Clinical Significance
To interpret the fluorescein angiography images, knowledge of the following terms is important:
Hyperfluorescence
- Preinjection Fluorescence
- Autofluorescence: It is the property of certain biological structures to emit fluorescent light when they absorb light in the absence of fluorescein dye. This occurs due to the presence of fluorophores like lipofuscins or porphyrins. A normal optic disc does not have any fluorophores. Optic disc drusen show excessive accumulation of mitochondria. Porphyrins are present in mitochondria. Optic disc-drusen superficial or buried show autofluorescence due to porphyrins in these dysfunctional mitochondria.[38][39] The role of calcium as a cause for autofluorescence of optic disc drusen needs further research.[40] Retinal astrocytic hamartomas also show autofluorescence.[41]
- Pseudofluorescence: This occurs due to the presence of old filters or allowing transmission of wavelength beyond blue and green light. This nonfluorescent light is reflected from various lesions, including exudates, sclera, and scar, and is perceived as a bright spot on the preinjection angiogram images. Dehemoglobinised blood also reflects blue light that passes through the barrier filters causing it to cause hyperfluorescence on preinjection images. This may happen even if the filters are not worn out or old if there are overlapping wavelengths of the barrier and excitation filters.
- Transmitted fluorescence: Healthy RPE blocks the choroidal fluorescence. In cases with atrophy of the RPE, the visibility of the underlying choroidal fluorescence is enhanced. This is known as transmitted fluorescence or window defect. This fluorescence follows the same pattern as the flow of dye through normal ocular circulation. It appears early and peaks early during the choroidal filling phase. It is of the same shape as that of pigment epithelial atrophy. Therefore, it doesn't increase in size or shape with time. The fluorescence fades as and when the dye washes out of the choroidal circulation.
- Leak: Fluorescence that shows an increase in size and intensity with time is defined as a leak, which happens due to dye leakage in a potential space.
- Pooling: In this case, the fluorescence shows an increase in intensity with time. However, the size of fluorescence remains the same throughout the angiogram. This is because of the dye collection in a defined anatomic space, either subretinal or sub-RPE space.
- Stain: Certain structures retain the fluorescein dye and show hyperfluorescence in the later stages of the angiography. This is known as staining.
Hypofluorescence
The area of reduced or absent fluorescence is seen as a dark area on the angiogram.
- Blocked fluorescence: This shows reduced visibility of the fluorescence of underlying retinal or choroidal circulation due to a barrier located anterior to the respective circulation. This needs to be correlated clinically or with the color photograph. The shape of the hypofluorescent area will correlate with that of the barrier, for example, bleeding in the subhyaloid space or subretinal space. Opacities involving the cornea, anterior chamber, lens, or vitreous affect the quality of the photograph as a whole and are not seen as hypofluorescent areas. However, asteroid hyalosis does not affect the angiography imaging because the light scattered by the suspended particles is filtered by the barrier filter. This makes FFA a better tool for investigation than the clinical examination with an ophthalmoscope in cases with asteroid hyalosis.[42]
- Vascular filling defect: Absence of visibility of fluorescence due to lack of retinal or choroidal circulation perfusion. A total lack of perfusion results in hypofluorescence that persists through all the phases of the angiogram. A partial lack of perfusion results in delayed filling of the circulation or delayed fluorescence.
A few clinical scenarios have been discussed herewith.
Epiretinal Membrane (ERM)
Abnormal retinal vessel tortuosity and dilation may be seen at the macula, and this is due to the contraction of the internal limiting membrane by the epiretinal membrane. In the late phases of the angiogram, small leakage from these vessels may be seen. Hyperfluorescence at the fovea due to cystoid macular edema may also be present. Other features of retinal vein occlusion, diabetic retinopathy, or coexisting pathology may be seen in cases of secondary epiretinal membranes.
RPE Rip
The RPE tears at one edge of a pigment epithelium detachment, and the RPE layer scrolls at the other end. This is seen in cases of PED with underlying CNVM or PED in cases of CSCR. The characteristic feature of angiography is transmitted hyperfluorescence in the area of the tear. This area of the exposed choroid may show staining in the later phases. The area where the RPE scrolls underneath shows blocked fluorescence.[19] This blocked fluorescence persists throughout the angiogram. In cases with associated CNVM, the CNVM usually lies at the end of the scrolled RPE.
Drusen
Areas of hard drusens show transmitted fluorescence due to atrophy of the overlying RPE. Areas of soft drusen show late mild hyperfluorescence or staining. This is due to the hydrophobic content of the soft drusen. Drusenoid pigment epithelium detachment also shows hyperfluorescence that starts in the mid-arteriovenous phase increases in the late venous phase and persists in the recirculation phase. Cuticular or basal laminar drusen may give a starry sky appearance. Subretinal drusenoid deposits or reticular pseudodrusen may not be evident on FFA or show minimal hypofluorescence.[43]
Serous PED
Serous PED shows fluorescence that starts in the early phase and increases uniformly in intensity in the subsequent phases. The borders of the lesion are well-demarcated with no leakage. The fluorescein dye pools in the sub-RPE space and causes this pattern of fluorescence. Cases of serous PED may show a notch in a few instances. These cases have an underlying CNVM.[44]
Choroidal Neovascular Membrane (CNVM)
Classic CNVM is identified by a well-defined lacy network of early hyperfluorescence, which shows an increase in the intensity and size of the fluorescence in the subsequent phases. This is due to the progressive leakage of the dye. The boundaries of the area of hyperfluorescence become indistinct due to leakage.[45]
Occult CNVM shows two patterns:
- Fibrovascular PED: This is characterized by irregularly elevated RPE with a gradually progressive stippled leak on FFA. Alternatively, a notched serous PED may be seen where the notch or hot spot harbors the CNVM. Such serous PEDs are called vascularized serous PEDs.
- Late leakage from an undetermined source: This is not recognizable until the later phases of the angiogram, where hyperfluorescence is seen. However, the exact source of the leakage cannot be distinguished.[45]
ICGA is a better investigation tool for imaging occult CNVM as the visibility of the CNVM network is better than on FFA.[46][47] ICGA also helps identify cases of idiopathic polypoidal choroidal vasculopathy.[48]
Central Serous Chorioretinopathy (CSCR)
Inkblot leak is seen as dot hyperfluorescence in the early phases, which increases in the following phases. The increase in size is limited as it does not fill the entire area of neurosensory detachment. The number of leaks may vary, and multifocal leakage points may be seen. In a few cases of CSCR, the dot leak is succeeded by a mushroom pattern leak. This is because the dye ascends up and then spreads laterally. The osmotic gradient of the fluid allows rapid diffusion of dye to the superior part of the detachment. Proteins accumulate in the inferior part delaying the entry of dye. This is known as smokestack or umbrella pattern leakage.[49] Chronic cases of CSCR may show transmitted fluorescence at the posterior pole, and a descending RPE track may be noted as a window defect in a teardrop pattern. These are areas of RPE alteration resembling prior fluid collection. The inkblot pattern of leakage is more prevalent than the smokestack pattern. Diffuse type of leakage is the least prevalent.[50][51]
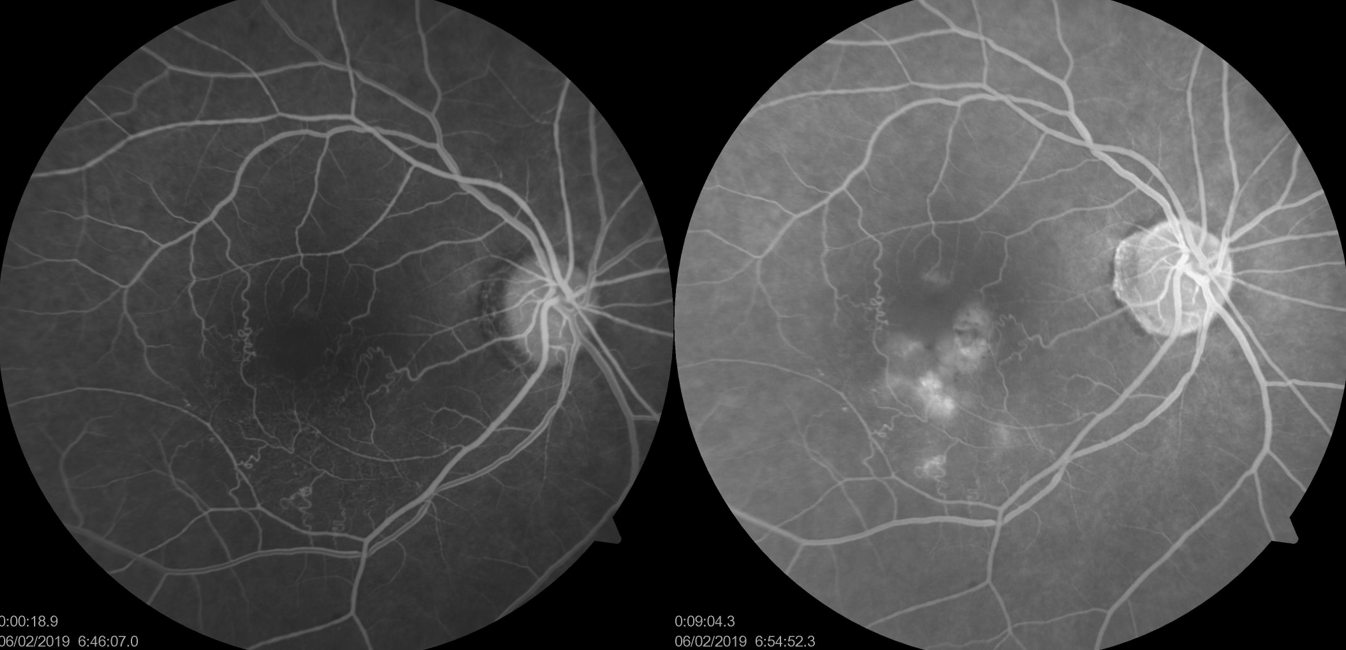
Cystoid Macular Edema (CME)
Leakage from the perifoveal capillaries is seen earlier in the arteriovenous phase, followed by dye accumulation in a petaloid pattern or a more diffuse hyperfluorescence in the later phases.[52]
Diabetic Retinopathy
Microaneurysms appear as dots that hyperfluoresce.[53] These areas of dot hyperfluorescence should correspond with microaneurysms on color photographs of the fundus. This helps differentiate microaneurysms from dot hyperfluorescence due to RPE defects. FFA is a better investigative tool to pick up microaneurysms, and subtle changes in diabetic retinopathy become evident on FFA. An additional number of microaneurysms are visible on FFA compared to clinical examination with the naked eye. Leakage in diabetic macular edema was historically graded as focal or diffuse based on the FFA features. The term focal leak was used for eyes where more than 67 percent of the leakage originated from microaneurysms. The term diffuse leak was used in cases where less than 33 percent of leakage was noted from microaneurysms (most of the leakage was noted from dilated capillaries).[54]
Clinically, focal diabetic macular edema is characterized by a circinate ring of hard exudates around a microaneurysm. Areas of superficial and deep hemorrhages and areas of nonperfusion are visualized as areas of hypofluorescence. The former are areas of blocked fluorescence and need to be correlated clinically. The latter are areas of ischemia. The foveal avascular zone may also show enlargement in size due to ischemia (macular ischemia) and should be examined carefully. In cases with diabetic retinopathy and unexplained vision loss, distortion of the anatomy of perifoveal capillaries indicates macular ischemia.[55][56][57][58]
Abnormal vessels connecting two veins in the areas of capillary nonperfusion may also be seen in cases of diabetic retinopathy. These do not leak, and these are intraretinal microvascular abnormalities (IRMA). IRMA is present within the retina, unlike neovascular vessels, which tend to protrude into the vitreous. Neovascularization networks leak dye from the early phases of the angiogram. Venous beading and loops can also be better appreciated on fluorescein angiography.
Vogt-Koyanagi-Harada Syndrome
Multiple hyperfluorescent point leaks are seen in the arteriovenous phase, increasing intensity in the venous phase. In the later phases, extensive dye pooling is seen in neurosensory detachment, unlike CSCR, where the dye does not fill the entire detachment area, even in the late phases. Another differentiating feature from CSCR is the absence of pigment epithelium detachments in eyes with Vogt-Koyanagi-Harada syndrome. The presence of intraocular inflammation in VKH causes staining of the disc in the late phases.
Best Dystrophy
These are inherited macular dystrophy characterized by yellowish subretinal deposits and sometimes subretinal collections of fluid, depending on the stage of the disease. With time these deposits may be partially absorbed with the gravitation of the remaining deposits. In the early stage, also known as the vitelliform stage, fundus fluorescein angiography shows blocked choroidal fluorescence in the area of the subretinal yellowish deposit.[59]
This area may slightly hyperfluoresce in the later stages or may remain hypofluorescent. In the later stages, when the yellowish deposits have reabsorbed, the FFA shows window defects in the area of absorbed deposits due to RPE changes. The area where the yellowish deposit gravitates shows blocked fluorescence.[60] Fibrous scar, when present in the later stages of the disease, shows staining on FFA. FFA is also useful in identifying CNVM in these cases. In cases of adult-onset vitelliform macular dystrophy, FFA is useful in differentiating these cases from CSCR.
Malignant Melanoma
The tumor shows a characteristic double circulation sign where the intrinsic vascularity of the tumor mass is seen along with normal retinal circulation.
Choroidal Hemangioma
These tumors are vascular hamartomas. They show intense hyperfluorescence in the early arterial phase itself because of their choroidal origin. The fluorescence increases with the leakage of dye in the later phase because of the vascularity of the tumor.[61]
Choroidal Metastasis
The choroidal metastases show hypofluorescence in the arterial phase and then hyperfluoresce in the venous phase of the angiogram. Pinpoint areas of fluorescence appear in the late venous phase at the margins of the lesion. Variable fluorescein patterns may be seen depending on the type of tumor mass.[62] Some lesions block choroidal fluorescence throughout the angiography, some show progressive fluorescence in the late phases, some show fluorescence in the early phase itself, and then show leakage in the subsequent phases.[62] However, FFA may show multiple pinpoint leaks in choroidal metastasis. FFA helps to differentiate it from other causes of choroidal mass, like choroidal hemangioma or melanoma. Other pathologies that may show multiple pinpoint leaks on FFA include[63][64][65][66]
- posterior scleritis,
- toxemia of pregnancy,
- hypertensive choroidopathy,
- IgA nephropathy,
- Vogt-Koyanagi-Harada disease,
- sympathetic ophthalmia,
- punctate inner choroidopathy, and
- leukemia or lymphoma of the uveal tract.
Choroidal Granuloma
They are discrete yellowish-white elevated lesions with indistinct borders. They show hypofluorescence in the early phases and hyperfluorescence in the late phases of angiography.[67] Common causes include sarcoidosis and tuberculosis.
Retinal Capillary Hemangioma
The feeder arteriole fills earlier with the rapid flow of the dye into the venule. The tumor shows intense hyperfluorescence. Angiography is useful in these cases as it identifies smaller angiomas that are not easily visible on naked eye examination.[61][68] These lesions are typically seen in Von Hippel-Lindau's disease.[61]
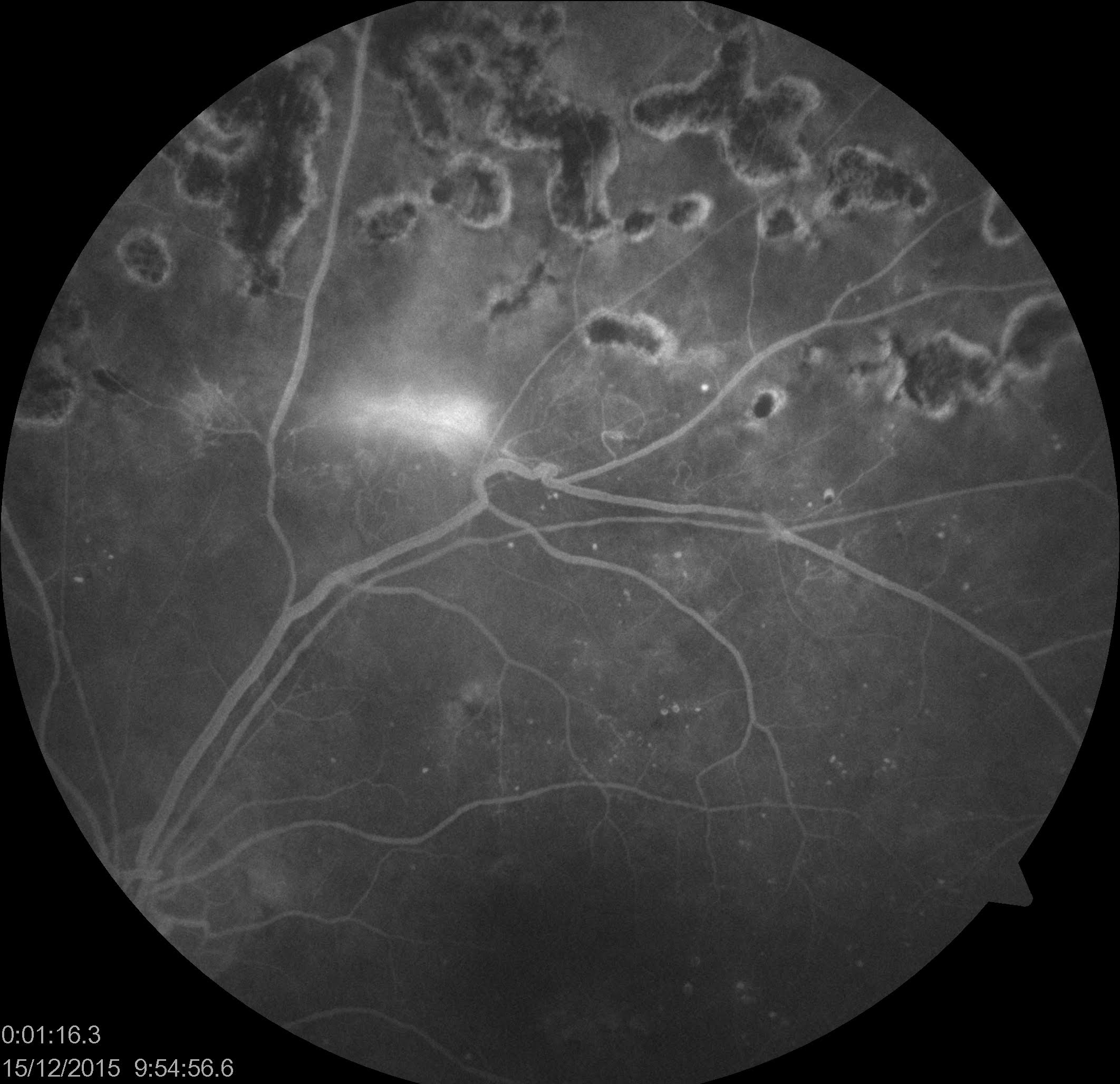
Branch Retinal Vein Occlusion (BRVO)
Recent vein occlusion cases will show blocked fluorescence corresponding to retinal hemorrhages. Therefore, it is better to wait for the hemorrhages to resolve. In branch retinal vein occlusion (BRVO) cases, a delay in filling the affected vein is noted. Dilated capillaries, microaneurysms, and telangiectatic changes may also be noted. Areas of capillary nonperfusion may be present and may involve the foveal avascular zone. Macular edema is noted as late leakage of dye by the perifoveal capillaries. Late staining of the affected vessels may also be seen. In chronic cases of BRVO, neovascularization of the retina or the optic disc may also be seen. These vessels show fluorescence in the early phases and excessive leakage of the dye subsequently.
Central Retinal Vein Occlusion (CRVO)
Tortuosity and dilatation of the veins are well-demarcated. The capillaries at the disc are also dilated. The filling of dye in the veins is delayed. A delay in the emptying of the veins is also noted. Leakage of dye from the perifoveal capillaries may also be seen. Capillaries may show microaneurysmal changes. Staining of the wall of the vein may be seen. If the size of the capillary nonperfusion area in CRVO is more than ten disc areas, the diagnosis of ischemic CRVO or non-perfused CRVO is confirmed.[69]
Myopic CNVM
FFA is useful in evaluating patients with myopia who have a history of recent vision loss. CNVM in myopia may not always be visible on clinical examination with an ophthalmoscope. Myopic CNVM on FFA shows hyperfluorescence in the early phases, followed by leakage. The hyperfluorescence increases in size and intensity. However, these changes may be very subtle.[70] Associated subretinal hemorrhage present in these cases shows blocked choroidal fluorescence. Lacquer cracks are visible as hyperfluorescent lines due to transmitted fluorescence. Staining of the sclera in the late phases in the areas of RPE atrophy may also be seen at the posterior pole.
Ocular Ischemic Syndrome
A delay in the filling of the choroidal circulation is noted. The retinal arterioles also show delayed entry of dye in the late phases. Due to incomplete filling of the arterioles, the leading edge of the dye may be seen in the arterioles. Microaneurysms may be seen distributed in the mid-periphery of the retina. Other features include capillary nonperfusion, perivascular staining, and staining of the optic disc.[71]
Takayasu Arteritis
It is an autoimmune disease affecting the aorta, aortic arch, and its main branches. Granulomatous inflammation involving all the layers of the arterial wall results in the narrowing of the lumen of the affected blood vessels and ischemia. Involvement of the carotid artery causes ischemic changes in the retinal and choroidal circulation. There is a delay in the circulation time of the dye from the arm to the retina. There is also a delay in the appearance of the dye in the choroidal circulation. After the dye enters the retinal arterioles, a delay in filling the retinal veins may also be noted. Multiple arteriovenous shunts are also seen in these cases.[72] Numerous microaneurysms are observed due to the dilatation of the capillaries. Capillary nonperfusion areas are noted peripheral to the arteriovenous shunts. Profuse leakage from neovascularization at the disc or elsewhere in the retina may also be observed.
Central Retinal Artery Occlusion (CRAO)
A delay in filling the retinal arterioles with no perfusion in the early phases is usually seen. The flow in the retinal arterioles may be very sluggish, and the front edge of the dye may be noted to travel slowly in consecutive FFA images. The choroidal filling is normal—a delay in choroidal filling points towards the obstruction of the ophthalmic artery or ipsilateral carotid artery. A delay in filling the retinal veins is also noted with delayed arteriovenous transit. A patent cilioretinal artery, if present, fills during the choroidal phase of FFA. Dye front reciprocation (systolic advancement and diastolic retraction of the dye front) within the cilioretinal artery may be noted in cases with CRVO and cilioretinal arterial infarction.[73] The FFA may show normal angiography features in cases with transient CRAO or reperfused cases.[74]
Branch Retinal Artery Occlusion (BRAO)
Branch retinal artery occlusions usually occur due to an embolus. A delay in filling the artery distal to the embolus is seen in the early phases of the angiography. Retinal opacification, if present, shows a corresponding area of blocked fluorescence on the FFA. The occluded artery may not show complete fill even in the late phases. Mild staining of the wall of the occluded artery may be seen in the late phases.
Hypertension
Hypertensive retinopathy is characterized by changes at the arteriovenous crossings. Compression of the vein at the area where an arteriole crosses a venule is better appreciated on FFA. Cotton wool spots, when present, show corresponding areas of blocked fluorescence. Leakage from the capillaries at the disc may be seen in cases of malignant hypertension with disc edema. Acute Elschnig spots appear as multiple spots of hyperfluorescence. These are seen in the early phase of the angiogram, with dye leakage seen in the later phases. Siegrist's streaks are areas of fibrinoid necrosis of choroidal arterioles. They are usually tongue-shaped areas that show hypofluorescence due to nonperfusion.[75] If pigmentation is present, then corresponding blocked fluorescence may be seen. Malignant hypertensive choroidopathy may reveal multiple pinpoint leaks and pooling of dye beneath the serous retinal detachment.[65]
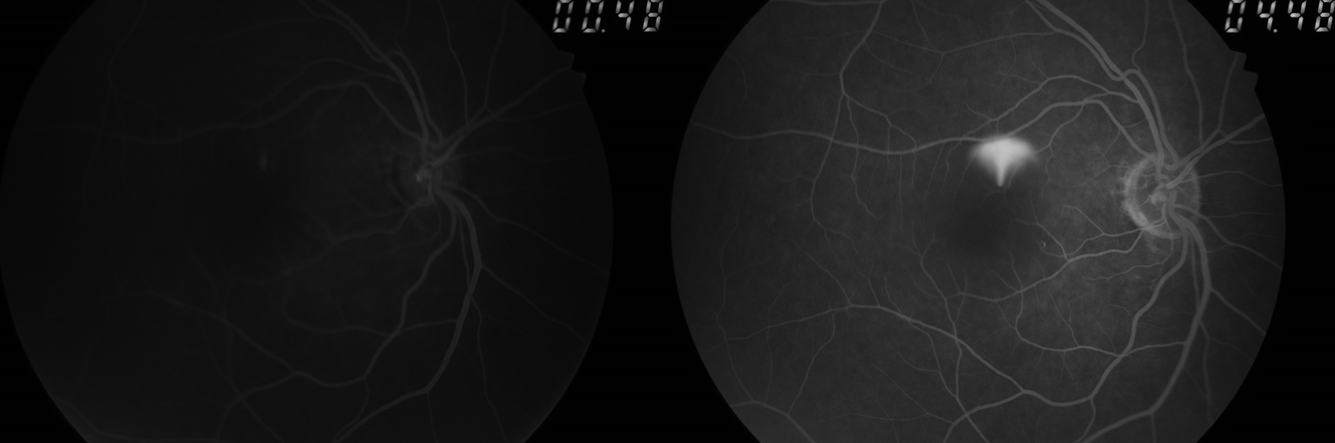
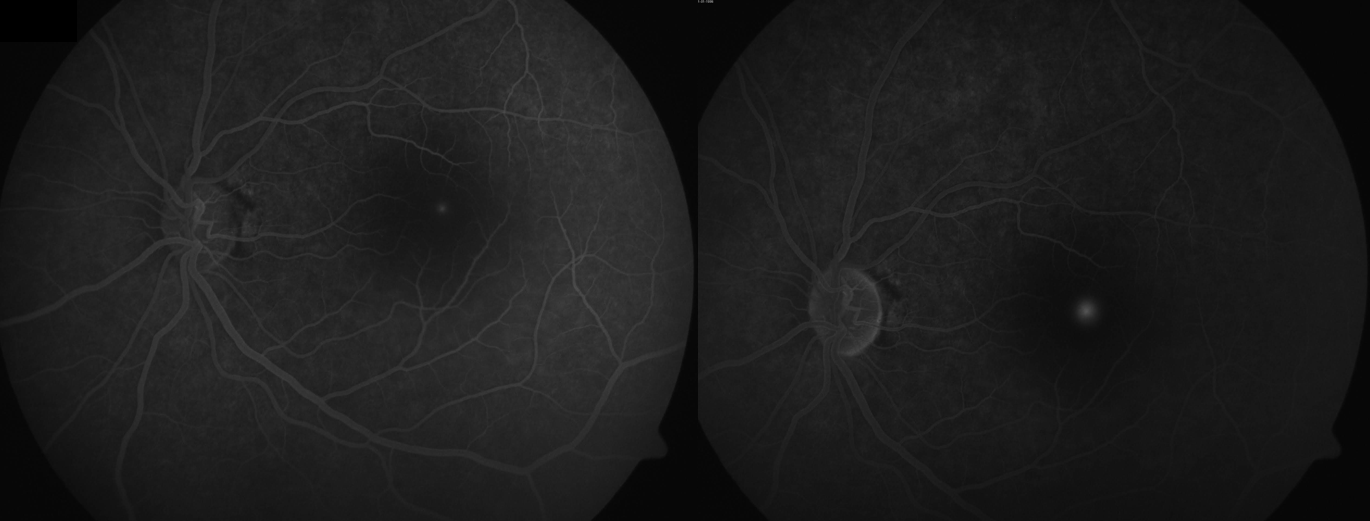
Disc Edema
Leakage of dye from the capillaries at the disc is seen in the early phases of the angiogram, followed by staining of the disc in the late phases. Cases with optic neuritis also show dilatation of the capillaries of the disc with leakage and staining of the disc in the subsequent phases. FFA can differentiate cases of papilledema (true disc edema) from pseudopapilledema. The latter does not show early or late dye leakage from the capillaries at the disc, and late hyperfluorescence of the disc is not a characteristic of pseudopapilledema.
Arteritic Anterior Ischemic Optic Neuropathy [Giant cell arteritis (GCA)] (A-AION)
A typical feature is a delay in the choroidal filling and prominence of choroidal watershed zones in the early phases. Differential filling of choroidal lobules or persistent non-filling of a few choroidal lobules may be noted.[76] The features suggest that in GCA, the flow impairment involves the short posterior ciliary arteries (SPCA) before these give branches to the optic nerve and choroid.[77]
Non-Arteritic Anterior Ischemic Optic Neuropathy (NA-AION)
Delay in the filling of the choroidal circulation is not usually seen in cases of non-arteritic ischemic optic neuropathy (NAION).[78] The characteristic feature is a difference in the filling of the dye in the capillaries at the disc. Poor filling of a segment of the disc is seen (usually superior or inferior) in the early phases of the angiogram with or without poor filling of the peripapillary choroid. Late phases show leakage at the disc, which may be diffuse (around 70%) and focal (around 30%).[79] Few unusual cases may show macular edema. In NA-AION, the distal branches of SPCA supplying the optic nerve (sparing branches to the choroid) are thought to be involved, which is more distal to the involvement seen in GCA.[77]
White Dot Syndrome
Multiple evanescent white dot syndrome (MEWDS) is identified in FFA by multiple small hyperfluorescent lesions in the early phase. These are arranged in a wreathlike configuration, and this hyperfluorescence persists in the late phase of the FFA.
FFA in cases with acute posterior multifocal placoid pigment epitheliopathy (APMPPE) reveals multiple hypofluorescent lesions in the early phase. These lesions show hyperfluorescence in the late venous phase. The early hypofluorescence results from blocked choroidal fluorescence due to infiltration of inflammatory cells or nonperfusion of the choriocapillaris.[80]
Sympathetic Ophthalmia
Two types of FFA patterns are seen in the acute stages of the disease. Multiple pinpoint hyperfluorescent leaks or multiple hypofluorescent spots in the early phase of the FFA may be seen.[81] The cases with pinpoint leaks[82] in the early phase show dye pooling in the subretinal space in the later phases. The cases with hypofluorescent spots in the early stages show staining of these spots in the late phases, similar to APMPPE.[64] The initial hypofluorescence may be due to blocked fluorescence by Dalen-Fuchs nodules or hypoperfusion of the choriocapillaris.
The chronic stages of sympathetic ophthalmia may show chorioretinal atrophy, nummular scars, subretinal fibrosis, and choroidal neovascularization. The nummular scars show transmitted fluorescence, and the subretinal fibrosis shows staining.
Uveal Effusion Syndrome
This is a less common ocular pathology where retinal detachment and ciliochoroidal detachment are noticed. These eyes are usually hypermetropic or nanophthalmic. Choroidal thickening is noticed on ultrasonography in these cases. FFA, in these cases, reveals a characteristic leopard spot appearance.[83] Multiple hypofluorescent and hyperfluorescence spots are observed in the early phase. These spots do not show any leakage in the late phases. FFA is not diagnostic in these cases. It is useful only to rule out other pathologies.
Intermediate Uveitis
Leakage from perifoveal capillaries leading to macular edema (petaloid leak) is seen in the arteriovenous phase. Staining of the disc is seen in the late phases.[84] The retinal vessels may also show leakage of dye and staining. Media may be hazy due to vitritis, which may limit the quality of captured images.
Retinitis
Retinitis appears as a whitish or yellowish lesion blocking the visibility of the choroid. On FFA, the early phase shows blocked fluorescence, and the late phase may show leakage of dye at the margin of the lesion.[24][67][85] FFA may also show vascular involvement and optic disc stain.[86]
Serpiginous Choroiditis
In its active phase, early hypofluorescence is seen due to blocked fluorescence resulting from the inflammatory process. Later phases of the angiography show staining of the margins of the lesion. Healed areas of the lesion show transmitted fluorescence without staining, and the areas of pigmentation show blocked fluorescence.[80]
Behçet disease
Fern-like fluorescein leakage has been described as a characteristic finding. It is prominent in the arteriovenous phase. It helps in the assessment of the status of inflammation. Leakage from retinal capillaries has been observed in cases of Behçet disease with no ocular symptoms or clinical signs of inflammation.[87] The presence of macular edema, disc hyperfluorescence, capillary nonperfusion areas, occlusion of retinal vessels, and neovascularization are other features observed in angiography, depending on the severity of the disease.
Posterior Scleritis
Ultrasonogram (USG) is the preferred tool for diagnosing posterior scleritis. USG reveals subtenon fluid (hypoechoic area) and a T sign. However, FFA helps rule out differential diagnoses like CSCR or choroidal hemangioma. Early hypofluorescence with pinpoint leaks in later phases may be seen on angiography in cases with posterior scleritis. Disc leakage may be seen in cases where disc edema is present. Choroidal folds may be evident on FFA.[63]
Foveoschisis
In cases with retinal dystrophies with clinically appreciable macular edema, including retinitis pigmentosa, juvenile X-linked retinoschisis, gyrate atrophy of the choroid and retina, no leakage of dye from perifoveal capillaries may be seen even in the late phases suggesting foveoschisis or maculoschisis.[88][89] This helps differentiate these cases from cystoid macular edema caused by inflammatory pathologies, which show a petaloid pattern of the leak around the foveola in the late phase of FFA. Similar non-leaking macular edema may also be seen in various conditions, including:
- Myopic foveoschisis or myopic traction maculopathy
- Vitreomacular traction
- Optic disc pit maculopathy
- Nicotinic acid maculopathy
- Enhanced S-cone syndrome/Goldman-Favre syndrome
- Cohen syndrome
- Hydroxychloroquine toxicity[90][91]
Cases of macular edema showing petaloid leak on FFA are more likely to respond to intraocular, periocular, or systemic steroid therapy. Non-leaking macular edema associated with retinal dystrophies may respond to topical or systemic carbonic anhydrase inhibitors.
Amalric Sign
This is seen in cases of choroidal ischemia. The extent of choroidal ischemia depends on the artery involved. Occlusion of the posterior ciliary artery causes extensive ischemia. These are seen as triangular-shaped lesions with their apex pointing towards the posterior pole and base towards the equator.[92] These are seen in the peripheral fundus. They show hypofluorescence in the early phases due to ischemia and staining in the late phases. Localized segmental involvement is seen in cases of occlusion of the large choroidal artery. Occlusion of smaller choroidal arteries or arterioles causes corresponding smaller ischemic lesions, either geographic in shape or small round ischemic areas depending on the area of supply of the affected vessel.
Anterior Segment Imaging
Fluorescein angiography has also been used to visualize neovascular vessels at the level of the iris, anterior chamber, or cornea.[93] FFA can also show areas of iris ischemia. In cases of neovascular glaucoma, neovascularization of the iris may not be visible clinically in the early pre-rubeotic phase. These cases can be diagnosed timely on fluorescein angiography imaging. These cases show leakage from neovascular vessels at the pupillary margin on imaging of the iris after intravenous fluorescein angiography injection. The aqueous humor may appear greenish on slit-lamp examination in various conditions, including in patients with new vessels in the iris or angle of the anterior chamber and active anterior uveitis.
Imaging of the cornea has also been documented. This is useful for documentation of corneal epithelial defects and their response to treatment at follow-up. In cases with corneal epithelial defects, topical fluorescein is used in the concerned eye, and images are taken with a fundus camera in fluorescein angiography mode with the exciting and barrier filters. The area of epithelial defect is captured as a hyperfluorescent area.[94]
Ultra-wide field retinal imaging
The standard fundus camera captures images of 30-50 degrees of the retina. Several wide-field imaging systems are now available. With the help of wide-field imaging, peripheral vascular leakage, peripheral vascular loops, and capillary nonperfusion areas can be easily assessed.[89][95]
Ultrawide field FFA can vividly show the peripheral avascular area in retinopathy of prematurity (ROP) and familial exudative vitreoretinopathy (FEVR). Both of these diseases can cause temporal dragging of the fovea and straightening of the peripheral retinal vessels. Peripheral avascular areas have also been noted in rhegmatogenous retinal detachments (RRD), possibly proving an explanation of retinal new vessels or iris new vessels seen in chronic RRDs.[96] Ultrawide field FFA using a scanning laser ophthalmoscope and ellipsoid mirror can image the retina through small pupils and hazy media in various conditions, including uveitis and diabetes mellitus.[97]
Ultrawide field FFA showed peripheral retinal vascular involvement and evidence of posterior segment inflammation in a majority of patients with uveitis related to juvenile idiopathic arthritis in a study by Tripathy and colleagues.[98] Targeted retinal photocoagulation to the areas of capillary nonperfusion on ultrawide field FFA has been used successfully in various disorders, including BRVO with macular edema, proliferative diabetic retinopathy, and diabetic macular edema.[99][100][101]