Continuing Education Activity
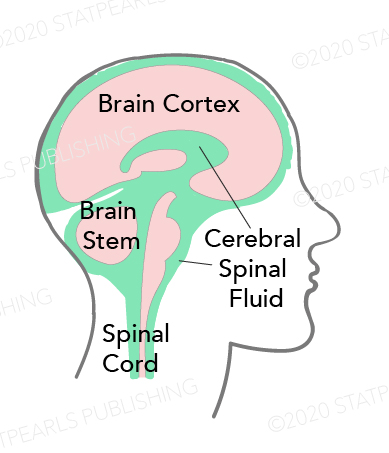
Cerebrospinal fluid (CSF) is a clear protein- and glucose-rich liquid in the subarachnoid space of the central nervous system. It is found in the ventricles, surrounding the brain, and within the central spinal column. CSF regulates central nervous system temperature, cushions the brain and spinal cord, and provides a delicately balanced buoyant force that allows the brain to retain its shape and circulatory integrity despite its weight and lack of intrinsic rigid support. A leak in this system, therefore, can be detrimental to brain blood supply and function and can increase the risk of direct trauma to brain parenchyma due to loss of fluid cushion. Additionally, the presence of a CSF leak indicates the need for further evaluation and management, as it may be due to a frontobasilar or temporal skull fracture. Open communication of the subarachnoid space with CSF leak presents a pathway for life-threatening CNS infection, including meningitis. This activity reviews the etiology, presentation, evaluation, and management of CSF leaks and reviews the role of the interprofessional team in evaluating, diagnosing, and managing the condition.
Objectives:
- Describe some of the most common etiologies for cerebrospinal fluid leaks.
- Review the appropriate steps in a workup and evaluation of a suspected CSF fluid leak.
- Outline management strategies for CSF fluid leaks based on various etiologies.
- Summarize the importance of collaboration and communication among the interprofessional team members to improve outcomes for patients affected by CSF leaking.
Introduction
Cerebrospinal fluid (CSF) is a clear, colorless liquid found in the subarachnoid space (SAS) that fills the neuroaxis cavities and surrounds the central nervous system (CNS). It consists predominantly of 99% water, and 1% accounts for electrolytes, proteins, neurotransmitters, and glucose.[1] According to Cushing and Weed's seminal theory, the CSF is mainly produced by the choroid plexuses, a network of blood vessels and tissues located within the ventricles of the brain, and circulates throughout the CNS to be finally reabsorbed into the venous sinuses at the level of arachnoid granulation (AGs).[2][3] However, much controversy remains regarding the basic CSF physiology, which led some researchers to defy the conventional theory and propose alternative homeostatic mechanisms of CSF regulation.[4][1][2]
The volume of CSF is estimated at 125 mL to 150 mL in the SAS and 25 mL in the ventricular system. This volume is defined by equal production and reabsorption rates of approximately 420 to 530 mL/day in adults.[5] The CSF regulates CNS temperature, cushions the brain and spinal cord, and provides a delicately balanced buoyant force that allows the brain to retain its shape and circulatory integrity despite its weight and lack of intrinsic rigid support. The CSF also forms the blood-CSF barrier (BCSFB) circulation, acting in the removal of waste products and metabolites through its continuous renewal.[6]
The CSF leak occurs when there is a tear or hole in the dura mater, the outermost layer of the meninges that protect the central nervous system, communicating the subarachnoid space with the epidural space and, sometimes, with the skin establishing a connexion between the CNS and the external environment.[7] This can cause low-pressure headaches, neck pain, ringing in the ear, and occasionally, loss of smell or taste. A leak can be detrimental to brain blood supply and function and can increase the risk of direct trauma to brain parenchyma due to loss of fluid cushion. Open communication of the subarachnoid space with CSF leak also presents a pathway for life-threatening CNS infection, including meningitis. Therefore, the presence of signs and symptoms of a CSF leak indicates the need for further evaluation and management.
Etiology
Cerebrospinal fluid (CSF) leak is the escape of the fluid surrounding the brain and spinal cord. It occurs when there is a tear or hole in the dura mater, the meninges' outermost layer that protects the central nervous system, communicating the subarachnoid space and other spaces via meningeal disruption. The most common symptoms are low-pressure (orthostatic) headaches (92%), nausea (54%), and neck pain (43%). [8] Ringing in the ear and loss of smell or taste is also possible clinical findings. The most common cause of leaking cerebrospinal fluid is a structural compromise secondary to craniofacial trauma, making up 80% of CSF leaks. Iatrogenic causes comprise 16% of CSF leaks, with the last 4% due to varied etiologies. CSF leak is typically classified into spontaneous/idiopathic, traumatic, and iatrogenic.[9][10]
Craniofacial trauma can lead to varied presentations of CSF leak, determined primarily by injury location and mechanism of action. Skull base CSF leaks occur when there is an abnormal communication between the subarachnoid space and air space in the paranasal sinuses, nasal cavity, middle ear, or mastoid cells.[11]Anterior skull base fractures are frequently associated with moderate-to-high-velocity impact. The cribriform plate, ethmoid bone, and sphenoid sinuses are thin and closely associated with the dura. Less regularly, fractures of the temporal bone (middle ears or mastoid cells) are associated with dural disruption, which can also result in CSF leak through the ears (otorrhea). Rarely, injury and disruption to the orbit can result in CSF occulorrhea.[12]
Iatrogenic CSF leaks occur most frequently as sequelae to functional endoscopic sinus surgery. The cribriform plate and ethmoid bone are the most commonly injured, followed by the frontal and sphenoid sinuses.[9][13] Neurosurgical interventions contribute to iatrogenic leaks, especially with the increased prevalence of endoscopic transnasal pituitary surgeries. The proposed etiologies of these leaks range from anatomical variations to technical factors and may be recognized intra- or postoperatively. In one study, pituitary tumor resection made up nearly half of the cases where tumor removal led to a confirmed CSF leak.[13] Spinal CSF leaks may also occur after procedures such as lumbar punctures, lumbar-abdominal shunt track, epidural anesthesia, and spinal surgery.[14]
Spontaneous CSF leaks occur without an obvious inciting event, often at the spine level, only rarely from the skull base. Spontaneous leaks are typically attributed to underlying conditions resulting in decreased intracranial pressure (ICP), leading to orthostatic headaches, the most common clinical manifestation.[15][16] Four types of spontaneous spinal CSF leaks and their incidence among 568 patients were recently proposed by Schievink et al.[17]
Type 1 - dural tear comprises 26.6% of the CSF leaks. They are subdivided into type 1a.ventral (96%), end type 1b.dorsolateral (4%). These ventral tears are typically vertically oriented along the fibers of the dura and are associated with calcifications at the level of the disc space. Ventral spine CSF leaks tend to become chronic, and if present for many years, they have been associated with various neurologic complications, such as non-aneurismal intracranial subarachnoid hemorrhage.[18] Type 2 - meningeal diverticulum (42.3% of CSF leaks). They were subdivided into type 2a. Simple single or multiple diverticula (90.8%); and into type 2b. complex meningel diverticula or dural ectasia (9.2%). The meningeal diverticula may represent an underlying dural friability predisposing patients to CSF leaks. Type 3 - CSF-venous fistula, found in 2.5% of the patients. These fistulas are more common among women with spontaneous intracranial hypotension. Type 4 - Indeterminate/Idiopathic: comprises 28.7% of the CSF leaks, and half of them presented radiological evidence of epidural collection.
Spontaneous CSF leaks are also associated with elevated ICP or idiopathic intracranial hypertension (IIH). These leaks were most likely secondary to an erosion of thin bony structures of the skull base discussed above due to chronically increased ICP.[19][20] Preexisting dural weakness likely related to a heritable disorder of the connective tissues matrix is also described as a significant contributory factor.[21] The non-traumatic CSF leak is also caused due to congenital skull base formation and skull base defect due to tumors.
Epidemiology
According to Ommaya et al., 80 % of CSF leak is due to nonsurgical trauma, 16 % is iatrogenic, and 4 % is spontaneous.[22] It is known that spontaneous spinal CSF leaks are more common in female adults than in children.[23] The average patient age at the presentation for various types of spontaneous CSF leaks ranges from 33 years (+/-10) to 52.4 years (+/-13.1).[24] Its most common location is at the upper thoracic level (T1 - T6), followed by the lower thoracic level (T7 - T12), and being the lumbar and cervical spine levels the less common regions.[25]
Nearly 2.8 million people in the United States sustained a head injury that resulted in an emergency department visit, hospitalization, or death between 2007 and 2013. Skull base fractures occur in approximately 4% of these injuries, accounting for 21% of all skull fractures. Male patients are predominant (78%), and the mean age was 49.[26] The widely cited incidence of CSF leaks resulting from skull base fractures has been reported in the literature as 10 to 30%. Still, recently the retrospective study from the Harward group reviewed the incidence of CSF leaks among 4,944 patients with cranial facial fractures and reported a lower incidence of 4%.[27][12][28]
A cerebrospinal fluid leak is also a known complication of post-trauma surgical procedures, endoscopic endonasal skull base surgery, operations involving the lumbosacral spine, as well as diagnostic or therapeutic lumbar puncture. The CSF leak incidence after primary spine surgery varies from 5.5% to 9% and 13.2% to 21% for second-look surgeries.[29] Non-identified intraoperative durotomies occurred in 6.8% of cases, and the incidence of CSF leaks was less frequent in minimally invasive surgeries (4.7%) compared to 9.0% of open cases, according to Wong et al.[30] A recent literature review on CSF leaks in the endoscopic endonasal approach for tumor resection showed an overall post-operative rate of 10.1%. It also suggests that the material and closure technique can influence that rate.[31]
Pathophysiology
Although CSF leaks can arise from the skull base, these types of leaks do not typically cause orthostatic headaches and are more associated with high (rather than low) CSF pressure when spontaneous or head trauma and post-surgical procedures.[32][33]
The mechanism of CSF leaks after head trauma and surgical procedures is mainly due to the breach in the intervening layers of mucosa, bone, dura, and arachnoid membrane, which results in rhinorrhea - the flow of CSF into the nose.[24]
Spinal CSF leaks are currently recognized to occur through three primary mechanisms: meningeal diverticula, ventral dural tears, and CSF-venous fistulas (CVFs).[32]
Meningeal diverticula: areas of dural dehiscence that permit protrusion of the leptomeninges through the dural defect, creating a fragile outpunching-prone rupture. Some diverticula involve large meningeal tears that allow rapid egress of CSF, whereas others produce slow weeping of CSF with the Valsalva maneuver.
Ventral dural tears are more commonly caused by calcified disk protrusions or sharp endplate osteophytes that incise the dura, producing a longitudinally oriented tear. Leaks from ventral tears are often rapid, resulting in extensive epidural CSF collections.
CSF - Venous Fistula: In this entity, there is a direct connection between the spinal subarachnoid space and the draining paraspinal vein that allows rapid loss of CSF into the venous circulation. The CSF is usually reabsorbed at the level of spinal nerve roots by the arachnoid villi regulated by vacuoles. In contrast, the CSF loss due to CVFs is unregulated, resulting in CSF volume depletion and intracranial hypotension. The thoracic spine is the most common location for CVFs.
History and Physical
As most CSF leaks are secondary to either accidental or iatrogenic trauma, the history of a patient presenting with rhinorrhea or otorrhea should raise suspicion for a CSF leak; thus, one should obtain a recent history of trauma or a surgical procedure. The most common presenting symptom across all skull base leaks is clear rhinorrhea that may be accompanied by a headache, neck pain, or stiffness.[34] Several uncommon symptoms can also result from compression of structures at the skull base, brain stem, or spinal cord due to brain herniations through the bone and dural defect. Examples include galactorrhea, quadriparesis, infarcts, and coma.[7]
Orthostatic headache is the hallmark symptom of SIH, presumably caused by positionally exacerbated meningeal traction. Occasionally, this evolves into a non-positional chronic headache. Rarely, the headache may be paradoxically improved when upright; prior authors have speculated that this may be due to worsening engorgement of the dural venous sinuses when recumbent.[35] A large systematic review reports numerous additional symptoms, most commonly nausea, neck pain or stiffness, tinnitus, and dizziness.[8]
Other atypical presentations of chronic CSF leak include obtundation, memory deficits, dementia with frontotemporal features (frontotemporal brain sagging syndrome), parkinsonism, and ataxia.[36] Recent evidence suggests that dural defects frequently underlie unexplained posterior fossa-predominant superficial siderosis due to bleeding from friable epidural veins at the site of the dural tear.[37]
Radiculopathy, brachial amyotrophy, and myelopathy can occur in SIH, both secondary to spinal cord superficial siderosis or/and CNS compression.[32]
Evaluation
A careful diagnostic evaluation of patients clinically suspected to have CSF leak is crucial, with the awareness of CSF components and imaging abnormalities.
Evaluating a suspected leak should include testing rhinorrhea or otorrhea for beta-2 transferrin, a compound found only in CSF and perilymph, making it a highly specific and sensitive test.[38][39] Beta-2 transferrin is a carbohydrate-free isoform of transferring, which is almost exclusively found in the CSF. It is not present in the blood, nasal mucus, tears, or mucosal discharge. Intense research over the last decade has validated characteristics and value in clinical use as a specific CSF marker. [40] If negative in the CSF from a suspected leak, there is a low likelihood. Glucose testing of rhinorrhea liquid was more frequently done in the past but had poor sensitivity and specificity compared to beta-2 transferrin. If beta-transferrin is positive during an acute leak or if there is a high index of suspicion, imaging is indicated to localize the source.
Traumatic rhinorrhea has been classified according to the latency of occurrence, and different evaluation and treatment methods can be applied accordingly. In the acute/early onset type, the patient presents with a CSF leak within the first two days of trauma, the delayed type presents at least one week after trauma, and the late onset/occult type presents within three months after trauma. According to several authors, 50% start in the first 2 or 3 days, 70% start within the first week, and almost all traumatic CSF leaks occur within three months of trauma.[24] A high level of suspicion is required to identify patients of occult/late onset CSF fistula; there are situations when patients may present with recurrent post-traumatic meningitis without an active leak. Evaluation of such patients with a history of trauma must include high-resolution CT (HRCT) to identify the defect.[24]
High-resolution CT (HRCT) of paranasal sinuses and temporal bone is typically sufficient for identifying single osseous defects. If multiple defects are suspected on HRCT, CT cisternography is useful to localize the lesions further. If initial CT is suspicious for meningoencephalocele, magnetic resonance (MR) cisternography is highly sensitive to soft tissue findings. If the leak is intermittent but active, HRCT is still the first-line imaging test of choice. If the suspected leak is inactive at the time of imaging, consider contrast-enhanced MR cisternography or radionucleotide cisternography.[41]
The cause of spontaneous CSF rhinorrhea may be attributed to idiopathic intracranial hypertension (IIH); therefore, imaging evaluation and fundoscopy are highly recommended. In cases of spontaneous CSF leak with intracranial hypotension, magnetic resonance imaging (MRI) of the brain may show intracranial pachymeningeal thickening and enhancement with contrast, subdural fluid collections, and downward displacement of the brain. MRI of the spine can also show dural collapse and may demonstrate CSF leakage from spinal dural defects.[42] First-line spinal imaging techniques are CT myelography (CTM) and MR imaging, which may be followed by dynamic techniques such as dynamic myelography, digital subtraction myelography, and dynamic CTM if a high flow leak is found and the suspected site of leak is yet to be identified. [43]
In a recent study, MRI findings of intracranial hypotension were present in 79% of patients with spontaneous CSF leaks. These findings have an overall sensitivity of 85.7%. With detailed analyses, enhancement of the pachymeninges showed the highest sensitivity of 78.6%, followed by subdural fluid accumulation and sagging brain, 21.4%, and subsequently by engorgement of venous structures and pituitary hyperemia, both 14.3%.[44]
Treatment / Management
Treatment of a CSF leak depends on the underlying cause, size, and location of the tear. If it is a small leak, it may resolve on its own, while larger leaks may require surgical intervention. Conservative treatment for a CSF leak typically includes bed rest and increased oral fluids. Other treatments, such as a lumbar epidural blood patch, may be recommended if the leak persists. A lumbar epidural blood patch involves injecting a small amount of the patient's own blood into the area around the spinal cord. This helps to seal the leak and reduce the symptoms associated with the leak. Other treatments for a CSF leak may include nasal packing, endoscopic repair, and surgical repair.
In cases of craniofacial trauma, it has been posited that conservative management and observation should be employed since a number of these resolve with no intervention. However, the risk of developing meningitis in these patients is up to 29%, so this course should be pursued cautiously.[45] Using lumbar shunts or repeat lumbar punctures to lower ICP, endoscopic repair, and in cases of refractory or particularly high ICP, ventriculoperitoneal (VP) shunts can be effective but have relatively high complication rates.[13]
Conservative treatment for a CSF leak may include bed rest: This helps reduce pressure on the affected area and allows the leak to heal. Pain management: Over-the-counter pain relievers such as ibuprofen or acetaminophen may be recommended to relieve headaches and neck pain. Hydration: Drinking plenty of fluids helps to maintain adequate fluid levels in the body and reduce symptoms. Caffeine restriction: Caffeine can worsen symptoms and should be avoided or limited. Position changes: Avoiding activities that increase pressure on the affected area, such as coughing, sneezing, or straining, can help reduce symptoms. Acetazolamide in the dosage of 500 mg twice daily for the first week, followed by a lower dosage to 250 mg twice daily for a second week, seems to have a 100% success rate in the closure of the primary defect in spontaneous CSF leak with controlled ICP.[24] According to numerous researchers, acetazolamide should always be administered to patients who exhibit spontaneous CSF leaks and signs of elevated CSF pressure. Acetazolamide reduces CSF synthesis by 48%.[34]
For mild occurrences of a CSF leak, conservative treatment is a viable option and may be successful in letting the leak heal on its own. However, more invasive therapies, such as surgical correction, may be required in more severe situations or if symptoms continue. Working closely with a skilled healthcare professional is crucial to choosing the right course of action for each unique instance.
The epidural blood patch (EBP) procedure is a commonly used treatment for cerebrospinal fluid (CSF) leaks. The procedure involves injecting a small volume of the patient's own blood into the epidural space surrounding the spinal cord to seal the leak and prevent further loss of CSF fluid.[46] The EBP procedure is typically performed under local anesthesia, with the patient lying on their stomach. The anesthesiologist numbs the skin and tissues over the lower back, then uses a needle to insert a catheter into the epidural space. The patient's own blood (ranging from 10 to 55 ml) is then slowly infused through the catheter, filling the epidural space and putting pressure on the leaking area to close it. The EBP procedure is usually considered a safe and effective treatment for CSF leaks, with a high success rate in resolving symptoms. It is often used for patients who have not responded to conservative treatment or have experienced a recurrent leak. There was no statistical difference in the success rate between target and non-target patching techniques.[43]
The epidural blood patch procedure is valuable for neurosurgeons and other specialists treating CSF leaks. The procedure is simple, low-risk, and can quickly relieve symptoms in most cases. The success rates for EBP have been shown to increase with the volume of autologous blood, with a rate of 80% achieved with 10 to 15 ml and more than 95% with 20 ml.[47]
The lumbar drain procedure involves the insertion of a catheter into the lumbar (lower back) region of the spinal canal, where the CSF fluid accumulates. The lumbar drain treatment is typically done to temporarily relieve symptoms and give the CSF leak time to mend. The size and location of the leak, as well as the patient's unique healing process, will determine how long the catheter must remain in place. During the surgery, a small incision is created in the lower back, and a thin tube is put through it. The CSF can then be removed from the body by connecting the tube to a drainage system. The patient often stays in the hospital for a few days following the procedure to be observed and get any necessary follow-up care. The surgery is often carried out under local or general anesthesia due to its low risk.[48]
Surgery is recommended when the leak site has been identified, symptoms have been refractory to less invasive treatments, and severe symptoms persist.
The endoscopic nasal packing procedure involves the placement of small, absorbent material into the nasal passages to block the leak and prevent further loss of CSF fluid. The endoscopic nasal packing procedure is typically performed under general anesthesia and uses an endoscope, a thin, flexible tube with a light and camera attached, to visualize the inside of the nose. The surgeon identifies the site of the leak and uses specialized instruments to place the absorbent material, such as Gelfoam and/or fibrin glue, to patch the defect into the nasal passages. The packing material seals the leak and helps promote healing. The endoscopic nasal packing procedure is considered a safe and effective option for treating CSF leaks that originate in the nasal passages. The procedure is minimally invasive and does not require a large incision or prolonged recovery time, making it a popular choice for patients who want to avoid more invasive surgical options.[49] In post-surgical cases with skull base fractures, an endonasal endoscopic approach has a first-attempt success rate ranging from 80 to 91%. The remaining patients may need further endoscopic revision, with less than 10% of cases requiring open surgical revision.[20] The surgical failure rate is higher among those with elevated ICP.[50]
External Ventricular Drainage (EVD) procedure is a neurosurgical treatment option for a CSF leak. The EVD procedure involves placing a drain in the brain's ventricles to remove cerebrospinal fluid (CSF) to reduce pressure and allow for better healing of the CSF leak region. During the procedure, a small burrhole is made two to three centimeters anterior to the coronal suture and three centimeters com the sagittal suture. Another landmark for the borehole is the mid-pupillary line. The catheter is then inserted through the hole and into the frontal horn of the lateral ventricle. The right side is preferred due to the risk of the lesion to the dominant motor cortex. The tube is then connected to an external drainage system, which is used to remove the CSF. The amount of CSF removed is monitored until the CFS leak heals completely. The procedure is usually performed in the operating room under general anesthesia. It is followed by a period of observation to ensure that the pressure is successfully reduced.[51]
In cases of iatrogenic injury during intracranial surgery, the obvious treatment is repairing the affected site, which may require multiple procedures or adjuvant therapy with the options described above.[52] In patients with higher ICP, it is thought that the implantation of an underlay bone graft will offer additional structural support to repair the skull base in more extensive lesions. Because the skull base in spontaneous CSF leaks is extensively attenuated and may fracture readily, great care should be used in shaping and placing this underlay bone graft. The specific overlay graft material chosen depends on the defect's size, location, shape, and the surgeon's personal preference. A variety of overlay graft materials have been demonstrated to be effective.[53]
Differential Diagnosis
The presentation of clear rhinorrhea and/or a headache is common in many conditions.[54][55] Those that should be specifically considered are:
- Allergic rhinitis
- Benign intracranial hypotension
- Carotid or vertebral artery dissection
- Infectious etiologies such as the common cold
- Meningitis
- Posttraumatic headache
- Spontaneous intracranial hypotension
- Subarachnoid hemorrhage
- Vasomotor rhinitis
- Sinus disease
- Spinal disease
- Migraine
- Brain tumor
Prognosis
As discussed in Treatment/Management, the overall prognosis of CSF leak is very favorable, with one study citing a 90% first-attempt success rate of endoscopic repair of CSF rhinorrhea and another study citing an overall success rate of 98% across traumatic, iatrogenic, and congenital causes.[56][13]
A recent study showed that when stratifying by first CSF leak treatment, EBP was more effective when applied to the right individuals with smaller dural abnormalities. On the other hand, the greater dural abnormalities can be the reason why surgical and lumbar drain treatments fail more frequently. Ninety-seven percent of the patients failed initial EBP, 21.1 failed lumbar drain, 13.5% failed surgery, and the overall failure rate was 10.9%.[57]
The success rate of endoscopic CSF repairs ranges from 87 to 100% after the first attempt. Patients with severe skull base abnormalities, lateral sphenoid leaks, raised body mass index, spontaneous CSF rhinorrhea, and elevated BMI are more likely to experience recurrence. Recurrence might also happen in patients in their middle age, who are obese and have an empty sella. A high recurrence rate is linked to elevated ICP, diabetes mellitus, and a body mass index of >30. Patients who have repeated CSF leaks may have a higher failure rate.[58]
Complications
A leak can be detrimental to brain blood supply and function and can increase the risk of direct trauma to brain parenchyma due to loss of fluid cushion. Chronic and untreated CSF leaks can cause low-pressure headaches, neck pain, ringing in the ear, and, occasionally, loss of smell or taste. Open communication of the subarachnoid space with CSF leak also presents a pathway for life-threatening CNS infection, including meningitis.
The most serious potential complication of CSF leak is meningitis. This risk appears to be highest in the preoperative period for those with confirmed CSF rhinorrhea, with those sustaining traumatic injuries having the highest risk of around 30%. Meningitis was found in 18.2% of trauma patients and a third of patients undergoing sphenoid, skull base, and calvaria operations. However, the risk of meningitis remains at 19% in those with persistent CSF leaks, and such persons remain at risk until successful operative closure.[59]
A recent study shows abducens nerve palsy as a potential complication of skull-base surgery for CSF treatment. Calvarian surgery has a more challenging clinical presentation, including headache, meningitis with fever, and conscious disturbance.[44]
Patients who had undergone lumbar puncture or spinal surgery did not express having meningitis. While skull-base surgery, calvaria surgery, and a lumbar puncture did not have such MRI results, trauma, and spinal surgery had more CSF accumulation imaging findings than these other procedures. Regarding the course of care, all groups experienced conservative treatment with similar success rates and lengths of time.
While surgery or trauma-associated CSF leak tended to favor surgical treatment, CSF leak related to lumbar puncture typically used blood patches as the most sophisticated therapy repair. Brain abscess (0.9%), subdural hematoma (0.3%), and smell disorders (0.6%) are so described as complications.[58]
Deterrence and Patient Education
One way to prevent CSF leaks is to avoid situations or activities that could injure the brain or spinal cord, such as high-impact sports, roller coasters, and other activities that could result in unexpected jolts or falls. Additionally, people with connective tissue diseases or a history of spinal or brain surgery may be more susceptible to CSF leaks and should take special care.
Another crucial component of controlling and preventing CSF leaks is patient education. The warning signs and symptoms of a CSF leak, which include headache, nausea, and a clear or salty discharge from the nose or ear, should be explained to patients. They should be told to get help from a doctor immediately if they encounter these symptoms.
Patients should also be made aware of the value of adhering to treatment recommendations, including bed rest, staying hydrated, and avoiding activities that raise intracranial pressure. Additionally, patients should be counseled against blowing their nose vigorously because doing so increases the possibility of a CSF leak.
Enhancing Healthcare Team Outcomes
The CSF leak workup affects numerous facets of healthcare, functioning as an interprofessional team. Any level of the care center and any professional, such as doctors, nurse practitioners, and physician assistants, may receive a patient's at their initial presentation. Laboratory technicians interpret testing CSF samples. Radiologic technicians obtain the proper investigations, and radiologists then interpret such studies. Surgical technologists, interventional radiologists, and surgeons then offer conclusive operative management. Pharmacists evaluate, and nurses administer prescriptions while monitoring the patient's vital signs during the patient's presentation, evaluation, and treatment. Effective communication between oimter[rpfessopma; team members is crucial for eliminating medical errors and minimizing patient damage because many of these patients' hospital stays may be protracted.