be applied over the donor site closure, especially if a skin graft is used; use of a VAC dressing improves take of skin grafts and helps to prevent wound dehiscence in primary closure.
In cases when ALT perforators are absent or not of good caliber (2%), there are four alternative options:
- Proximal dissection and exploration of the transverse branch of LCFA and harvest of the tensor fasciae latae perforator flap.
- Exploration of the rectus femoris branch (medial branch of descending LCFA) and raising of the anteromedial thigh flap.
- Elevation of a free vastus lateralis muscle flap.
- Explore the contralateral thigh or abandon the procedure and look for alternative donor sites.[1][2][6][20][22][23][24]
Complications
The most dreaded complication following a free flap transfer is flap failure, hence the importance of postoperative flap monitoring. The causes of flap failure include:
- Venous or arterial insufficiency, more often venous (8-9:1)
- Thrombosis
- Venous congestion
- Twisting of the pedicle
- Compression of the pedicle
- Tension at flap edges leading to marginal flap loss
Other complications include:
- Color mismatch between flap and recipient site
- Excessively bulky flaps leading to fat necrosis
- Hematoma
- Seroma
- Infection
- Delayed wound healing
- Wound dehiscence
- Hypertrophic scarring of the donor site
- Failure of skin graft take over the donor site and sensory loss over the donor thigh[25]
Clinical Significance
The success rate of anterolateral thigh flap transfer is higher than 95%, and the ALT flap has become a workhorse of reconstructive microsurgery, with broad indications for reconstructing defects from head to foot. The advantages of using the ALT flap include:
- Ease of harvest due to reliable anatomy
- Long and large calibre vascular pedicle
- Versatility in flap modification like flap thinning or harvesting of chimeric flaps, depending on the donor site requirement
- Ability to use as a sensate flap
- Little donor site morbidity
- The use of a two-team approach, which decreases operating time[26][27]
Enhancing Healthcare Team Outcomes
The anterolateral thigh flap has become an essential part of the plastic surgeon's armamentarium, and its widespread use involves a team approach wherein the plastic surgeon plays an important role in the management of a multispecialty problem. The ALT flap can be used to cover difficult open complex lower extremity fractures, which will involve the plastic surgeon, orthopedic surgeon, anesthetist, physical medicine specialist, and physiotherapist. This orthoplastic approach in extremity reconstruction involves the simultaneous application of the principles and practice of orthopedic and plastic surgery to enhance and optimize patient outcomes following limb reconstructive surgery.
Similarly, in head and neck reconstruction where a multidisciplinary team approach is essential to prevent the locoregional and distant spread of tumors, the concept of wide local excision combined with immediate reconstruction with free flaps and adjuvant chemoradiation is gaining popularity. In these cases, the ablative and reconstructive surgeon are often one and the same, although plastic surgeons, facial plastic surgeons, and oral-maxillofacial surgeons are often called upon by the head and neck surgical oncologist or otorhinolaryngologist. [Level 3]
Nursing, Allied Health, and Interprofessional Team Monitoring
One of the most important factors contributing to the success of free flap surgery is timely intervention in the case of early complications, especially flap failure; to that end, recognition of complications through proper monitoring and good postoperative care is critical. The role of ward nurses and other allied staff is critical to increase the likelihood that patients who undergo ALT flap reconstructions experience good outcomes. To ensure these goals are met, there should be a good understanding and coordination among the members of the plastic and reconstructive surgery team, with each member having a clear understanding of his or her own roles, responsibilities, and limitations.
Postoperative monitoring of ALT free flaps is most critical during the first 24 to 72 hours and should be performed diligently until at least the first week after surgery. Proper monitoring improves salvage rates of failing free flaps, as there is an inverse relationship between the length of time a failed flap takes to get to the operating room and its chance of survival. Hourly flap monitoring by nurses includes: color, warmth, turgor, capillary refill, handheld doppler signals from flap pedicle, signs of hematoma, bleeding, and checking for compression or pressure over the flap pedicle. During the initial 48-72 hours, hourly flap monitoring is mandatory. Once the danger period has passed, monitoring the flap every three to four hours for a few days should be sufficient.