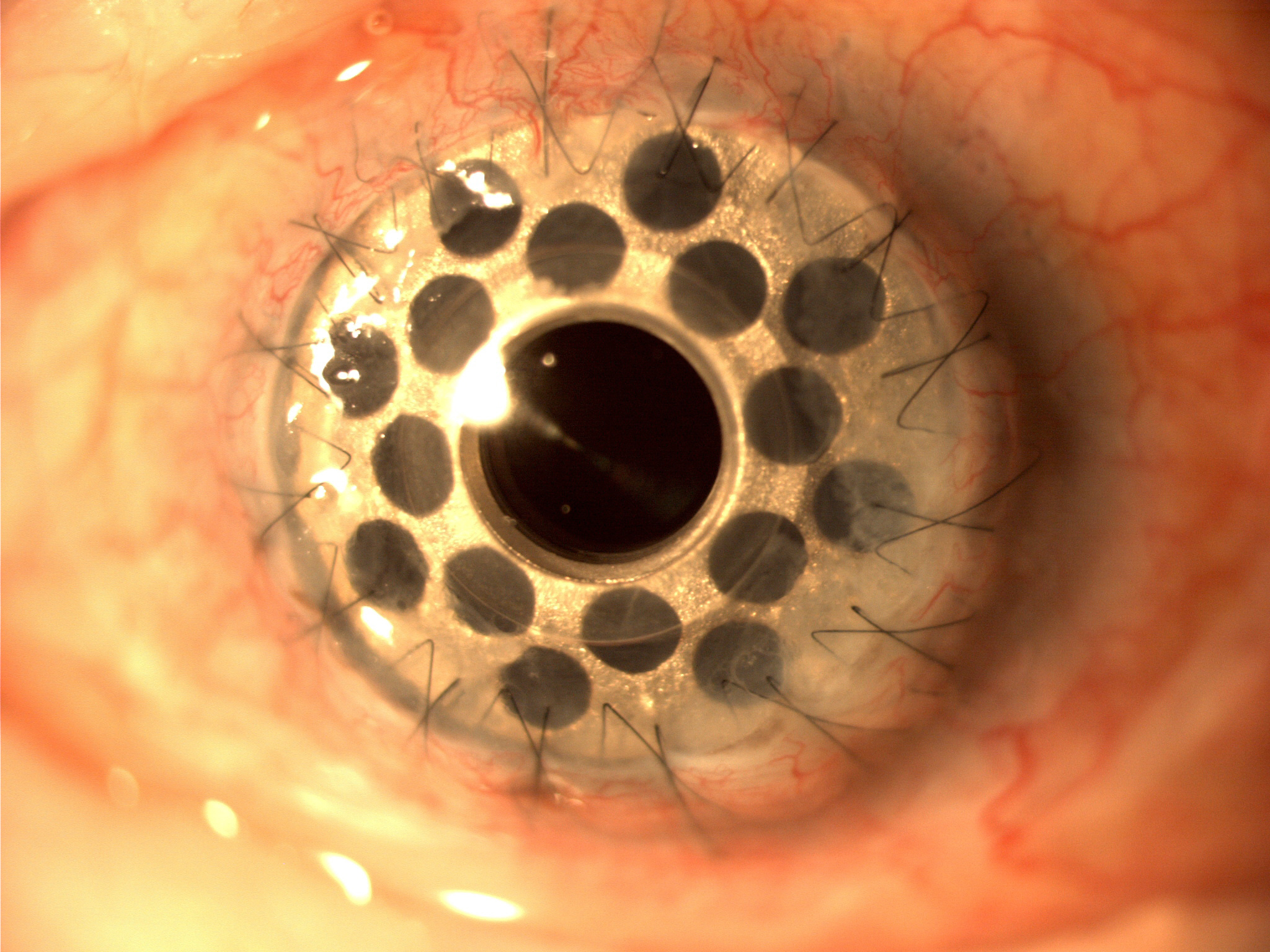
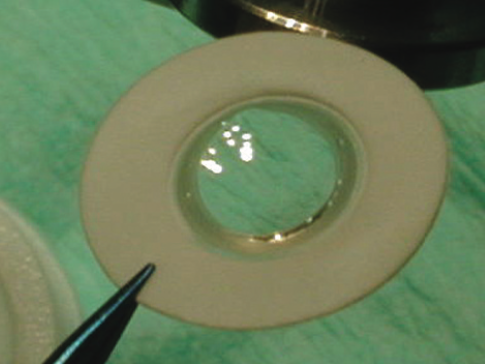
[1]
Pascolini D, Mariotti SP. Global estimates of visual impairment: 2010. The British journal of ophthalmology. 2012 May:96(5):614-8. doi: 10.1136/bjophthalmol-2011-300539. Epub 2011 Dec 1
[PubMed PMID: 22133988]
[2]
Mathews PM, Lindsley K, Aldave AJ, Akpek EK. Etiology of Global Corneal Blindness and Current Practices of Corneal Transplantation: A Focused Review. Cornea. 2018 Sep:37(9):1198-1203. doi: 10.1097/ICO.0000000000001666. Epub
[PubMed PMID: 29912039]
[3]
Avadhanam VS, Smith HE, Liu C. Keratoprostheses for corneal blindness: a review of contemporary devices. Clinical ophthalmology (Auckland, N.Z.). 2015:9():697-720. doi: 10.2147/OPTH.S27083. Epub 2015 Apr 16
[PubMed PMID: 25945031]
[4]
Mitry D, Bhogal M, Patel AK, Lee BS, Chai SM, Price MO, Price FW Jr, Jun AS, Aldave AJ, Mehta JS, Busin M, Allan BD. Descemet stripping automated endothelial keratoplasty after failed penetrating keratoplasty: survival, rejection risk, and visual outcome. JAMA ophthalmology. 2014 Jun:132(6):742-9. doi: 10.1001/jamaophthalmol.2014.352. Epub
[PubMed PMID: 24763830]
[5]
Giles CL, Henderson JW. Keratoprosthesis: current status. The American journal of the medical sciences. 1967 Feb:253(2):239-42
[PubMed PMID: 5334643]
[6]
STONE W Jr. Alloplasty in surgery of the eye. The New England journal of medicine. 1958 Mar 6:258(10):486-90 contd
[PubMed PMID: 13517505]
[7]
Barnham JJ, Roper-Hall MJ. Keratoprosthesis: a long-term review. The British journal of ophthalmology. 1983 Jul:67(7):468-74
[PubMed PMID: 6860613]
[8]
Hicks CR, Crawford GJ, Dart JK, Grabner G, Holland EJ, Stulting RD, Tan DT, Bulsara M. AlphaCor: Clinical outcomes. Cornea. 2006 Oct:25(9):1034-42
[PubMed PMID: 17133049]
Level 2 (mid-level) evidence
[9]
Hollick EJ, Watson SL, Dart JK, Luthert PJ, Allan BD. Legeais BioKpro III keratoprosthesis implantation: long term results in seven patients. The British journal of ophthalmology. 2006 Sep:90(9):1146-51
[PubMed PMID: 16929061]
[10]
Nonpassopon M, Niparugs M, Cortina MS. Boston Type 1 Keratoprosthesis: Updated Perspectives. Clinical ophthalmology (Auckland, N.Z.). 2020:14():1189-1200. doi: 10.2147/OPTH.S219270. Epub 2020 Apr 29
[PubMed PMID: 32425503]
Level 3 (low-level) evidence
[11]
Talati RK, Hallak JA, Karas FI, de la Cruz J, Cortina MS. Retroprosthetic Membrane Formation in Boston Keratoprosthesis: A Case-Control-Matched Comparison of Titanium Versus PMMA Backplate. Cornea. 2018 Feb:37(2):145-150. doi: 10.1097/ICO.0000000000001462. Epub
[PubMed PMID: 29140862]
Level 2 (mid-level) evidence
[12]
Falcinelli G, Falsini B, Taloni M, Colliardo P, Falcinelli G. Modified osteo-odonto-keratoprosthesis for treatment of corneal blindness: long-term anatomical and functional outcomes in 181 cases. Archives of ophthalmology (Chicago, Ill. : 1960). 2005 Oct:123(10):1319-29
[PubMed PMID: 16219722]
Level 3 (low-level) evidence
[13]
Tan A, Tan DT, Tan XW, Mehta JS. Osteo-odonto keratoprosthesis: systematic review of surgical outcomes and complication rates. The ocular surface. 2012 Jan:10(1):15-25. doi: 10.1016/j.jtos.2012.01.003. Epub 2012 Jan 8
[PubMed PMID: 22330056]
Level 1 (high-level) evidence
[14]
Liu C, Okera S, Tandon R, Herold J, Hull C, Thorp S. Visual rehabilitation in end-stage inflammatory ocular surface disease with the osteo-odonto-keratoprosthesis: results from the UK. The British journal of ophthalmology. 2008 Sep:92(9):1211-7. doi: 10.1136/bjo.2007.130567. Epub 2008 May 29
[PubMed PMID: 18511541]
[15]
Marchi V, Ricci R, Pecorella I, Ciardi A, Di Tondo U. Osteo-odonto-keratoprosthesis. Description of surgical technique with results in 85 patients. Cornea. 1994 Mar:13(2):125-30
[PubMed PMID: 8156783]
[16]
Bakshi SK, Graney J, Paschalis EI, Agarwal S, Basu S, Iyer G, Liu C, Srinivasan B, Chodosh J. Design and Outcomes of a Novel Keratoprosthesis: Addressing Unmet Needs in End-Stage Cicatricial Corneal Blindness. Cornea. 2020 Apr:39(4):484-490. doi: 10.1097/ICO.0000000000002207. Epub
[PubMed PMID: 31724985]
[17]
Fariselli C, Toprak I, Al-Shymali O, Alio Del Barrio JL, Alio JL. Corneal transplantation outcomes after the extrusion of an intrastromal keratoprosthesis: a pilot study. Eye and vision (London, England). 2020:7():26. doi: 10.1186/s40662-020-00193-4. Epub 2020 May 8
[PubMed PMID: 32411808]
Level 3 (low-level) evidence
[18]
Basu S, Serna-Ojeda JC, Senthil S, Pappuru RR, Bagga B, Sangwan V. The Aurolab Keratoprosthesis (KPro) versus the Boston Type I Kpro: 5-year Clinical Outcomes in 134 Cases of Bilateral Corneal Blindness. American journal of ophthalmology. 2019 Sep:205():175-183. doi: 10.1016/j.ajo.2019.03.016. Epub 2019 Mar 22
[PubMed PMID: 30905723]
Level 2 (mid-level) evidence
[19]
Shanbhag SS, Senthil S, Mohamed A, Basu S. Outcomes of the Boston type 1 and the Aurolab keratoprosthesis in eyes with limbal stem cell deficiency. The British journal of ophthalmology. 2021 Apr:105(4):473-478. doi: 10.1136/bjophthalmol-2020-316369. Epub 2020 Jun 17
[PubMed PMID: 32554443]
[20]
Gomaa A, Comyn O, Liu C. Keratoprostheses in clinical practice - a review. Clinical & experimental ophthalmology. 2010 Mar:38(2):211-24. doi: 10.1111/j.1442-9071.2010.02231.x. Epub
[PubMed PMID: 20398109]
[21]
Tan XW, Thompson B, Konstantopoulos A, Goh TW, Setiawan M, Yam GH, Tan D, Khor KA, Mehta JS. Application of Graphene as Candidate Biomaterial for Synthetic Keratoprosthesis Skirt. Investigative ophthalmology & visual science. 2015 Oct:56(11):6605-11. doi: 10.1167/iovs.15-17306. Epub
[PubMed PMID: 26465888]
[22]
Zarei-Ghanavati M, Avadhanam V, Vasquez Perez A, Liu C. The osteo-odonto-keratoprosthesis. Current opinion in ophthalmology. 2017 Jul:28(4):397-402. doi: 10.1097/ICU.0000000000000388. Epub
[PubMed PMID: 28441214]
Level 3 (low-level) evidence
[23]
Hille K, Grabner G, Liu C, Colliardo P, Falcinelli G, Taloni M, Falcinelli G. Standards for modified osteoodontokeratoprosthesis (OOKP) surgery according to Strampelli and Falcinelli: the Rome-Vienna Protocol. Cornea. 2005 Nov:24(8):895-908
[PubMed PMID: 16227830]
[24]
Colby K. Pediatric Keratoprosthesis: A Promise Unfulfilled. Ophthalmology. 2018 Feb:125(2):147-149. doi: 10.1016/j.ophtha.2017.10.030. Epub
[PubMed PMID: 29389402]
[25]
Fung SSM, Jabbour S, Harissi-Dagher M, Tan RRG, Hamel P, Baig K, Ali A. Visual Outcomes and Complications of Type I Boston Keratoprosthesis in Children: A Retrospective Multicenter Study and Literature Review. Ophthalmology. 2018 Feb:125(2):153-160. doi: 10.1016/j.ophtha.2017.07.009. Epub 2017 Aug 12
[PubMed PMID: 28807636]
Level 2 (mid-level) evidence
[26]
Lim JI, Machen L, Arteaga A, Karas FI, Hyde R, Cao D, Niec M, Vajaranant TS, Cortina MS. COMPARISON OF VISUAL AND ANATOMICAL OUTCOMES OF EYES UNDERGOING TYPE I BOSTON KERATOPROSTHESIS WITH COMBINATION PARS PLANA VITRECTOMY WITH EYES WITHOUT COMBINATION VITRECTOMY. Retina (Philadelphia, Pa.). 2018 Sep:38 Suppl 1(Suppl 1):S125-S133. doi: 10.1097/IAE.0000000000002036. Epub
[PubMed PMID: 29370031]
[27]
Huang Y, Dong Y, Wang L, Du G, Yu J, Song J, Chiang HH. Long-term outcomes of MICOF keratoprosthesis in the end stage of autoimmune dry eyes: an experience in China. The British journal of ophthalmology. 2012 Jan:96(1):28-33. doi: 10.1136/bjo.2010.193029. Epub 2011 Mar 31
[PubMed PMID: 21454379]
[28]
Behlau I, Martin KV, Martin JN, Naumova EN, Cadorette JJ, Sforza JT, Pineda R 2nd, Dohlman CH. Infectious endophthalmitis in Boston keratoprosthesis: incidence and prevention. Acta ophthalmologica. 2014 Nov:92(7):e546-55. doi: 10.1111/aos.12309. Epub 2014 Jan 25
[PubMed PMID: 24460594]
[29]
Geoffrion D, Harissi-Dagher M. Glaucoma Risk Factors and Outcomes Following Boston Keratoprosthesis Type 1 Surgery. American journal of ophthalmology. 2021 Jun:226():56-67. doi: 10.1016/j.ajo.2021.01.006. Epub 2021 Jan 22
[PubMed PMID: 33493469]
[30]
Nascimento E Silva R, Taniguchi EV, Cruzat A, Paschalis EI, Pasquale LR, Colby KA, Dohlman CH, Chodosh J, Shen LQ. Angle Anatomy and Glaucoma in Patients With Boston Keratoprosthesis. Cornea. 2020 Jun:39(6):713-719. doi: 10.1097/ICO.0000000000002216. Epub
[PubMed PMID: 31764284]
[31]
Quercia AZF, Silva LD, de Oliveira F, Teixeira SH, de Sousa LB, de Oliveira LA. Visual Field Characteristics of Type I Boston Keratoprosthesis Patients Without Glaucoma. Journal of glaucoma. 2021 Jun 1:30(6):532-536. doi: 10.1097/IJG.0000000000001737. Epub
[PubMed PMID: 33149106]
[32]
Samarawickrama C, Strouthidis N, Wilkins MR. Boston keratoprosthesis type 1: outcomes of the first 38 cases performed at Moorfields Eye Hospital. Eye (London, England). 2018 Jun:32(6):1087-1092. doi: 10.1038/s41433-018-0016-4. Epub 2018 Feb 14
[PubMed PMID: 29440740]
Level 3 (low-level) evidence
[33]
Vajaranant TS, Liu J, Wilensky J, Cortina MS, Aref AA. Innovative approaches to glaucoma management of Boston keratoprosthesis type 1. Current ophthalmology reports. 2016 Sep:4(3):147-153. doi: 10.1007/s40135-016-0102-3. Epub 2016 Jul 26
[PubMed PMID: 28529825]
[34]
Kanu LN, Niparugs M, Nonpassopon M, Karas FI, de la Cruz JM, Cortina MS. Predictive factors of Boston Type I Keratoprosthesis outcomes: A long-term analysis. The ocular surface. 2020 Oct:18(4):613-619. doi: 10.1016/j.jtos.2020.07.012. Epub 2020 Jul 21
[PubMed PMID: 32702418]
[35]
Priddy J, Bardan AS, Tawfik HS, Liu C. Systematic Review and Meta-Analysis of the Medium- and Long-Term Outcomes of the Boston Type 1 Keratoprosthesis. Cornea. 2019 Nov:38(11):1465-1473. doi: 10.1097/ICO.0000000000002098. Epub
[PubMed PMID: 31403526]
Level 1 (high-level) evidence
[36]
Daoud R, Sabeti S, Harissi-Dagher M. Management of corneal melt in patients with Boston Keratoprosthesis Type 1: Repair versus repeat. The ocular surface. 2020 Oct:18(4):713-717. doi: 10.1016/j.jtos.2020.07.005. Epub 2020 Aug 7
[PubMed PMID: 32777438]
[37]
Rishi P, Rishi E, Agarwal V, Nair S, Iyer G, Srinivasan B, Agarwal S. Vitreoretinal Complications and Outcomes in 92 Eyes Undergoing Surgery for Modified Osteo-Odonto-Keratoprosthesis: A 10-Year Review. Ophthalmology. 2018 Jun:125(6):832-841. doi: 10.1016/j.ophtha.2017.12.003. Epub 2018 Jan 17
[PubMed PMID: 29342438]
[38]
Avadhanam VS, Smith J, Poostchi A, Chervenkoff J, Al Raqqad N, Francis I, Liu CS. Detection of laminar resorption in osteo-odonto-keratoprostheses. The ocular surface. 2019 Jan:17(1):78-82. doi: 10.1016/j.jtos.2018.09.004. Epub 2018 Sep 15
[PubMed PMID: 30227262]
[39]
Iyer G, Srinivasan B, Agarwal S, Rachapalle SR. Laminar resorption in modified osteo-odonto-keratoprosthesis procedure: a cause for concern. American journal of ophthalmology. 2014 Aug:158(2):263-269.e2. doi: 10.1016/j.ajo.2014.03.004. Epub 2014 Mar 12
[PubMed PMID: 24631477]
[40]
Jeng BH, Ahmad S. In Pursuit of the Elimination of Corneal Blindness: Is Establishing Eye Banks and Training Surgeons Enough? Ophthalmology. 2021 Jun:128(6):813-815. doi: 10.1016/j.ophtha.2020.06.042. Epub 2020 Jul 29
[PubMed PMID: 32739177]
[41]
Gain P, Jullienne R, He Z, Aldossary M, Acquart S, Cognasse F, Thuret G. Global Survey of Corneal Transplantation and Eye Banking. JAMA ophthalmology. 2016 Feb:134(2):167-73. doi: 10.1001/jamaophthalmol.2015.4776. Epub
[PubMed PMID: 26633035]
Level 3 (low-level) evidence
[42]
Thuret G,Courrier E,Poinard S,Gain P,Baud'Huin M,Martinache I,Cursiefen C,Maier P,Hjortdal J,Sanchez Ibanez J,Ponzin D,Ferrari S,Jones G,Griffoni C,Rooney P,Bennett K,Armitage WJ,Figueiredo F,Nuijts R,Dickman M, One threat, different answers: the impact of COVID-19 pandemic on cornea donation and donor selection across Europe. The British journal of ophthalmology. 2020 Nov 26;
[PubMed PMID: 33243832]
[43]
Belin MW, Güell JL, Grabner G. Suggested Guidelines for Reporting Keratoprosthesis Results: Consensus Opinion of the Cornea Society, Asia Cornea Society, EuCornea, PanCornea, and the KPRO Study Group. Cornea. 2016 Feb:35(2):143-4. doi: 10.1097/ICO.0000000000000703. Epub
[PubMed PMID: 26619387]
Level 3 (low-level) evidence