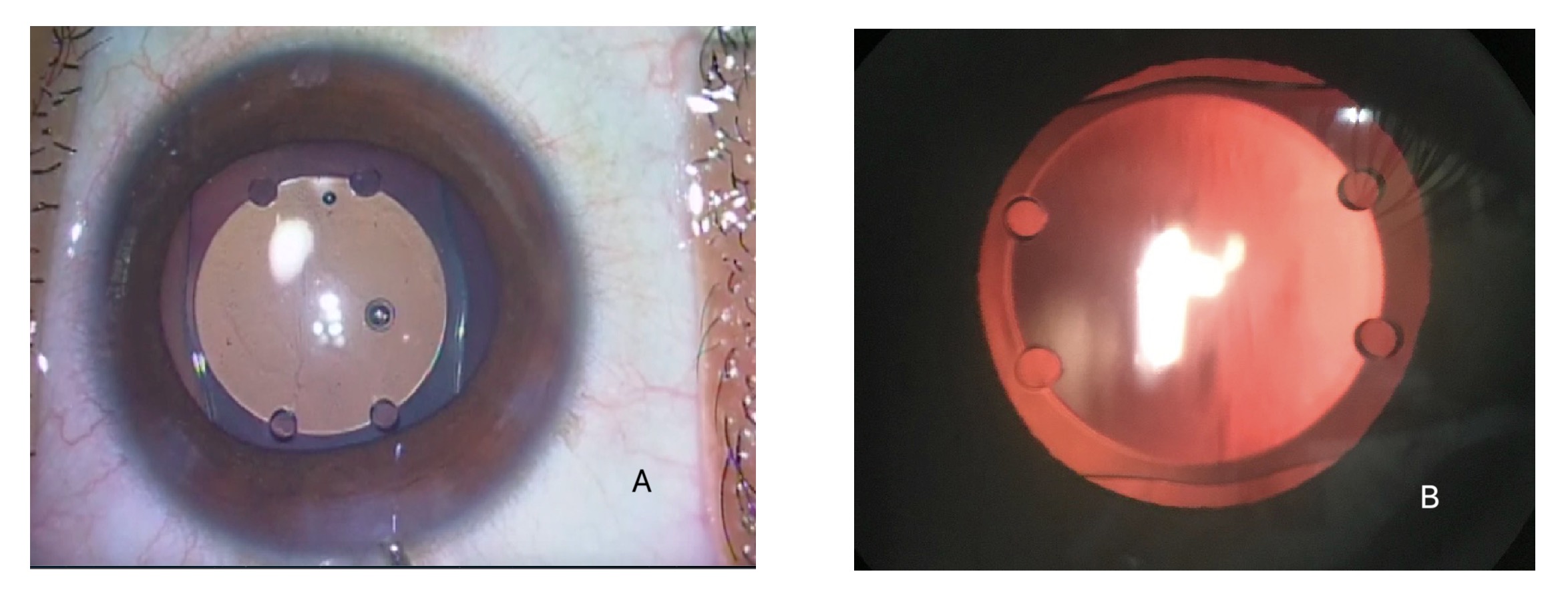
[1]
Baird PN, Saw SM, Lanca C, Guggenheim JA, Smith Iii EL, Zhou X, Matsui KO, Wu PC, Sankaridurg P, Chia A, Rosman M, Lamoureux EL, Man R, He M. Myopia. Nature reviews. Disease primers. 2020 Dec 17:6(1):99. doi: 10.1038/s41572-020-00231-4. Epub 2020 Dec 17
[PubMed PMID: 33328468]
[2]
Kaiti R , Pradhan A , Dahal HN , Shrestha P . Pattern and Prevalence of Refractive Error and Secondary Visual Impairment in Patients Attending a Tertiary Hospital in Dhulikhel, Nepal. Kathmandu University medical journal (KUMJ). 2018 Apr-Jun:16(62):114-119
[PubMed PMID: 30636750]
[3]
Wong CW, Tsai A, Jonas JB, Ohno-Matsui K, Chen J, Ang M, Ting DSW. Digital Screen Time During the COVID-19 Pandemic: Risk for a Further Myopia Boom? American journal of ophthalmology. 2021 Mar:223():333-337. doi: 10.1016/j.ajo.2020.07.034. Epub 2020 Jul 30
[PubMed PMID: 32738229]
[4]
Saunders KJ, McCullough SJ. Normative data for emmetropic and myopic eye growth in childhood. Ophthalmic & physiological optics : the journal of the British College of Ophthalmic Opticians (Optometrists). 2021 Nov:41(6):1382-1383. doi: 10.1111/opo.12873. Epub 2021 Aug 17
[PubMed PMID: 34402539]
[5]
Kleinstein RN, Jones LA, Hullett S, Kwon S, Lee RJ, Friedman NE, Manny RE, Mutti DO, Yu JA, Zadnik K, Collaborative Longitudinal Evaluation of Ethnicity and Refractive Error Study Group. Refractive error and ethnicity in children. Archives of ophthalmology (Chicago, Ill. : 1960). 2003 Aug:121(8):1141-7
[PubMed PMID: 12912692]
[6]
Ding BY, Shih YF, Lin LLK, Hsiao CK, Wang IJ. Myopia among schoolchildren in East Asia and Singapore. Survey of ophthalmology. 2017 Sep-Oct:62(5):677-697. doi: 10.1016/j.survophthal.2017.03.006. Epub 2017 Mar 28
[PubMed PMID: 28359704]
Level 3 (low-level) evidence
[7]
Vitale S, Sperduto RD, Ferris FL 3rd. Increased prevalence of myopia in the United States between 1971-1972 and 1999-2004. Archives of ophthalmology (Chicago, Ill. : 1960). 2009 Dec:127(12):1632-9. doi: 10.1001/archophthalmol.2009.303. Epub
[PubMed PMID: 20008719]
[8]
Li M, Tan CS, Foo LL, Sugianto R, Toh JY, Sun CH, Yap F, Sabanayagam C, Chong FM, Saw SM. Dietary intake and associations with myopia in Singapore children. Ophthalmic & physiological optics : the journal of the British College of Ophthalmic Opticians (Optometrists). 2022 Mar:42(2):319-326. doi: 10.1111/opo.12929. Epub 2021 Dec 3
[PubMed PMID: 34862645]
[9]
He X, Sankaridurg P, Xiong S, Li W, Naduvilath T, Lin S, Weng R, Lv M, Ma Y, Lu L, Wang J, Zhao R, Resnikoff S, Zhu J, Zou H, Xu X. Prevalence of myopia and high myopia, and the association with education: Shanghai Child and Adolescent Large-scale Eye Study (SCALE): a cross-sectional study. BMJ open. 2021 Dec 23:11(12):e048450. doi: 10.1136/bmjopen-2020-048450. Epub 2021 Dec 23
[PubMed PMID: 34949607]
Level 2 (mid-level) evidence
[10]
O'Donoghue L, Kapetanankis VV, McClelland JF, Logan NS, Owen CG, Saunders KJ, Rudnicka AR. Risk Factors for Childhood Myopia: Findings From the NICER Study. Investigative ophthalmology & visual science. 2015 Feb 5:56(3):1524-30. doi: 10.1167/iovs.14-15549. Epub 2015 Feb 5
[PubMed PMID: 25655799]
[11]
Villarreal MG, Ohlsson J, Abrahamsson M, Sjöstrom A, Sjöstrand J. Myopisation: the refractive tendency in teenagers. Prevalence of myopia among young teenagers in Sweden. Acta ophthalmologica Scandinavica. 2000 Apr:78(2):177-81
[PubMed PMID: 10794252]
[12]
Attebo K, Ivers RQ, Mitchell P. Refractive errors in an older population: the Blue Mountains Eye Study. Ophthalmology. 1999 Jun:106(6):1066-72
[PubMed PMID: 10366072]
[13]
Flitcroft DI, He M, Jonas JB, Jong M, Naidoo K, Ohno-Matsui K, Rahi J, Resnikoff S, Vitale S, Yannuzzi L. IMI - Defining and Classifying Myopia: A Proposed Set of Standards for Clinical and Epidemiologic Studies. Investigative ophthalmology & visual science. 2019 Feb 28:60(3):M20-M30. doi: 10.1167/iovs.18-25957. Epub
[PubMed PMID: 30817826]
[14]
Gessesse SA, Teshome AW. Prevalence of myopia among secondary school students in Welkite town: South-Western Ethiopia. BMC ophthalmology. 2020 May 4:20(1):176. doi: 10.1186/s12886-020-01457-2. Epub 2020 May 4
[PubMed PMID: 32366285]
[15]
Maduka Okafor FC, Okoye OI, Eze BI. Myopia: a review of literature. Nigerian journal of medicine : journal of the National Association of Resident Doctors of Nigeria. 2009 Apr-Jun:18(2):134-8
[PubMed PMID: 19630316]
[16]
Alvarez-Peregrina CC, Sanchez-Tena MAMA, Martinez-Perez CC, Villa-Collar CC. Prevalence and Risk Factors of Myopia in Spain. Journal of ophthalmology. 2019:2019():3419576. doi: 10.1155/2019/3419576. Epub 2019 Aug 18
[PubMed PMID: 31531234]
[17]
Yeter V, Koçak N, Eser-Ozturk H. Changes in corneal thickness, upper and lower tear film in seasonal allergic conjunctivitis by steroid treatment: anterior segment optical coherence tomography study. International ophthalmology. 2020 Sep:40(9):2275-2281. doi: 10.1007/s10792-020-01410-8. Epub 2020 May 14
[PubMed PMID: 32409942]
[18]
Gellrich MM, Kandzia C. [Purkinje images in slit lamp videography : Video article]. Der Ophthalmologe : Zeitschrift der Deutschen Ophthalmologischen Gesellschaft. 2016 Sep:113(9):789-93. doi: 10.1007/s00347-016-0343-4. Epub
[PubMed PMID: 27558688]
[19]
Tang SM, Chan RY, Bin Lin S, Rong SS, Lau HH, Lau WW, Yip WW, Chen LJ, Ko ST, Yam JC. Refractive Errors and Concomitant Strabismus: A Systematic Review and Meta-analysis. Scientific reports. 2016 Oct 12:6():35177. doi: 10.1038/srep35177. Epub 2016 Oct 12
[PubMed PMID: 27731389]
Level 1 (high-level) evidence
[20]
Smith EL 3rd, Hung LF, Arumugam B, Wensveen JM, Chino YM, Harwerth RS. Observations on the relationship between anisometropia, amblyopia and strabismus. Vision research. 2017 May:134():26-42. doi: 10.1016/j.visres.2017.03.004. Epub 2017 Apr 18
[PubMed PMID: 28404522]
[21]
Stephenson KAJ, Power B, Malata D, Quill B, Murphy CC, Power WJ. Management of Keratoconus in Down Syndrome and Other Intellectual Disability. Cornea. 2022 Apr 1:41(4):456-461. doi: 10.1097/ICO.0000000000002793. Epub
[PubMed PMID: 35244626]
[22]
Arnon R, Rozen-Knisbacher I, Yahalomi T, Stanescu N, Niazov Y, Goldberg D, Sharabi-Nov A, Mostovoy D. Rise of the Machines? Comparison of Cycloplegic Refraction Using Retinoscopy and the Retinomax K-Plus 5 in Children. Journal of pediatric ophthalmology and strabismus. 2022 Nov-Dec:59(6):380-387. doi: 10.3928/01913913-20220211-01. Epub 2022 Mar 11
[PubMed PMID: 35275776]
[23]
Padhy D, Bharadwaj SR, Nayak S, Rath S, Das T. Does the Accuracy and Repeatability of Refractive Error Estimates Depend on the Measurement Principle of Autorefractors? Translational vision science & technology. 2021 Jan:10(1):2. doi: 10.1167/tvst.10.1.2. Epub 2021 Jan 5
[PubMed PMID: 33505769]
[24]
Chaurasiya RK. Refractive changes during off-the-axis retinoscopy in myopia. Indian journal of ophthalmology. 2022 Mar:70(3):779-781. doi: 10.4103/ijo.IJO_1123_21. Epub
[PubMed PMID: 35225512]
[25]
Liu X, Feng J, Wang L, Tong H, Chen W. Spot Refractive Screening With or Without Maximum Atropine Cycloplegia in Preschool Chinese Children. Journal of pediatric ophthalmology and strabismus. 2021 May-Jun:58(3):146-153. doi: 10.3928/01913913-20210128-01. Epub 2021 May 1
[PubMed PMID: 34039157]
[26]
Karabulut M, Karabulut S, Karalezli A. Refractive outcomes of table-mounted and hand-held auto-refractometers in children: an observational cross-sectional study. BMC ophthalmology. 2021 Dec 9:21(1):424. doi: 10.1186/s12886-021-02199-5. Epub 2021 Dec 9
[PubMed PMID: 34879852]
Level 2 (mid-level) evidence
[27]
Mirzajani A, Qasemi F, Asharlous A, Yekta A, Doostdar A, Khabazkhoob M, Hashemi H. Are the results of handheld auto-refractometer as valid as the result of table-mounted refractometer? Journal of current ophthalmology. 2019 Sep:31(3):305-311. doi: 10.1016/j.joco.2018.10.012. Epub 2018 Nov 22
[PubMed PMID: 31528766]
[28]
Domínguez-Vicent A, Al-Soboh L, Brautaset R, Venkataraman AP. Effect of Instrument Design and Technique on the Precision and Accuracy of Objective Refraction Measurement. Journal of clinical medicine. 2020 Sep 23:9(10):. doi: 10.3390/jcm9103061. Epub 2020 Sep 23
[PubMed PMID: 32977411]
[29]
Wan XH, Lin Z, Cai XG, Qiao LY, Yang XD, Wang NL, Liang YB. [Comparison between binocular, open-field auto ref/keratometer and conventional autorefractor]. [Zhonghua yan ke za zhi] Chinese journal of ophthalmology. 2012 Jun:48(6):519-23
[PubMed PMID: 22943807]
Level 2 (mid-level) evidence
[30]
Vargas V, Radner W, Allan BD, Reinstein DZ, Burkhard Dick H, Alió JL, Near vision and accommodation committee of the American-European Congress of Ophthalmology (AECOS). Methods for the study of near, intermediate vision, and accommodation: an overview of subjective and objective approaches. Survey of ophthalmology. 2019 Jan-Feb:64(1):90-100. doi: 10.1016/j.survophthal.2018.08.003. Epub 2018 Sep 3
[PubMed PMID: 30189234]
Level 3 (low-level) evidence
[31]
McKendrick AM, Brennan NA. Clinical evaluation of refractive techniques. Journal of the American Optometric Association. 1995 Dec:66(12):758-65
[PubMed PMID: 8557954]
[32]
Schittkowski M, Hucks-Sievers S, Krentz H, Guthoff R. [Accuracy of the autorefractor power refractor in clinical work--a comparative study]. Klinische Monatsblatter fur Augenheilkunde. 2005 Dec:222(12):983-92
[PubMed PMID: 16380885]
Level 2 (mid-level) evidence
[33]
Namkung S, Boyle AB, Li Y, Gokul A, McGhee C, Ziaei M. Repeatability and Agreement of Horizontal Corneal Diameter Measurements Between Scanning-Slit Topography, Dual Rotating Scheimpflug Camera With Placido Disc Tomography, Placido Disc Topography, and Optical Coherence Tomography. Cornea. 2022 Nov 1:41(11):1392-1397. doi: 10.1097/ICO.0000000000002964. Epub 2021 Dec 22
[PubMed PMID: 34935660]
[34]
Cairns G, McGhee CN. Orbscan computerized topography: attributes, applications, and limitations. Journal of cataract and refractive surgery. 2005 Jan:31(1):205-20
[PubMed PMID: 15721715]
[35]
Motlagh MN, Moshirfar M, Murri MS, Skanchy DF, Momeni-Moghaddam H, Ronquillo YC, Hoopes PC. Pentacam® Corneal Tomography for Screening of Refractive Surgery Candidates: A Review of the Literature, Part I. Medical hypothesis, discovery & innovation ophthalmology journal. 2019 Fall:8(3):177-203
[PubMed PMID: 31598520]
[36]
Clover J. Slit-Lamp Biomicroscopy. Cornea. 2018 Jun:37 Suppl 1():S5-S6. doi: 10.1097/ICO.0000000000001641. Epub
[PubMed PMID: 29734202]
[37]
Martínez-Abad A, Piñero DP. Pellucid marginal degeneration: Detection, discrimination from other corneal ectatic disorders and progression. Contact lens & anterior eye : the journal of the British Contact Lens Association. 2019 Aug:42(4):341-349. doi: 10.1016/j.clae.2018.11.010. Epub 2018 Nov 22
[PubMed PMID: 30473322]
[38]
Gellrich MM. A simple method for panretinal imaging with the slit lamp. International ophthalmology. 2016 Dec:36(6):775-780
[PubMed PMID: 26879088]
[39]
Goss DA. Effect of spectacle correction on the progression of myopia in children--a literature review. Journal of the American Optometric Association. 1994 Feb:65(2):117-28
[PubMed PMID: 8144839]
[40]
Schiefer U, Kraus C, Baumbach P, Ungewiß J, Michels R. Refractive errors. Deutsches Arzteblatt international. 2016 Oct 14:113(41):693-702. doi: 10.3238/arztebl.2016.0693. Epub
[PubMed PMID: 27839543]
[41]
Leung TW, Li RW, Kee CS. Blue-Light Filtering Spectacle Lenses: Optical and Clinical Performances. PloS one. 2017:12(1):e0169114. doi: 10.1371/journal.pone.0169114. Epub 2017 Jan 3
[PubMed PMID: 28045969]
[42]
Stephens GL, Polasky M. New options for aniseikonia correction: the use of high index materials. Optometry and vision science : official publication of the American Academy of Optometry. 1991 Nov:68(11):899-906
[PubMed PMID: 1766654]
[43]
Paulson CA, Price JJ, Koch KW, Kim CG, Oh JH, Lin L, Subramanian AN, Zhang B, Amin J, Mayolet A, Kosik Williams C, Hart SD. Industrial-grade anti-reflection coatings with extreme scratch resistance. Optics letters. 2019 Dec 15:44(24):5977-5980. doi: 10.1364/OL.44.005977. Epub
[PubMed PMID: 32628197]
Level 3 (low-level) evidence
[44]
Sankaridurg P. Contact lenses to slow progression of myopia. Clinical & experimental optometry. 2017 Sep:100(5):432-437. doi: 10.1111/cxo.12584. Epub 2017 Jul 28
[PubMed PMID: 28752898]
[45]
Bhamra TS, Tighe BJ. Mechanical properties of contact lenses: The contribution of measurement techniques and clinical feedback to 50 years of materials development. Contact lens & anterior eye : the journal of the British Contact Lens Association. 2017 Apr:40(2):70-81. doi: 10.1016/j.clae.2016.11.005. Epub 2016 Nov 22
[PubMed PMID: 27884616]
[46]
Scanzera AC, Deeley M, Joslin C, McMahon TT, Shorter E. Contact Lens Prescribing Trends for Keratoconus at an Academic Medical Center: Increased Utilization of Scleral Lenses for Severe Disease. Eye & contact lens. 2022 Feb 1:48(2):58-62. doi: 10.1097/ICL.0000000000000869. Epub
[PubMed PMID: 35058416]
[47]
Lim CHL, Stapleton F, Mehta JS. Review of Contact Lens-Related Complications. Eye & contact lens. 2018 Nov:44 Suppl 2():S1-S10. doi: 10.1097/ICL.0000000000000481. Epub
[PubMed PMID: 29373389]
[48]
Covey M, Sweeney DF, Terry R, Sankaridurg PR, Holden BA. Hypoxic effects on the anterior eye of high-Dk soft contact lens wearers are negligible. Optometry and vision science : official publication of the American Academy of Optometry. 2001 Feb:78(2):95-9
[PubMed PMID: 11265932]
[49]
Chalmers RL, Wagner H, Mitchell GL, Lam DY, Kinoshita BT, Jansen ME, Richdale K, Sorbara L, McMahon TT. Age and other risk factors for corneal infiltrative and inflammatory events in young soft contact lens wearers from the Contact Lens Assessment in Youth (CLAY) study. Investigative ophthalmology & visual science. 2011 Aug 24:52(9):6690-6. doi: 10.1167/iovs.10-7018. Epub 2011 Aug 24
[PubMed PMID: 21527379]
[50]
Sankaridurg PR, Vuppala N, Sreedharan A, Vadlamudi J, Rao GN. Gram negative bacteria and contact lens induced acute red eye. Indian journal of ophthalmology. 1996 Mar:44(1):29-32
[PubMed PMID: 8828303]
[51]
Vinciguerra R, Borgia A, Tredici C, Vinciguerra P. Excimer laser tissue interactions in the cornea. Experimental eye research. 2021 May:206():108537. doi: 10.1016/j.exer.2021.108537. Epub 2021 Mar 12
[PubMed PMID: 33716013]
[52]
Lu LW, Rocha-de-Lossada C, Rachwani-Anil R, Flikier S, Flikier D. The role of posterior corneal power in 21st century biometry: A review. Journal francais d'ophtalmologie. 2021 Sep:44(7):1052-1058. doi: 10.1016/j.jfo.2020.11.031. Epub 2021 Jun 17
[PubMed PMID: 34148699]
[53]
Mello GR, Rocha KM, Santhiago MR, Smadja D, Krueger RR. Applications of wavefront technology. Journal of cataract and refractive surgery. 2012 Sep:38(9):1671-83. doi: 10.1016/j.jcrs.2012.07.004. Epub
[PubMed PMID: 22906449]
[54]
Ehlke GL, Krueger RR. Laser Vision Correction in Treating Myopia. Asia-Pacific journal of ophthalmology (Philadelphia, Pa.). 2016 Nov/Dec:5(6):434-437
[PubMed PMID: 27898448]
[55]
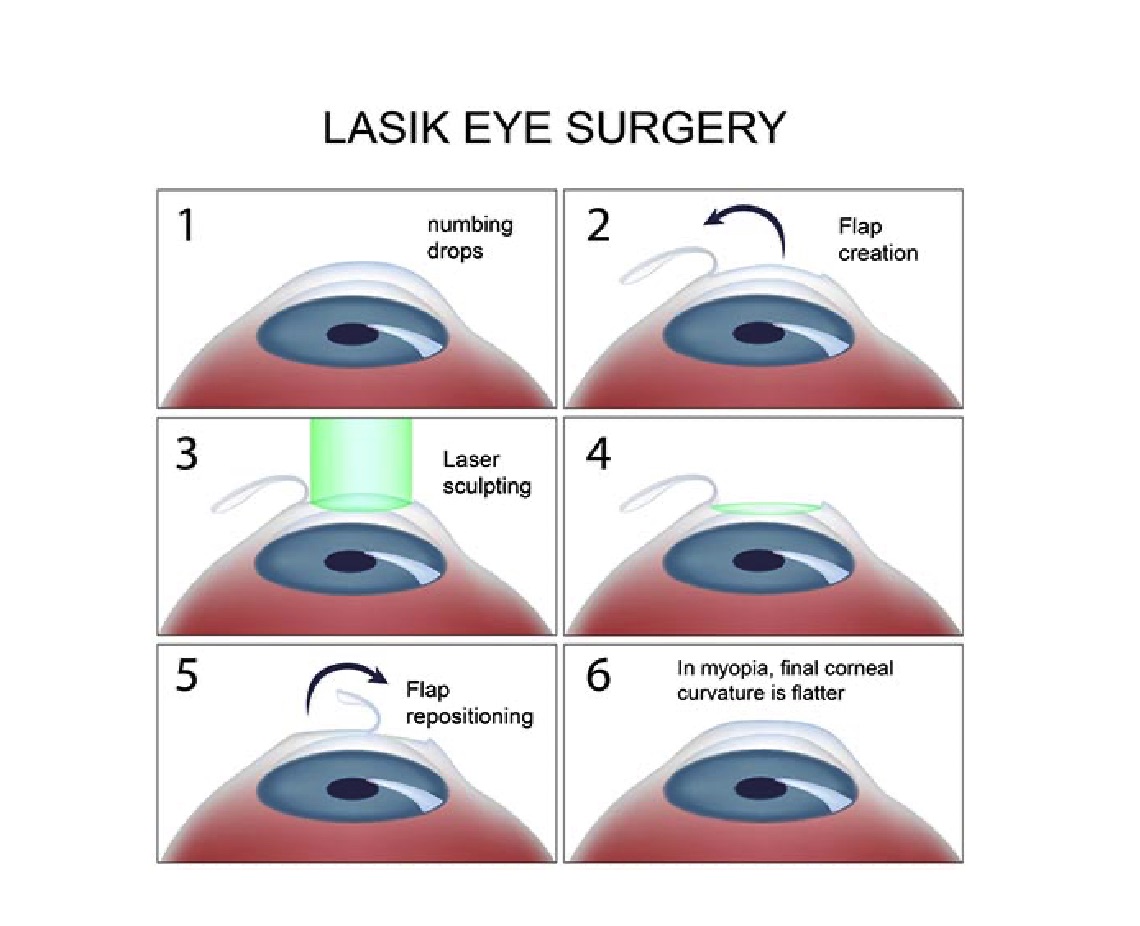
Ambrósio R Jr, Wilson S. LASIK vs LASEK vs PRK: advantages and indications. Seminars in ophthalmology. 2003 Mar:18(1):2-10
[PubMed PMID: 12759854]
[56]
Fogla R, Luthra G, Chhabra A, Gupta K, Dalal R, Khamar P. Preferred practice patterns for photorefractive keratectomy surgery. Indian journal of ophthalmology. 2020 Dec:68(12):2847-2855. doi: 10.4103/ijo.IJO_2178_20. Epub
[PubMed PMID: 33229660]
[57]
Bashir ZS, Ali MH, Anwar A, Ayub MH, Butt NH. Femto-lasik: The recent innovation in laser assisted refractive surgery. JPMA. The Journal of the Pakistan Medical Association. 2017 Apr:67(4):609-615
[PubMed PMID: 28420926]
[58]
Lee JK,Chuck RS,Park CY, Femtosecond laser refractive surgery: small-incision lenticule extraction vs. femtosecond laser-assisted LASIK. Current opinion in ophthalmology. 2015 Jul
[PubMed PMID: 26058022]
Level 3 (low-level) evidence
[59]
Wilson SE. Biology of keratorefractive surgery- PRK, PTK, LASIK, SMILE, inlays and other refractive procedures. Experimental eye research. 2020 Sep:198():108136. doi: 10.1016/j.exer.2020.108136. Epub 2020 Jul 10
[PubMed PMID: 32653492]
[60]
Jaycock PD, Lobo L, Ibrahim J, Tyrer J, Marshall J. Interferometric technique to measure biomechanical changes in the cornea induced by refractive surgery. Journal of cataract and refractive surgery. 2005 Jan:31(1):175-84
[PubMed PMID: 15721710]
[61]
Kenia VP, Kenia RV, Pirdankar OH. Short term changes in corneal stress-strain index and other corneal biomechanical parameters post-laser in situ keratomileusis. Indian journal of ophthalmology. 2021 Oct:69(10):2650-2656. doi: 10.4103/ijo.IJO_3365_20. Epub
[PubMed PMID: 34571607]
[62]
Tse SM, Farley ND, Tomasko KR, Amin SR. Intraoperative LASIK Complications. International ophthalmology clinics. 2016 Spring:56(2):47-57. doi: 10.1097/IIO.0000000000000110. Epub
[PubMed PMID: 26938337]
[63]
Davis EA, Hardten DR, Lindstrom RL. LASIK complications. International ophthalmology clinics. 2000 Summer:40(3):67-75
[PubMed PMID: 10941647]
[64]
Martínez-Plaza E, López-Miguel A, Holgueras A, Barraquer RI, Alió JL, Maldonado MJ. Phakic intraocular lenses: Recent advances and innovations. Archivos de la Sociedad Espanola de Oftalmologia. 2020 Apr:95(4):178-187. doi: 10.1016/j.oftal.2020.02.001. Epub 2020 Mar 7
[PubMed PMID: 32156485]
Level 3 (low-level) evidence
[65]
Alió JL, Abdelrahman AM, Javaloy J, Iradier MT, Ortuño V. Angle-supported anterior chamber phakic intraocular lens explantation causes and outcome. Ophthalmology. 2006 Dec:113(12):2213-20
[PubMed PMID: 16996608]
[66]
Pinto C, Monteiro T, Franqueira N, Faria-Correia F, Mendes J, Vaz F. Posterior chamber collamer phakic intraocular lens implantation: Comparison of efficacy and safety for low and moderate-to-high myopia. European journal of ophthalmology. 2021 Apr 22:():11206721211012861. doi: 10.1177/11206721211012861. Epub 2021 Apr 22
[PubMed PMID: 33887990]
[67]
Subudhi P, Patro S, Agarwal P, Khan Z, Subudhi BNR, Mekap C, Padhi A. Safety and Efficacy of a New Posterior Chamber Phakic Intraocular Lens in Cases of High Myopia: Early Results. Clinical ophthalmology (Auckland, N.Z.). 2020:14():3681-3689. doi: 10.2147/OPTH.S270690. Epub 2020 Oct 30
[PubMed PMID: 33162752]
Level 3 (low-level) evidence
[68]
Yildirim TM, Khoramnia R, Son HS, Mayer CS, Łabuz G, Munro DJ, Auffarth GU. Reasons for explantation of phakic intraocular lenses and associated perioperative complications: cross-sectional explant registry analysis. BMC ophthalmology. 2021 Feb 12:21(1):80. doi: 10.1186/s12886-021-01847-0. Epub 2021 Feb 12
[PubMed PMID: 33579238]
Level 2 (mid-level) evidence
[69]
Subudhi P, Patro S, Khan Z, Subudhi BNR, Sitaram S. Refractive outcomes of implantation of an implantable phakic copolymer lens with peripheral holes in the intraocular posterior chamber in moderate to high myopia patients: a single-surgeon series. Clinical ophthalmology (Auckland, N.Z.). 2019:13():1887-1894. doi: 10.2147/OPTH.S215821. Epub 2019 Sep 23
[PubMed PMID: 31576104]
[70]
Huang D, Schallhorn SC, Sugar A, Farjo AA, Majmudar PA, Trattler WB, Tanzer DJ. Phakic intraocular lens implantation for the correction of myopia: a report by the American Academy of Ophthalmology. Ophthalmology. 2009 Nov:116(11):2244-58. doi: 10.1016/j.ophtha.2009.08.018. Epub
[PubMed PMID: 19883852]
[71]
Fairaq R, Almutlak M, Almazyad E, Badawi AH, Ahad MA. Outcomes and complications of implantable collamer lens for mild to advance keratoconus. International ophthalmology. 2021 Jul:41(7):2609-2618. doi: 10.1007/s10792-021-01820-2. Epub 2021 Mar 27
[PubMed PMID: 33772698]
[72]
Yao Y, Lu Q, Wei L, Cheng K, Lu Y, Zhu X. Efficacy and complications of cataract surgery in high myopia. Journal of cataract and refractive surgery. 2021 Nov 1:47(11):1473-1480. doi: 10.1097/j.jcrs.0000000000000664. Epub
[PubMed PMID: 33929806]
[73]
Chong EW, Mehta JS. High myopia and cataract surgery. Current opinion in ophthalmology. 2016 Jan:27(1):45-50. doi: 10.1097/ICU.0000000000000217. Epub
[PubMed PMID: 26569522]
Level 3 (low-level) evidence
[74]
Iusef IuN, Iusef SIu, Reznikova EV, Vvedenskiĭ AS. [Cataract surgery in patients with high myopia]. Vestnik oftalmologii. 2005 Nov-Dec:121(6):47-9
[PubMed PMID: 16405068]
[75]
Lam JK, Chan TC, Ng AL, Chow VW, Wong VW, Jhanji V. Outcomes of cataract operations in extreme high axial myopia. Graefe's archive for clinical and experimental ophthalmology = Albrecht von Graefes Archiv fur klinische und experimentelle Ophthalmologie. 2016 Sep:254(9):1811-7. doi: 10.1007/s00417-016-3414-y. Epub 2016 Jun 16
[PubMed PMID: 27313161]
[76]
Khalid K, Padda J, Pokhriyal S, Hitawala G, Khan MS, Upadhyay P, Cooper AC, Jean-Charles G. Pseudomyopia and Its Association With Anxiety. Cureus. 2021 Aug:13(8):e17411. doi: 10.7759/cureus.17411. Epub 2021 Aug 24
[PubMed PMID: 34589322]
[77]
Ohno-Matsui K, Lai TY, Lai CC, Cheung CM. Updates of pathologic myopia. Progress in retinal and eye research. 2016 May:52():156-87. doi: 10.1016/j.preteyeres.2015.12.001. Epub 2016 Jan 6
[PubMed PMID: 26769165]
[78]
Shukla M, Ahuja OP. Peripheral retina in myopia. Indian journal of ophthalmology. 1983 Nov-Dec:31(6):719-22
[PubMed PMID: 6676251]
[79]
Kumar A, Chawla R, Kumawat D, Pillay G. Insight into high myopia and the macula. Indian journal of ophthalmology. 2017 Feb:65(2):85-91. doi: 10.4103/ijo.IJO_863_16. Epub
[PubMed PMID: 28345561]