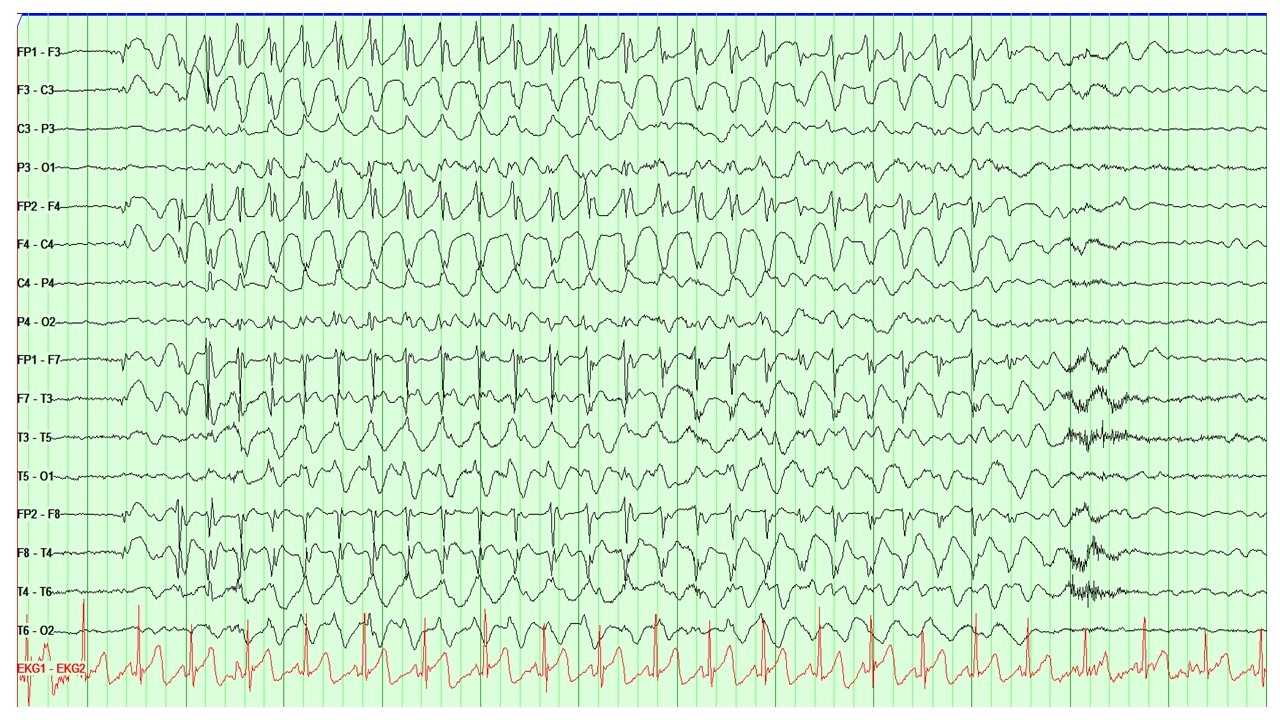
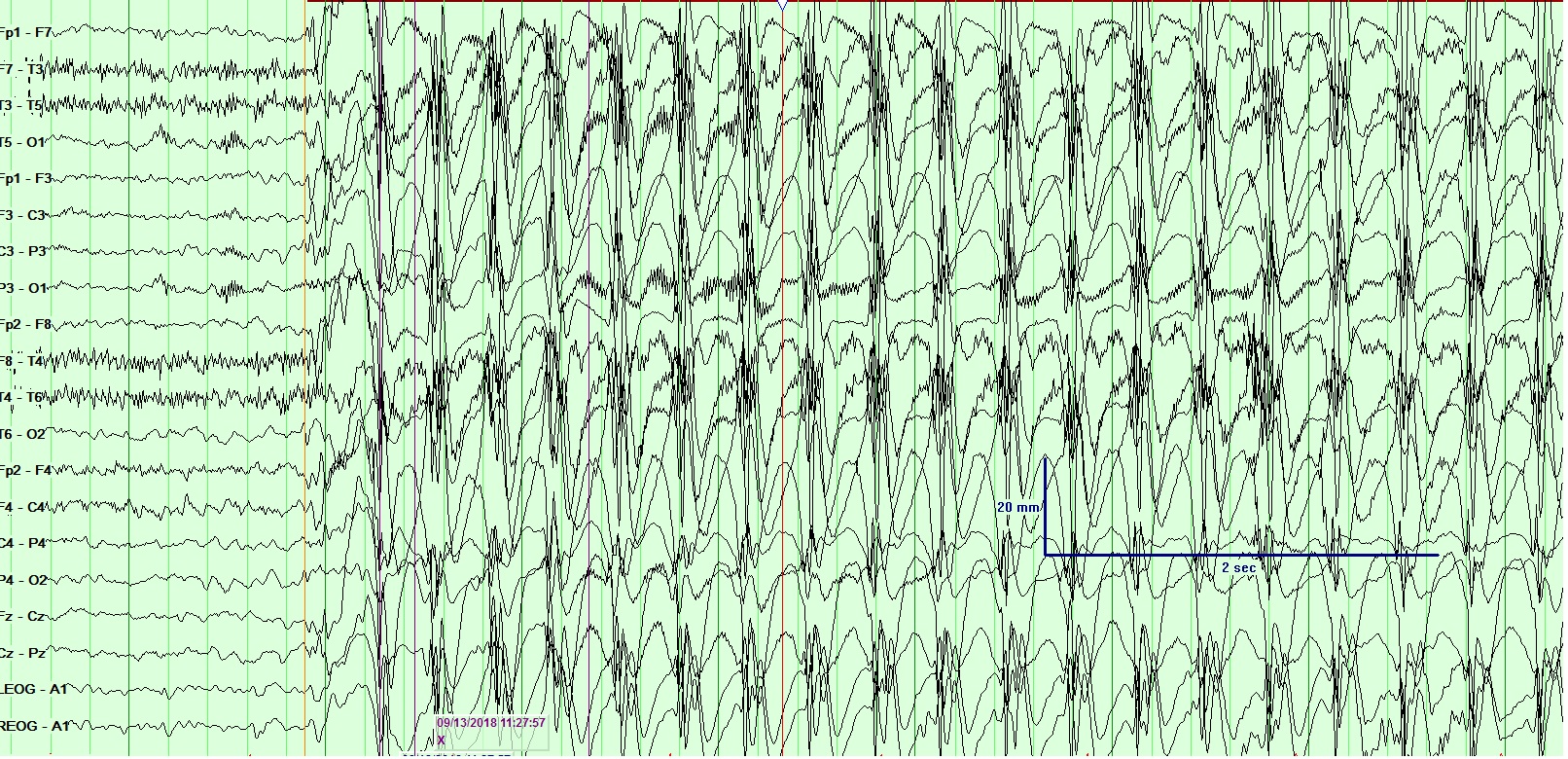
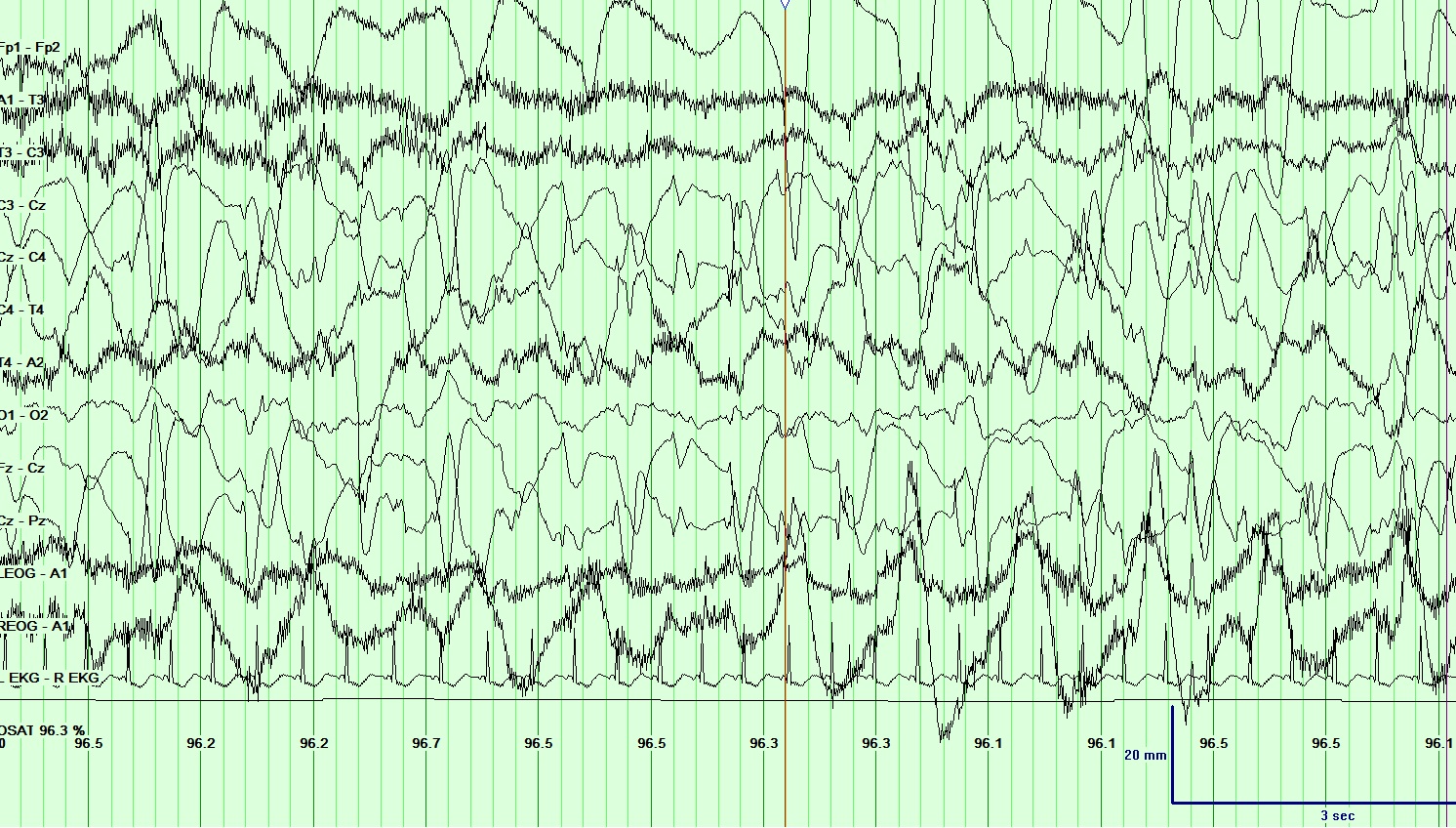
[1]
Scheffer IE, Berkovic S, Capovilla G, Connolly MB, French J, Guilhoto L, Hirsch E, Jain S, Mathern GW, Moshé SL, Nordli DR, Perucca E, Tomson T, Wiebe S, Zhang YH, Zuberi SM. ILAE classification of the epilepsies: Position paper of the ILAE Commission for Classification and Terminology. Epilepsia. 2017 Apr:58(4):512-521. doi: 10.1111/epi.13709. Epub 2017 Mar 8
[PubMed PMID: 28276062]
[2]
Donos C, Maliia MD, Dümpelmann M, Schulze-Bonhage A. Seizure onset predicts its type. Epilepsia. 2018 Mar:59(3):650-660. doi: 10.1111/epi.13997. Epub 2018 Jan 11
[PubMed PMID: 29322500]
[3]
Skidmore CT. Adult Focal Epilepsies. Continuum (Minneapolis, Minn.). 2016 Feb:22(1 Epilepsy):94-115. doi: 10.1212/CON.0000000000000290. Epub
[PubMed PMID: 26844732]
[4]
Guerreiro CA. Epilepsy: Is there hope? The Indian journal of medical research. 2016 Nov:144(5):657-660. doi: 10.4103/ijmr.IJMR_1051_16. Epub
[PubMed PMID: 28361817]
[5]
Baumgartner C, Koren JP. Seizure detection using scalp-EEG. Epilepsia. 2018 Jun:59 Suppl 1():14-22. doi: 10.1111/epi.14052. Epub
[PubMed PMID: 29873826]
[7]
Halgren M, Fabó D, Ulbert I, Madsen JR, Erőss L, Doyle WK, Devinsky O, Schomer D, Cash SS, Halgren E. Superficial Slow Rhythms Integrate Cortical Processing in Humans. Scientific reports. 2018 Feb 1:8(1):2055. doi: 10.1038/s41598-018-20662-0. Epub 2018 Feb 1
[PubMed PMID: 29391596]
[8]
Biasiucci A, Franceschiello B, Murray MM. Electroencephalography. Current biology : CB. 2019 Feb 4:29(3):R80-R85. doi: 10.1016/j.cub.2018.11.052. Epub
[PubMed PMID: 30721678]
[9]
Kirschstein T, Köhling R. What is the source of the EEG? Clinical EEG and neuroscience. 2009 Jul:40(3):146-9
[PubMed PMID: 19715175]
[10]
Mei T, Wei X, Chen Z, Tian X, Dong N, Li D, Zhou Y. Epileptic foci localization based on mapping the synchronization of dynamic brain network. BMC medical informatics and decision making. 2019 Jan 31:19(Suppl 1):19. doi: 10.1186/s12911-019-0737-8. Epub 2019 Jan 31
[PubMed PMID: 30700279]
[11]
Stam CJ. Modern network science of neurological disorders. Nature reviews. Neuroscience. 2014 Oct:15(10):683-95. doi: 10.1038/nrn3801. Epub 2014 Sep 4
[PubMed PMID: 25186238]
[12]
Ravindra VM, Sweney MT, Bollo RJ. Recent developments in the surgical management of paediatric epilepsy. Archives of disease in childhood. 2017 Aug:102(8):760-766. doi: 10.1136/archdischild-2016-311183. Epub 2017 Jan 17
[PubMed PMID: 28096104]
[13]
Ye BS, Cho YJ, Jang SH, Lee MK, Lee BI, Heo K. The localizing and lateralizing value of auras in lesional partial epilepsy patients. Yonsei medical journal. 2012 May:53(3):477-85. doi: 10.3349/ymj.2012.53.3.477. Epub
[PubMed PMID: 22476989]
[14]
Chatzikonstantinou A. Epilepsy and the hippocampus. Frontiers of neurology and neuroscience. 2014:34():121-42. doi: 10.1159/000356435. Epub 2014 Apr 16
[PubMed PMID: 24777136]
[15]
National Clinical Guideline Centre (UK). The Epilepsies: The Diagnosis and Management of the Epilepsies in Adults and Children in Primary and Secondary Care: Pharmacological Update of Clinical Guideline 20. 2012 Jan:():
[PubMed PMID: 25340221]
[16]
Renzel R, Baumann CR, Poryazova R. EEG after sleep deprivation is a sensitive tool in the first diagnosis of idiopathic generalized but not focal epilepsy. Clinical neurophysiology : official journal of the International Federation of Clinical Neurophysiology. 2016 Jan:127(1):209-213. doi: 10.1016/j.clinph.2015.06.012. Epub 2015 Jun 18
[PubMed PMID: 26118491]
[17]
Sohrabpour A, Lu Y, Kankirawatana P, Blount J, Kim H, He B. Effect of EEG electrode number on epileptic source localization in pediatric patients. Clinical neurophysiology : official journal of the International Federation of Clinical Neurophysiology. 2015 Mar:126(3):472-80. doi: 10.1016/j.clinph.2014.05.038. Epub 2014 Jul 11
[PubMed PMID: 25088733]
[18]
Gu Y, Cleeren E, Dan J, Claes K, Van Paesschen W, Van Huffel S, Hunyadi B. Comparison between Scalp EEG and Behind-the-Ear EEG for Development of a Wearable Seizure Detection System for Patients with Focal Epilepsy. Sensors (Basel, Switzerland). 2017 Dec 23:18(1):. doi: 10.3390/s18010029. Epub 2017 Dec 23
[PubMed PMID: 29295522]
[19]
Kutluay E, Kalamangalam GP. Montages for Noninvasive EEG Recording. Journal of clinical neurophysiology : official publication of the American Electroencephalographic Society. 2019 Sep:36(5):330-336. doi: 10.1097/WNP.0000000000000546. Epub
[PubMed PMID: 31490450]
[20]
Wu Z, Su S. A dynamic selection method for reference electrode in SSVEP-based BCI. PloS one. 2014:9(8):e104248. doi: 10.1371/journal.pone.0104248. Epub 2014 Aug 6
[PubMed PMID: 25100038]
[21]
Gordon R, Rzempoluck EJ. Introduction to Laplacian montages. American journal of electroneurodiagnostic technology. 2004 Jun:44(2):98-102
[PubMed PMID: 15328706]
[22]
Foldvary N, Caruso AC, Mascha E, Perry M, Klem G, McCarthy V, Qureshi F, Dinner D. Identifying montages that best detect electrographic seizure activity during polysomnography. Sleep. 2000 Mar 15:23(2):221-9
[PubMed PMID: 10737339]
[23]
Kang JY, Krauss GL. Normal Variants Are Commonly Overread as Interictal Epileptiform Abnormalities. Journal of clinical neurophysiology : official publication of the American Electroencephalographic Society. 2019 Jul:36(4):257-263. doi: 10.1097/WNP.0000000000000613. Epub
[PubMed PMID: 31274688]
[24]
Rosenow F, Klein KM, Hamer HM. Non-invasive EEG evaluation in epilepsy diagnosis. Expert review of neurotherapeutics. 2015 Apr:15(4):425-44. doi: 10.1586/14737175.2015.1025382. Epub 2015 Mar 16
[PubMed PMID: 25779862]
[25]
Aanestad E, Gilhus NE, Brogger J. Interictal epileptiform discharges vary across age groups. Clinical neurophysiology : official journal of the International Federation of Clinical Neurophysiology. 2020 Jan:131(1):25-33. doi: 10.1016/j.clinph.2019.09.017. Epub 2019 Nov 4
[PubMed PMID: 31751836]
[26]
Janati AB, Umair M, Alghasab NS, Al-Shurtan KS. Positive sharp waves in the EEG of children and adults. Neurological sciences : official journal of the Italian Neurological Society and of the Italian Society of Clinical Neurophysiology. 2014 May:35(5):707-13. doi: 10.1007/s10072-013-1588-1. Epub 2013 Nov 27
[PubMed PMID: 24281945]
[27]
Noachtar S, Rémi J. The role of EEG in epilepsy: a critical review. Epilepsy & behavior : E&B. 2009 May:15(1):22-33. doi: 10.1016/j.yebeh.2009.02.035. Epub 2009 Feb 25
[PubMed PMID: 19248841]
[28]
Siegel J, Tatum WO 4th. Index-finger pointing in generalized tonic-clonic seizures. Epilepsy & behavior : E&B. 2016 May:58():18-21. doi: 10.1016/j.yebeh.2016.02.022. Epub 2016 Mar 18
[PubMed PMID: 26994878]
[29]
Yao R, Xue J, Yang P, Wang Q, Gao P, Yang X, Deng H, Tan S, Li H. Dynamic Changes of Brain Networks during Working Memory Tasks in Schizophrenia. Neuroscience. 2021 Jan 15:453():187-205. doi: 10.1016/j.neuroscience.2020.11.007. Epub 2020 Nov 27
[PubMed PMID: 33249224]
[30]
Grant AC, Abdel-Baki SG, Omurtag A, Sinert R, Chari G, Malhotra S, Weedon J, Fenton AA, Zehtabchi S. Diagnostic accuracy of microEEG: a miniature, wireless EEG device. Epilepsy & behavior : E&B. 2014 May:34():81-5. doi: 10.1016/j.yebeh.2014.03.015. Epub 2014 Apr 12
[PubMed PMID: 24727466]
[31]
Gondeck AR, Smith JR. Dynamics of human sleep sigma spindles. Electroencephalography and clinical neurophysiology. 1974 Sep:37(3):293-7
[PubMed PMID: 4136646]
[32]
Worrell G, Gotman J. High-frequency oscillations and other electrophysiological biomarkers of epilepsy: clinical studies. Biomarkers in medicine. 2011 Oct:5(5):557-66. doi: 10.2217/bmm.11.74. Epub
[PubMed PMID: 22003904]
[33]
Kawano T, Hattori N, Uno Y, Kitajo K, Hatakenaka M, Yagura H, Fujimoto H, Yoshioka T, Nagasako M, Otomune H, Miyai I. Large-Scale Phase Synchrony Reflects Clinical Status After Stroke: An EEG Study. Neurorehabilitation and neural repair. 2017 Jun:31(6):561-570. doi: 10.1177/1545968317697031. Epub 2017 Mar 22
[PubMed PMID: 28506148]
[34]
Bartolomei F, McGonigal A, Naccache L. Alteration of consciousness in focal epilepsy: the global workspace alteration theory. Epilepsy & behavior : E&B. 2014 Jan:30():17-23. doi: 10.1016/j.yebeh.2013.09.012. Epub 2013 Oct 6
[PubMed PMID: 24103816]
[35]
Binnie CD, Prior PF. Electroencephalography. Journal of neurology, neurosurgery, and psychiatry. 1994 Nov:57(11):1308-19
[PubMed PMID: 7964803]
[36]
Dahal P, Ghani N, Flinker A, Dugan P, Friedman D, Doyle W, Devinsky O, Khodagholy D, Gelinas JN. Interictal epileptiform discharges shape large-scale intercortical communication. Brain : a journal of neurology. 2019 Nov 1:142(11):3502-3513. doi: 10.1093/brain/awz269. Epub
[PubMed PMID: 31501850]
[37]
Bettus G, Ranjeva JP, Wendling F, Bénar CG, Confort-Gouny S, Régis J, Chauvel P, Cozzone PJ, Lemieux L, Bartolomei F, Guye M. Interictal functional connectivity of human epileptic networks assessed by intracerebral EEG and BOLD signal fluctuations. PloS one. 2011:6(5):e20071. doi: 10.1371/journal.pone.0020071. Epub 2011 May 19
[PubMed PMID: 21625517]
[38]
Englot DJ, Konrad PE, Morgan VL. Regional and global connectivity disturbances in focal epilepsy, related neurocognitive sequelae, and potential mechanistic underpinnings. Epilepsia. 2016 Oct:57(10):1546-1557. doi: 10.1111/epi.13510. Epub 2016 Aug 24
[PubMed PMID: 27554793]
[39]
Lagarde S, Roehri N, Lambert I, Trebuchon A, McGonigal A, Carron R, Scavarda D, Milh M, Pizzo F, Colombet B, Giusiano B, Medina Villalon S, Guye M, Bénar CG, Bartolomei F. Interictal stereotactic-EEG functional connectivity in refractory focal epilepsies. Brain : a journal of neurology. 2018 Oct 1:141(10):2966-2980. doi: 10.1093/brain/awy214. Epub
[PubMed PMID: 30107499]
[40]
Tong X, An D, Xiao F, Lei D, Niu R, Li W, Ren J, Liu W, Tang Y, Zhang L, Zhou B, Gong Q, Zhou D. Real-time effects of interictal spikes on hippocampus and amygdala functional connectivity in unilateral temporal lobe epilepsy: An EEG-fMRI study. Epilepsia. 2019 Feb:60(2):246-254. doi: 10.1111/epi.14646. Epub 2019 Jan 17
[PubMed PMID: 30653664]
[41]
Foldvary N, Klem G, Hammel J, Bingaman W, Najm I, Lüders H. The localizing value of ictal EEG in focal epilepsy. Neurology. 2001 Dec 11:57(11):2022-8
[PubMed PMID: 11739820]
[42]
Vossler DG, Kraemer DL, Bell AJ. The Hippocampus and Cortex Together Generate the Scalp EEG Ictal Discharge in Temporal Lobe Epilepsy. Journal of clinical neurophysiology : official publication of the American Electroencephalographic Society. 2017 Sep:34(5):448-455. doi: 10.1097/WNP.0000000000000394. Epub
[PubMed PMID: 28574952]
[43]
Pourmotabbed H, Wheless JW, Babajani-Feremi A. Lateralization of epilepsy using intra-hemispheric brain networks based on resting-state MEG data. Human brain mapping. 2020 Aug 1:41(11):2964-2979. doi: 10.1002/hbm.24990. Epub 2020 May 13
[PubMed PMID: 32400923]
[44]
Bercovici E, Kumar BS, Mirsattari SM. Neocortical temporal lobe epilepsy. Epilepsy research and treatment. 2012:2012():103160. doi: 10.1155/2012/103160. Epub 2012 Jul 16
[PubMed PMID: 22953057]
[45]
Tatum WO 4th. Mesial temporal lobe epilepsy. Journal of clinical neurophysiology : official publication of the American Electroencephalographic Society. 2012 Oct:29(5):356-65. doi: 10.1097/WNP.0b013e31826b3ab7. Epub
[PubMed PMID: 23027091]
[46]
Murro AM, Park YD, King DW, Gallagher BB, Smith JR, Yaghmai F, Toro V, Figueroa RE, Loring DW, Littleton W. Seizure localization in temporal lobe epilepsy: a comparison of scalp-sphenoidal EEG and volumetric MRI. Neurology. 1993 Dec:43(12):2531-3
[PubMed PMID: 8255452]
[47]
V KR, Rajagopalan SS, Bhardwaj S, Panda R, Reddam VR, Ganne C, Kenchaiah R, Mundlamuri RC, Kandavel T, Majumdar KK, Parthasarathy S, Sinha S, Bharath RD. Machine learning detects EEG microstate alterations in patients living with temporal lobe epilepsy. Seizure. 2018 Oct:61():8-13. doi: 10.1016/j.seizure.2018.07.007. Epub 2018 Jul 10
[PubMed PMID: 30044996]
[48]
Maizuliana H, Usui N, Terada K, Kondo A, Inoue Y. Clinical, semiological, electroencephalographic, and neuropsychological features of "pure" neocortical temporal lobe epilepsy. Epileptic disorders : international epilepsy journal with videotape. 2020 Feb 1:22(1):55-65. doi: 10.1684/epd.2020.1132. Epub
[PubMed PMID: 32031536]
[49]
Blair RD. Temporal lobe epilepsy semiology. Epilepsy research and treatment. 2012:2012():751510. doi: 10.1155/2012/751510. Epub 2012 Mar 7
[PubMed PMID: 22957241]
[50]
Avoli M, de Curtis M, Gnatkovsky V, Gotman J, Köhling R, Lévesque M, Manseau F, Shiri Z, Williams S. Specific imbalance of excitatory/inhibitory signaling establishes seizure onset pattern in temporal lobe epilepsy. Journal of neurophysiology. 2016 Jun 1:115(6):3229-37. doi: 10.1152/jn.01128.2015. Epub 2016 Apr 13
[PubMed PMID: 27075542]
[51]
Vaessen MJ, Jansen JF, Braakman HM, Hofman PA, De Louw A, Aldenkamp AP, Backes WH. Functional and structural network impairment in childhood frontal lobe epilepsy. PloS one. 2014:9(3):e90068. doi: 10.1371/journal.pone.0090068. Epub 2014 Mar 4
[PubMed PMID: 24594874]
[52]
Gold JA, Sher Y, Maldonado JR. Frontal Lobe Epilepsy: A Primer for Psychiatrists and a Systematic Review of Psychiatric Manifestations. Psychosomatics. 2016 Sep-Oct:57(5):445-64. doi: 10.1016/j.psym.2016.05.005. Epub 2016 May 16
[PubMed PMID: 27494984]
Level 1 (high-level) evidence
[53]
Feyissa AM, Britton JW, Van Gompel J, Lagerlund TL, So E, Wong-Kisiel LC, Cascino GC, Brinkman BH, Nelson CL, Watson R, Worrell GA. High density scalp EEG in frontal lobe epilepsy. Epilepsy research. 2017 Jan:129():157-161. doi: 10.1016/j.eplepsyres.2016.12.016. Epub 2017 Jan 2
[PubMed PMID: 28073096]
[54]
Bonini F, McGonigal A, Trébuchon A, Gavaret M, Bartolomei F, Giusiano B, Chauvel P. Frontal lobe seizures: from clinical semiology to localization. Epilepsia. 2014 Feb:55(2):264-77. doi: 10.1111/epi.12490. Epub 2013 Dec 24
[PubMed PMID: 24372328]
[55]
Beleza P, Pinho J. Frontal lobe epilepsy. Journal of clinical neuroscience : official journal of the Neurosurgical Society of Australasia. 2011 May:18(5):593-600. doi: 10.1016/j.jocn.2010.08.018. Epub 2011 Feb 23
[PubMed PMID: 21349720]
[56]
Whitehead K, Gollwitzer S, Millward H, Wehner T, Scott C, Diehl B. The additional lateralizing and localizing value of the postictal EEG in frontal lobe epilepsy. Clinical neurophysiology : official journal of the International Federation of Clinical Neurophysiology. 2016 Mar:127(3):1774-80. doi: 10.1016/j.clinph.2015.11.050. Epub 2015 Dec 11
[PubMed PMID: 26750581]
[57]
Laskowitz DT, Sperling MR, French JA, O'Connor MJ. The syndrome of frontal lobe epilepsy: characteristics and surgical management. Neurology. 1995 Apr:45(4):780-7
[PubMed PMID: 7723970]
[58]
Unnwongse K, Wehner T, Foldvary-Schaefer N. Mesial frontal lobe epilepsy. Journal of clinical neurophysiology : official publication of the American Electroencephalographic Society. 2012 Oct:29(5):371-8. doi: 10.1097/WNP.0b013e31826b3c60. Epub
[PubMed PMID: 23027093]
[59]
Benbadis S. The differential diagnosis of epilepsy: a critical review. Epilepsy & behavior : E&B. 2009 May:15(1):15-21. doi: 10.1016/j.yebeh.2009.02.024. Epub 2009 Feb 21
[PubMed PMID: 19236946]
[60]
Lee RW, Worrell GA. Dorsolateral frontal lobe epilepsy. Journal of clinical neurophysiology : official publication of the American Electroencephalographic Society. 2012 Oct:29(5):379-84. doi: 10.1097/WNP.0b013e31826b3c7c. Epub
[PubMed PMID: 23027094]
[61]
Kellinghaus C, Lüders HO. Frontal lobe epilepsy. Epileptic disorders : international epilepsy journal with videotape. 2004 Dec:6(4):223-39
[PubMed PMID: 15634619]
[62]
Chibane IS, Boucher O, Dubeau F, Tran TPY, Mohamed I, McLachlan R, Sadler RM, Desbiens R, Carmant L, Nguyen DK. Orbitofrontal epilepsy: Case series and review of literature. Epilepsy & behavior : E&B. 2017 Nov:76():32-38. doi: 10.1016/j.yebeh.2017.08.038. Epub 2017 Sep 18
[PubMed PMID: 28928072]
Level 2 (mid-level) evidence
[63]
Salanova V. Parietal lobe epilepsy. Journal of clinical neurophysiology : official publication of the American Electroencephalographic Society. 2012 Oct:29(5):392-6. doi: 10.1097/WNP.0b013e31826c9ebc. Epub
[PubMed PMID: 23027096]
[64]
Ristić AJ, Alexopoulos AV, So N, Wong C, Najm IM. Parietal lobe epilepsy: the great imitator among focal epilepsies. Epileptic disorders : international epilepsy journal with videotape. 2012 Mar:14(1):22-31. doi: 10.1684/epd.2012.0484. Epub
[PubMed PMID: 22426412]
[65]
Akimura T, Fujii M, Ideguchi M, Yoshikawa K, Suzuki M. Ictal onset and spreading of seizures of parietal lobe origin. Neurologia medico-chirurgica. 2003 Nov:43(11):534-40
[PubMed PMID: 14705319]
[66]
Siegel AM, Williamson PD. Parietal lobe epilepsy. Advances in neurology. 2000:84():189-99
[PubMed PMID: 11091867]
Level 3 (low-level) evidence
[67]
Khan SA, Carney PW, Archer JS. Brief asymmetric tonic posturing with diffuse low-voltage fast activity in seizures arising from the mesial parietal region. Epilepsy research. 2014 Dec:108(10):1950-4. doi: 10.1016/j.eplepsyres.2014.09.011. Epub 2014 Oct 7
[PubMed PMID: 25445240]
[68]
Adcock JE, Panayiotopoulos CP. Occipital lobe seizures and epilepsies. Journal of clinical neurophysiology : official publication of the American Electroencephalographic Society. 2012 Oct:29(5):397-407. doi: 10.1097/WNP.0b013e31826c98fe. Epub
[PubMed PMID: 23027097]
[69]
AIRD RB, GASTAUT Y. Occipital and posterior electroencephalographic rhythms. Electroencephalography and clinical neurophysiology. 1959 Nov:11():637-56
[PubMed PMID: 13792196]
[70]
Harward SC, Chen WC, Rolston JD, Haglund MM, Englot DJ. Seizure Outcomes in Occipital Lobe and Posterior Quadrant Epilepsy Surgery: A Systematic Review and Meta-Analysis. Neurosurgery. 2018 Mar 1:82(3):350-358. doi: 10.1093/neuros/nyx158. Epub
[PubMed PMID: 28419330]
Level 1 (high-level) evidence
[71]
Marchi A, Bonini F, Lagarde S, McGonigal A, Gavaret M, Scavarda D, Carron R, Aubert S, Villeneuve N, Médina Villalon S, Bénar C, Trebuchon A, Bartolomei F. Occipital and occipital "plus" epilepsies: A study of involved epileptogenic networks through SEEG quantification. Epilepsy & behavior : E&B. 2016 Sep:62():104-14. doi: 10.1016/j.yebeh.2016.06.014. Epub 2016 Jul 22
[PubMed PMID: 27454330]
[72]
Epstein NE. Multidisciplinary in-hospital teams improve patient outcomes: A review. Surgical neurology international. 2014:5(Suppl 7):S295-303. doi: 10.4103/2152-7806.139612. Epub 2014 Aug 28
[PubMed PMID: 25289149]
[73]
Bosch B, Mansell H. Interprofessional collaboration in health care: Lessons to be learned from competitive sports. Canadian pharmacists journal : CPJ = Revue des pharmaciens du Canada : RPC. 2015 Jul:148(4):176-9. doi: 10.1177/1715163515588106. Epub
[PubMed PMID: 26448769]