Continuing Education Activity
Mucous membrane graft acts as a basement membrane and is indicated in many cases like Steven Johnson syndrome, chemical injury, socket reconstruction, and Ocular Cicatricial Pemphigoid. The graft can be harvested from labial or buccal mucosa. Though being safe and simple, the procedure can have untoward complications like graft failure, graft shrinkage, dislodgement of graft, and donor site morbidity. The article, in brief, describes the indication, technique, and complications of the procedure.
Objectives:
- Review the basic anatomy of labial mucosa, which is the chief donor site of the graft.
- Describe the indications of the mucous membrane graft.
- Explain the procedure of performing mucous membrane graft.
- Outline the role of the interprofessional team in the management of patients undergoing mucous membrane grafts.
Introduction
It is important to understand the anatomy of the conjunctiva when considering the reconstruction and replacement of the conjunctiva.[1] The human conjunctiva is an epithelial layer that is a non-keratinized stratified squamous and stratified columnar epithelium. The conjunctiva has goblet cells. The conjunctiva is a complex structure that contains lymphatic channels, blood vessels, fibrous tissue, melanocytes, T- and B-cell lymphocytes, accessory lacrimal glands, and Langerhans cells. Glands of Krause are found within the deep fibrous layer of the conjunctiva and the goblet cells, which are greatest in the fornices, secrete mucin. The conjunctiva contributes to the protection of the eye with the production of mucus and tears. It provides mechanical protection to the eye and assists in the free movement of the globe and the eyelids. The conjunctiva lines the tarsus (or palpebral conjunctiva), where it lines the eyelid margin of the tarsus and the Tenon's capsule. The flexibility of the forniceal conjunctiva is important in the free movement of the globe and the eyelids.
Loss or scarring of the conjunctiva from injury or disease can result in eyelid malposition, restriction of ocular movement with double vision, and dryness. When replacing the injured conjunctiva, materials such as amniotic membrane, hard palate grafts, nasal septal mucosa, autogenous conjunctiva, and tarsus with conjunctiva have been used. Ideally, it is wise to replace like for like. Unfortunately, except for small defects, autogenous conjunctiva is limited by availability.
Oral mucosal grafts are used in two forms:
- Oral mucous membrane grafting (MMG)
- Minor salivary gland grafting (MSGG) for the treatment of severe dryness
Oral mucosa has multiple advantages:
- It has similar biological properties to the conjunctiva
- The donor site is easily accessible and universally available
- Obtaining donor tissue is technically easy
- Obtaining tissue is cheap
- Repeat harvesting is possible
- Complications are few
- It is an autograft avoiding the risks of allogeneic grafts.
In 1912, Denig first described the use of mucous membrane for lime burns; later, mucous membrane grafts were used by Weeks for the correction of trichiasis and symblepharon.[2] The mucous membrane acts as a scaffold for the proliferation of growing epithelial cells. The membrane does not contain the goblet cells which are present in the conjunctiva. Hence it does not help in treating dry eyes unless transplanted with minor salivary glands.[3] Moreover, in patients with concurrent limbal stem cell deficiency of more than 3-4 clock hours, a limbal stem cell transplant procedure should be performed in the same sitting. The oral mucosal membrane is rich in elastin, making it resistant to shearing and compression, and being highly vascular, and the graft can be easily taken up. It is histo-compatible and has minimal contraction in the transplanted site.[4]
Indications for the use of mucous membrane grafts in Ophthalmology include the following:
- After removal of pterygia
- Repair of contracted anophthalmic sockets
- Globe surface and fornix reconstruction after tumor resection
- Deformities of the eyelids (cicatricial entropion, keratinization of the eyelid margin after chemical burns, or Stevens-Johnson syndrome)
- Repair of erosions over glaucoma drainage devices
- Repair over scleral buckle exposure
- Support of keratoprosthesis associated with corneal melt
- Formation of the lining during conjunctivodacryocystorhinostomy or dacryocystorhinostomy
Anatomy and Physiology
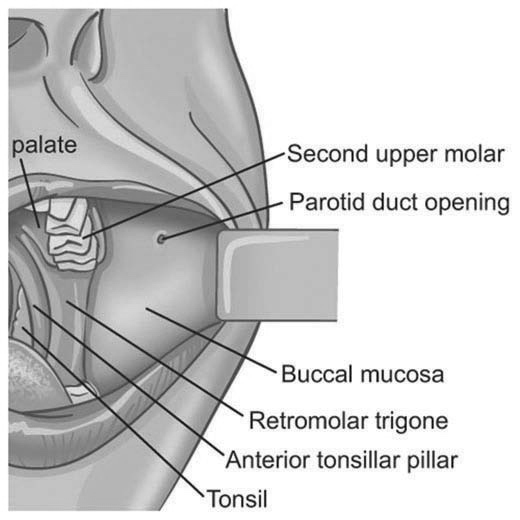
Harvesting sites of Mucous Membrane Grafts[4]
- buccal mucosa of the inner cheek
- mandibular labial mucosa of the lower lip
Boundaries of the Lower Labial Mucosa
- Laterally and anteriorly by the lower lip commissure
- Posteriorly by the anterior tonsillar pillar
- Superiorly by the maxillary vestibular fold
- Inferiorly by the mandibular vestibular fold
Blood Supply
Branches of the maxillary artery, including the buccal artery, the middle and posterior superior alveolar arteries, and the anterior superior alveolar branch of the infraorbital artery, supply the mucosa. Additional blood supply is from the transverse facial artery, which is a branch of the superficial temporal artery.
Innervation
The buccal mucosa is innervated by the long buccal nerve, a branch of the third division of the trigeminal nerve (CNV3), and the anterior, middle, and posterior superior alveolar branches of the second division of the trigeminal nerve (CNV2). The facial nerve also contributes sensation to the buccal mucosa.[4]
Histologically the mucosal epithelium is lined by the stratified non-keratinized squamous epithelium without any goblet cells. Below the mucosa is the vascular layer of connective tissue, which is termed lamina proparia. The loose connective tissue of the lamina propria inserts into the overlying epithelial layer through finger-like projections or papillae.
The inferior labial mucosa is supplied by the inferior labial branch of the facial artery and the buccal branch of the maxillary artery.
Minor Salivary Glands
Labial minor salivary glands are more numerous than at other sites. They are exocrine glands with a less developed ductal system than seen with major salivary glands. Minor salivary glands secrete mucinous or seromucinous secretions. Secretion is stimulated by the lingual nerve (postganglionic parasympathetic innervation).
Properties of Oral Mucosa
Buccal and labial mucosa have non-keratinized stratified squamous avascular epithelium with vascular connective tissue called lamina propria. The oral mucosa has a thick elastin-rich epithelium which is resistant to shearing or stretching forces. There are connective tissue projections from the lamina propria into the epithelium, which further adds to the resilience of the mucosa. The lamina propria has extensive nerve fibers and blood vessels from the submucosa. It is the highly vascular lamina propria that allows excellent take of mucosal grafts. The lack of hair and minimal contracture, when placed in a recipient bed further, makes mucous membrane grafts ideal for mucosal cover. The inherent biological properties of oral mucosa make it resistant to infections and inflammation. Antimicrobial peptides, cytokines, and defensins secreted by the mucosal epithelium together with mucosa-associated lymphoid tissue limit micro-bacterial colonization of the mucosa. Additional resistance to microbial invasion is provided by the rich lymphocyte, neutrophil, macrophage, mast cell, and plasma cell populations in the lamina propria. The mucosal epithelium has also been shown to have epithelial stem cells which promote rapid epithelialization when injured. The density of mucosal salivary glands is higher in the lateral aspect of the labial mucosa, which should allow the proper choice of donor mucosa when treating dry eyes.
Comparing Physiology of Saliva to Tears
Mucin is responsible for coating the ocular epithelium reducing the surface tension of the eye, and forms a stable and durable wet layer. Evaporation is reduced by mucins which form a hydrophobic barrier and increase the tear film break-up time. Saliva also contains immunoglobulin A, lysozyme, lactoferrin, and human beta-defensin, which have antimicrobial properties and protect the ocular surface from infection. Mucosal salivary gland secretions also contain epidermal growth factor and transforming growth factor-B, promoting normal growth of ocular surface epithelium and corneal reepithelization. Salivary secretions have lower osmolality when compared to normal tears and can lead to corneal epithelial microcystic edema in some patients with transplanted mucosa. Mucosal salivary glands produce secretions continuously, even during sleep which is useful in maintaining ocular surface lubrication.
Indications
Ocular Surface Reconstruction
A symblepharon is an abnormal adhesion between the palpebral and bulbar conjunctiva, potentially resulting from a large number of etiologies like chemical injury, ocular cicatricial pemphigoid, burns, recurrent pterygium, and Steven Jhonson syndrome. A bare conjunctival surface predisposes to the development of symblepharon. Symblephron can limit extra-ocular movements and can also be sight-threatening. Amniotic membrane graft can be used in patients with mild symblepharon. The mucous membrane graft(MMG) prevents the recurrence of symblepharon by providing epithelial basement membrane support for the growing cells.[5][6]
Contracted Socket
A healthy socket with well-formed fornices is essential for a well-fitting prosthesis. A contracted socket can result after enucleation or evisceration due to excessive tissue loss during the surgery, inflammation, and radiation. MMG acts as a physiological substitute for the reconstruction of the contracted socket. MMG, along with fornix formation sutures, is indicated in patients with a moderately contracted socket with surface area loss. A large number of studies have shown good results with MMG in patients with anophthalmia.[7][8] In severely contracted sockets, Putterman and Karesh successfully used a large mucous membrane graft over a custom-designed C-shaped conformer, achieving stable fornices in 40 of 41 severely contracted sockets.[9] Mucous membrane grafts may be used in conjunction with hard palate grafts to provide rigidity to the eyelid and avoid lower eyelid laxity. In mild orbital volume loss with mild socket contracture, it is possible to use a composite graft of the oral mucosa with the attached underlying fat although this only gives a small improvement in volume.[10]
Post-enucleation Socket Syndrome
The post-enucleation socket is not a contracted socket per se. It is a sunken, depressed, and posteriorly rotated socket, as described by Ton Smit and Leo Koornneef.[11] When repairing these sockets with orbital floor wedged implants, there can be shortening of the inferior fornix: this is simultaneously repairable with a mucous membrane graft.[7] Similarly, in patients with very dry anophthalmic cavities, Franca et al. showed a significant improvement when the sockets were transplanted with mucosa bearing minor salivary glands. Patients had an increase in prosthetic wear comfort and were able to retain their prostheses.
Orbital Implant Exposure After Enucleation or Evisceration
Full-thickness buccal mucous membrane grafts have been used together with the underlying submucosal fat to repair hydroxyapatite orbital implant exposure.[12]
Lid Margin Abnormalities
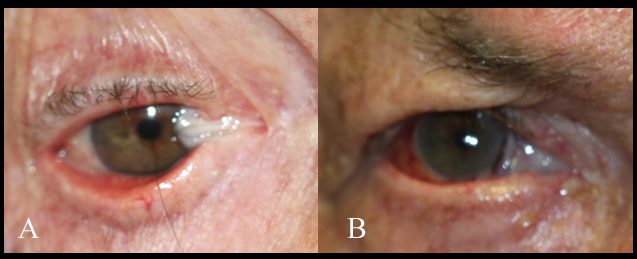
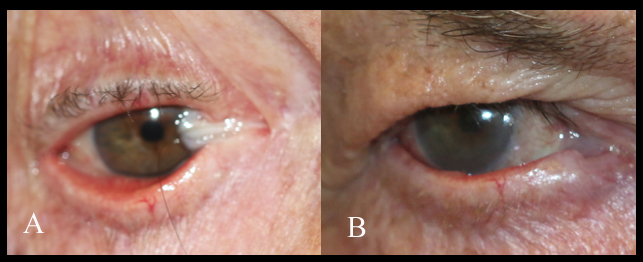
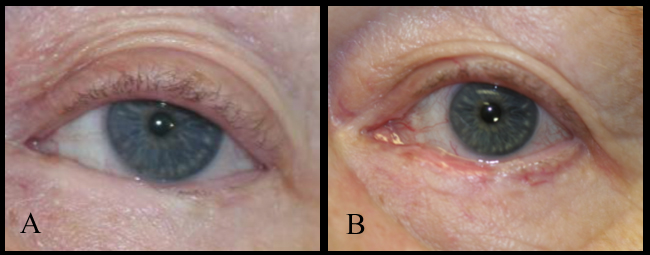
Steven Johnson syndrome, ocular cicatricial pemphigoid, toxic epidermal necrolysis, trachoma, congenital ichthyosis, and chemical burns cause shortening of the fornices and posterior lamella. This leads to cicatricial entropion, trichiasis, and keratinization of the lid margin, which can further result in corneal epithelium damage and persistent epithelial defects due to the repeated blinking and eventual blindness. Reconstruction aims to correct the cicatricial entropion, correct the eyelid margin keratinization, and trichiasis, together with any lagophthalmos. Eyelid margin keratinization in patients with cicatricial entropion was first treated with removing the lid margin keratin and grafting with MMGs by McCord and Chen.[13] Most such cases are now treated with anterior lamellar recession together with eyelid margin mucous membrane grafting. The endpoint with these procedures is to prevent keratinized epithelium and cilia from touching the ocular surface. The procedure of mucous membrane grafting may be combined with mechanical removal of keratin, partial excision of the eyelid margin and tarsus or tarsal conjunctiva, cryotherapy, and anterior lamellar recession. In the presence of significant symblepharon, local treatment is needed separately from the eyelid margin treatment. In cases of ocular cicatricial pemphigoid, it is important to have the underlying disease medically controlled. The reconstruction of posterior lamella can be performed with mucous membrane graft, and tarsus can be replaced with a hard palate. Labial mucosa has also been used as a spacer in eyelid retraction due to Steven Johnson syndrome.[14]
Trachoma, caused by Chlamydia trachomatis, is the third leading cause of blindness worldwide (after cataract and glaucoma) and the commonest cause of blindness by infection. The chronic immune response elicited by the infection causes severe scarring, most commonly of the upper tarsal plate. The resulting entropion and trichiasis cause corneal scarring and eventual blindness. Surgical repair of the cicatricial entropion can prevent blindness. Surgical intervention may include tarsal rotation, tarsal repositioning, posterior lamellar lengthening with an interpositional mucous membrane or tarsal graft, anterior lamellar repositioning, eyelid margin split, eversion, and use of an interpositional mucous membrane graft.[15][16]
Distichiasis can also be treated with resection of the portion of the tarsus containing the abnormal row of lashes and replacing it with a mucous membrane graft. However, in doing so, there is a risk of injuring the openings of the meibomian glands as the abnormal row of lashes is posterior to the normal row of lashes and in close proximity to the meibomian gland orifices.
With eyelid margin repairs, the mucous membrane graft replaces keratinized epithelium, mechanically prevents the contact of keratinized surfaces and trichiasis to the ocular surface, or lengthen contracted tissues.
Eyelid Reconstruction
Buccal mucosa was used to reconstruct the lower eyelid by van der Meulen in 1982.[17] Subsequently, mucous membrane grafts for the posterior lamella reconstruction have been used with rotational myocutaneous flaps. A composite skin-muscle-mucosa graft from the lower lip has also been used to repair an eyelid margin defect in the elderly.[18] Mucous membrane grafts have also been used to repair the cicatricial ectropion seen in congenital ichthyosis because the oral mucosa is not involved in this disease process. The transplanted mucosa undergoes metaplasia into keratinized skin, with less shrinkage than with traditional skin grafts.[19] Patients with cryptophthalmos have also been repaired using mucous membrane grafts.[20]
Ocular Surface Reconstruction
Severe symblepharon necessitates excision of scar tissue, creating raw surfaces on the tarsus and loss of conjunctiva in the fornices and bulbar surfaces, often with pannus formation because of loss of limbal stem cells. In these advanced cases, the amniotic membrane may be used to resurface the bulbar conjunctiva, but mucous membrane grafts are needed to reconstruct the fornices and coverage over the tarsus. Mitomycin C may be used intraoperatively to reduce the formation of secondary scarring. Kheirkhah et obtained deep fornices without motility restrictions in 84% of their 32 cases.[21]
Ocular resurfacing with mucous membrane grafts is often needed when implanting osteo-odonto-keratoprosthesis.[22] The conjunctiva is often diseased in advanced cases such as Stevens-Johnson syndrome or chemical burns, and the use of mucous membrane grafts allows coverage of the sclera, cornea and provides a more hospitable environment for the keratoprosthesis. Corneal stromal melts may be repaired with corneal grafts, which are covered by buccal mucous membrane grafts.
In the presence of total limbal stem cell deficiency where allogeneic limbal stem cells are not available or have failed, the use of mucous membrane grafts to the limbal region has been shown to improve corneal clarity. It may allow re-epithelialization of the cornea when combined with a vascular pedicle in the form of a tenonplasty. Similar repair of intractable sclerocorneal melts has been performed with mucous membrane grafts.[23][24]
Pterygium
Although early studies used mucosal grafts for primary pterygium resection and repair, these are now reserved for advanced recurrences with insufficient donor conjunctiva. With the use of glued conjunctival autografts, the recurrence rate of pterygia has been reduced dramatically. In cases of severe recurrence with motility problems and a lack of sufficient donor conjunctiva, split-thickness buccal grafts give a reasonable cosmetic outcome. [25]
Mucous Membrane Grafts as a Corneal Cover
When an eye does not have a useful vision but requires a cosmetic scleral shell to be fitted, a Gundersen flap is traditionally used. This reduces the sensitivity of the cornea, thereby allowing cosmetic scleral she'll wear. Mucous membrane graft has also been used as an alternative to Gunderson's flap in patients with phthisis bulbi and micro-ophthalmos with the sensitive cornea, thus aiding in the fitting of a scleral shell.[26] However, with a Gundersen flap, there may be a compromise of the fornix. The use of a mucous membrane graft allows preservation of the fornices and avoids the risk of flap retraction.[27][26]
Glaucoma Surgery
Glaucoma aqueous drainage devices may suffer from erosion of the overlying conjunctiva leading to exposure of the tube or plate. Free autologous conjunctival flaps and grafts are always the initial approach but may fail if there is surrounding scar tissue with secondary contracture. The amniotic membrane only acts as a substrate for healing and is usually inadequate for actual coverage of the device. Mucous membrane grafts involve minimal cost, are easily obtained, and achieve easy vascularization while avoiding local contracture in the presence of conjunctival scarring. Lamellar corneal patch grafts can be covered by a buccal mucous membrane which is secured to the recipient conjunctiva.[28] Similarly, persistently leaking trabeculectomy blebs can be repaired with buccal mucous membrane grafts where conjunctival flaps have failed.[29]
Retina Surgery
Atrophy of tissues overlying scleral explants placed for retinal detachment surgery may cause wound dehiscence and scleral exposure. Because of the risk of increased retinal detachment with the removal of the external explant, attempts may be made to retain the explant with coverage using a banked scleral patch graft and an overlying mucous membrane graft as a conjunctival substitute.[30]
Lacrimal Drainage Surgery
Conjunctivodacryocystorhinostomy (CDCR) is the creation of a passage from the conjunctiva to the nasal mucosa and the placement of a Jones tube, but without an epithelial lining. Therefore, if the Jones tube falls out or is buried, the passage fails to function. Several surgeons have used mucous membrane grafts to ensure there is a mucosal lining when performing CDCR: this negates the possibility of closure if the tube migrates or falls out.[31][32][33] In patients undergoing dacryocystorhinostomy where there is scarring with mucosal shortage, mucous membrane grafts have been shown to maintain patency of the anastomosis.[34]
Severe Dry Eyes
Murube del-Castillo in 1998 was the first to describe transplantation of minor salivary glands with the mucosa to the eyelids in patients with severe aqueous tear deficiency where all other treatments had failed.[3] He observed an improvement in the mean Schirmer test from 507 mm to 11.7 mm and reduced dry eye symptoms. This was an important advancement in the management of severe drys. This technique may be performed together with limbal stem cell and corneal transplantation in patients with the Stevens-Johnson syndrome or secondary to chemical burns. Following this initial report, multiple studies have shown significant improvement in the Schirmer test measurement, subjective comfort, and vision after such minor salivary gland mucosa transplantation. This has been shown in patients with Sjogren's syndrome, chemical burns, and Steven Johnson syndrome with transplantation into the inferior conjunctival fornix.[35][36][3][37] Significant clinical improvement in comfort has been noted in a many as 92% of cases and 86% noting an improvement in vision after receiving fornix mucosal grafts with minor salivary glands.[37]
Complications tend to be minor with partial ptosis, eyelid thickening, and increased mucus discharge. Repeat labial grafting was successful in patients with Steven-Johnson syndrome and pemphigoid disease during follow-up. Labial minor salivary gland transplantation into the conjunctival fornix in patients with a preoperative Schirmer test of zero was found to increase Schirmer test reading by 74% and improve corneal transparency, corneal neovascularization, and visual acuity. In the foreign body sensation, photophobia and pain were noted in more than 50% of patients. Murube's contribution to the management of severely dry eyes is a major one that is as yet not utilized widely.
Contraindications
Avoid in Patients With
- Severe kerato-conjunctivitis sicca
- Advanced ocular cicatricial pemphigoid
- Active conjunctival inflammation that is uncontrolled by immunosuppressants.[38]
Equipment
For the Preparation of the Recipient Bed
- Westcott conjunctival scissors
- Serrated, non-toothed forceps
- Blotting paper
For Fornix Formation
- 4-0 silk
- Castervejo forceps
- For harvesting of the mucosal graft
- Westcott conjunctival scissors
Personnel
The labial mucosal site is an easily approachable site, and a trained oculoplastic surgeon can harvest grafts from the labial mucosa; however, in patients needing buccal mucosal grafts, one can seek the help of a dental surgeon.
Preparation
The patient in the pre-operative period is advised to use chlorhexidine/ betadine mouth wash at least for one week.
Technique or Treatment
Mucosal grafts can be harvested from the oral cavity, nose, vagina, or rectum. The oral mucosal grafts are most commonly performed due to the simple harvesting technique and easy accessibility. The oral mucosal grafts can be harvested from either the labial mucosa, the buccal mucosa, or the hard palate.
Full-thickness Mucous Membrane Grafts from the Buccal Mucosa
Mucosal harvesting may be performed under local anesthesia, sedation anesthesia, or general anesthesia with the tube secured to one side of the mouth. Exposure of the mucosa may be achieved using several different instruments, including Sewell retractors, Babcock clamps, Dingman retractors, or a Steinhauser mucosal stretcher. The use of one of these instruments allows the mucosa to be kept under tension which aids dissection. Additional pressure on the outside of the cheek or lower lip by an assistant further helps keep the mucosa taut.
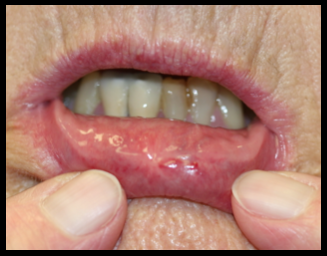
Full-thickness mucous membrane grafts have less post-operative contracture and are commonly performed. The scar tissue is dissected, following which fornix-forming sutures are passed with the help of 4-0 silk sutures. The size of the defect is then measured with the help of blotting paper to get an approximate size of the graft. The donor site is cleaned with povidone-iodine, the size of the graft is marked with the help of the blotting paper. The donor site is infiltrated with bupivacaine 0.25% mixed with 1:200000 epinephrine which facilitates hemostasis and allows hydro-dissection of the mucosa. The incision is made along the marking with the help of a 15 Bard-Parker blade, and the dissection is performed with the help of conjunctival scissors.
When harvesting the mucous membrane graft, care should be taken to dissect it off the submucosal fat and minor salivary glands, which line the inner surface of the buccinator muscle. The buccinator muscle is a muscle of facial expression, and trauma to the muscle may lead to contracture of the wound with restriction of the opening of the mouth. Aggressive posterior dissection can injure the buccal nerve, which can lead to perioral numbness. Care is also necessary to avoid damage to the frenulum and the vermilion lid margin. The surgeon should be aware of the structures that lie within the buccal space lateral to the buccinator muscle: the buccal fat pad, Stensen's duct of the parotid gland, the facial artery and vein, the buccal artery posteriorly, lymphatic vessels, and buccal branches of the facial and trigeminal nerve. Stensen's duct is lateral to the masseter muscle. It continues forward and turns medially at the anterior masseter muscle border, and passes through the buccal fat pad to penetrate the buccinator muscle with its orifice on the mucosa opposite the upper second molar tooth. Trauma to Stensen's duct can result in a decrease in parotid salivary flow. Squeezing the parotid gland allows one to visually identify the opening of the duct through the mucosa. It is best to harvest the graft staying 1 cm inferior to the Stensen duct papilla and 1. 5 cm away from the lip vermillion, posterior to the labial commissure, and anterior to the pterygomandibular raphe. Skin grafts generally contract by 25% (although the degree of contracture may be more if the bed is on severely damaged skin), and mucosal grafts shrink by about 20%.[39] Graft shrinkage is usually seen within six months of surgery. Split-thickness mucous membrane grafts are liable to shrink more than full-thickness grafts. The mucosal grafts can be placed under tension with a symblepharon ring, a PMMA conformed, or a silicone conformer.
The graft should be harvested as thinly as possible. The donor site is then cauterized minimally to prevent thermal nerve injury. In the postoperative period, the patient is advised to use betadine gargles along with systemic antibiotics; the patient can also be asked to opt for a liquid diet until the wound heals. After harvesting, the graft can be placed on the index finger to carefully dissect the sub-mucosal fat from the graft with the help of Westcott or Stevens scissors. The graft is placed on the bed and can be either sutured with a 6-0 polyglactin suture or fixed with the help of fibrin glue.[40] When grafts are taken with mucosal salivary glands, it is best to harvest the mucosa from the lower lip sulcus, 1 cm away from the mucocutaneous junction and 2 - 3 cm in length. The graft is dissected off the quadratus labii muscle.
Labial Mucosal grafts
The mandibular labial alveolar mucosa has the following borders:
- The vermilion border of the lower lip superiorly
- Vestibular fold between the lower lip and anterior border of the mandible inferiorly
- Outer commissures of the lower lip laterally.
The mental and buccal branches of the maxillary artery and the inferior labial branch from the facial artery (all from the external carotid artery) supply this mucosa.
The mental nerve, a terminal branch of the inferior alveolar nerve arising from the mandibular branch of the trigeminal nerve, supplies sensation. The mental nerve traverses the mental foramen, which is located between the first and second premolar teeth. Injury to the mental nerve is avoided by placing the initial incision medial to the middle of the canines. Lip contracture and inversion can occur from scarring if the incision is closer than 1.5 cm from the lower lip vermillion. Injury to the orbicularis oris muscle should be avoided as mobility of the lips may be impaired with an abnormal smile. Finally, mucosa near the teeth should be protected to prevent periodontal defects.
Mucosal Grafts with Minor Salivary Glands
Minor salivary glands are 1 to 5 mm in size. They are found in the labial mucosa, the buccal mucosa, the base of the tongue, and the posterior hard palate. In the lips, these are found as a continuous tightly packed layer of lobules between the quadratus labii and the labial mucosa.[3]
Split Thickness Mucous Membrane Graft
Split thickness mucosal grafts are less commonly performed. They are indicated when the graft has to be placed in the exposed areas, like after pterygium surgery, for better cosmesis. A microtome is used to harvest a mucosal strip of approximately 0.3mm thickness mucosa.
Suture Versus No Suture of the Donor Site
The donor site, as mentioned above, can either be sutured or can be allowed to heal by secondary intention. Suturing causes discomfort and may cause shortening and scaring of wound edges leading to poor cosmesis. The mucosal wound, when left open, has a tremendous self-healing property. The oral mucosa can undergo rapid healing due to the presence of a large number of growth factors in the saliva. Moreover, the presence of lymphoid tissue, macrophages, and plasma cells acts as a barrier for microbial invasion.
In the postoperative period, the patient is advised to avoid hot and spicy food, and it is advisable to apply lignocaine gel before meals. Chlorhexidine mouth wash and betadine gargles after meals for one week should also be added.
Complications
The complications include graft displacement, shrinkage, necrosis, wound site infection, granuloma formation, and lid thickening in the long term. Donor site complications include infection, mucosal necrosis, non-healing of the site, lip margin scarring, hematoma formation, persistent pain, and perioral numbness due to inadvertent damage to the mental nerve. Oral tightness may occur with wound contracture leading to limitation of jaw opening. Bleeding and buccal hematoma may require surgical intervention. Parotid duct injury can lead to abnormal salivary gland function.
Contracture after buccal mucosal grafts leading to difficulty opening the mouth is variable and reported to occur from 0% to 32% of patients.[41] Surgical release with Z-plasties may be necessary.
Lower lip paresthesia may persist for many months after surgery but generally resolves unless the mental nerve has been injured. Cauterization in this area can cause nerve injury as well.
Although lip mucosa is best for harvesting mucosal salivary glands, the risk of lip contracture and lip inversion of the vermilion is significant if the donor site is not carefully chosen.
Transient decrease in salivary flow may occur because of trauma to Stenson's duct or due to edema following surgery.
Alcohol abuse and smoking may lead to dysplastic changes in the oral mucosa with poor donor and recipient site outcomes. Medications such as clindamycin, angiotensin-converting enzyme inhibitors, and nonsteroidal anti-inflammatory drugs may alter mucosal anatomy.
Full-thickness mucous membrane grafts will remain pink and can be a cosmetic concern when visible. Split-thickness mucous membrane grafts are less pink but liable to shrink more and should not be used for fornix reconstruction. They are more suitable for bulbar conjunctival replacement as done after extensive recurrent pterygium surgery.
Failure of mucosal grafts with necrosis may be seen when used with osteo-odonto keratoprosthesis with necrosis rates of 8% to 50% reported.[41] Excessive mucosal salivary gland secretions may also occur and necessitate graft excision, cryotherapy, or injection with botulinum toxin.
Nodules (Fordyce nodules) may develop on the mucosa when transplanted to the ocular surface and necessitate surgical excision.
Disturbance of the conjunctiva in patients with ocular pemphigoid disease when applying mucous membrane grafts can activate local disease. It is important to only perform reconstructive surgery in these patients after the use of systemic immunosuppression.
Overall, mucosal grafts from the inner cheek give less postoperative discomfort, fewer neuro-sensory deficits, and fewer patients have salivary flow changes when compared to lower lip mucosal grafts.
Clinical Significance
Mucous membrane graft is a simple and viable option for the ocular surface reconstruction in patients with severe cicatrizing disorders like thermal injury, Steven Johnson's syndrome, and ocular cicatricial pemphigoid. They have also been used for the reconstruction of the severely contracted socket. The technique for harvesting labial mucosal graft is simple, easily accessible, and can be easily performed by an ophthalmologist. Moreover, the site is easily accessible, and there is no scarcity of donor grafts. Oral mucosa is thick and remains stable, thus minimizing the recurrences.
Enhancing Healthcare Team Outcomes
Oral mucosal grafts are a physiological substitute for reconstructing the posterior lamella in patients with posterior lamella shortening. In patients with Steven Johnson's syndrome, ocular cicatricial pemphigoid, and chemical injury with severe posterior lamella shortening and symblepharon formation, the oral mucosal grafts have been shown to provide good results in the various studies.[42][43][44]
In a study done by Iyer et al., in 54 eyes of 31 patients, with Steven-Johnson syndrome (SJS) and lid margin keratinization, mucous membrane graft with keratinized lid margin excision was performed. 92.6% of patients had a stable ocular surface, reduced conjunctival hyperemia, and improved visual acuity over 6 months of follow-up.[40] Osaki et al. did a retrospective review of 44 patients with cicatricial entropion who underwent mucous membrane graft and found improvement in the ocular symptoms in 98% of the patients, with no graft-related complication.[45] Similar observations were made by Singh et al. in 6 patients with cicatricial entropion.[46]
Lee et al. used mucous membrane graft and hard palate mucosal grafts to reconstruct the contracted socket in 13 patients. They found that all the patients after surgery were able to wear the artificial eye, though 5 of their patients required repeat surgeries.[8]
Besides ocular surface reconstruction, mucous membrane grafts have also been used as a conjunctival substitute in patients with recalcitrant leaking trabeculectomy bleb.[29]
An interprofessional team approach (including specialists, clinicians, surgical nurses, and surgical assistants) to performing mucous membrane grafts will yield the best patient results. [Level 5]