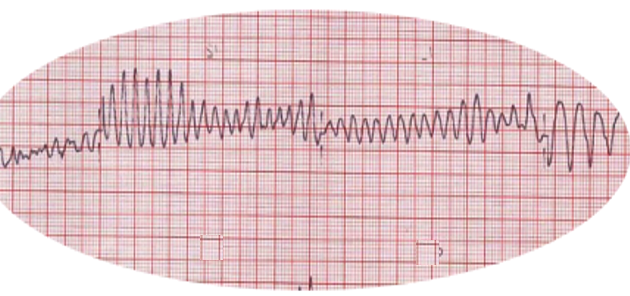
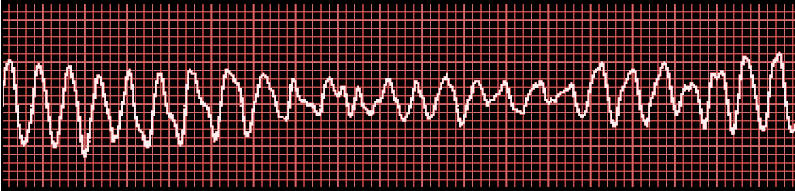
[1]
American College of Cardiology/American Heart Association Task Force on Clinical Data Standards (ACC/AHA/HRS Writing Committee to Develop Data Standards on Electrophysiology), Buxton AE, Calkins H, Callans DJ, DiMarco JP, Fisher JD, Greene HL, Haines DE, Hayes DL, Heidenreich PA, Miller JM, Poppas A, Prystowsky EN, Schoenfeld MH, Zimetbaum PJ, Goff DC, Grover FL, Malenka DJ, Peterson ED, Radford MJ, Redberg RF. ACC/AHA/HRS 2006 key data elements and definitions for electrophysiological studies and procedures: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Data Standards (ACC/AHA/HRS Writing Committee to Develop Data Standards on Electrophysiology). Circulation. 2006 Dec 5:114(23):2534-70
[PubMed PMID: 17130345]
[2]
Glinge C, Sattler S, Jabbari R, Tfelt-Hansen J. Epidemiology and genetics of ventricular fibrillation during acute myocardial infarction. Journal of geriatric cardiology : JGC. 2016 Sep:13(9):789-797
[PubMed PMID: 27899944]
[3]
Jabbari R, Engstrøm T, Glinge C, Risgaard B, Jabbari J, Winkel BG, Terkelsen CJ, Tilsted HH, Jensen LO, Hougaard M, Chiuve SE, Pedersen F, Svendsen JH, Haunsø S, Albert CM, Tfelt-Hansen J. Incidence and risk factors of ventricular fibrillation before primary angioplasty in patients with first ST-elevation myocardial infarction: a nationwide study in Denmark. Journal of the American Heart Association. 2015 Jan 5:4(1):e001399. doi: 10.1161/JAHA.114.001399. Epub 2015 Jan 5
[PubMed PMID: 25559012]
[4]
Samie FH,Jalife J, Mechanisms underlying ventricular tachycardia and its transition to ventricular fibrillation in the structurally normal heart. Cardiovascular research. 2001 May
[PubMed PMID: 11334828]
[5]
Bezzina CR, Pazoki R, Bardai A, Marsman RF, de Jong JSSG, Blom MT, Scicluna BP, Jukema JW, Bindraban NR, Lichtner P, Pfeufer A, Bishopric NH, Roden DM, Meitinger T, Chugh SS, Myerburg RJ, Jouven X, Kääb S, Dekker LRC, Tan HL, Tanck MWT, Wilde AAM. Genome-wide association study identifies a susceptibility locus at 21q21 for ventricular fibrillation in acute myocardial infarction. Nature genetics. 2010 Aug:42(8):688-691. doi: 10.1038/ng.623. Epub 2010 Jul 11
[PubMed PMID: 20622880]
[6]
Adabag AS, Luepker RV, Roger VL, Gersh BJ. Sudden cardiac death: epidemiology and risk factors. Nature reviews. Cardiology. 2010 Apr:7(4):216-25. doi: 10.1038/nrcardio.2010.3. Epub 2010 Feb 9
[PubMed PMID: 20142817]
[7]
Al-Khatib SM, Stevenson WG, Ackerman MJ, Bryant WJ, Callans DJ, Curtis AB, Deal BJ, Dickfeld T, Field ME, Fonarow GC, Gillis AM, Granger CB, Hammill SC, Hlatky MA, Joglar JA, Kay GN, Matlock DD, Myerburg RJ, Page RL. 2017 AHA/ACC/HRS Guideline for Management of Patients With Ventricular Arrhythmias and the Prevention of Sudden Cardiac Death: Executive Summary: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Rhythm Society. Journal of the American College of Cardiology. 2018 Oct 2:72(14):1677-1749. doi: 10.1016/j.jacc.2017.10.053. Epub 2017 Oct 30
[PubMed PMID: 29097294]
Level 1 (high-level) evidence
[8]
Dumas F, Cariou A, Manzo-Silberman S, Grimaldi D, Vivien B, Rosencher J, Empana JP, Carli P, Mira JP, Jouven X, Spaulding C. Immediate percutaneous coronary intervention is associated with better survival after out-of-hospital cardiac arrest: insights from the PROCAT (Parisian Region Out of hospital Cardiac ArresT) registry. Circulation. Cardiovascular interventions. 2010 Jun 1:3(3):200-7. doi: 10.1161/CIRCINTERVENTIONS.109.913665. Epub 2010 May 18
[PubMed PMID: 20484098]
[9]
Honarmand K, Mepham C, Ainsworth C, Khalid Z. Adherence to advanced cardiovascular life support (ACLS) guidelines during in-hospital cardiac arrest is associated with improved outcomes. Resuscitation. 2018 Aug:129():76-81. doi: 10.1016/j.resuscitation.2018.06.005. Epub 2018 Jun 6
[PubMed PMID: 29885353]
[10]
Chan PS, Krumholz HM, Nichol G, Nallamothu BK, American Heart Association National Registry of Cardiopulmonary Resuscitation Investigators. Delayed time to defibrillation after in-hospital cardiac arrest. The New England journal of medicine. 2008 Jan 3:358(1):9-17. doi: 10.1056/NEJMoa0706467. Epub
[PubMed PMID: 18172170]
[11]
Laina A, Karlis G, Liakos A, Georgiopoulos G, Oikonomou D, Kouskouni E, Chalkias A, Xanthos T. Amiodarone and cardiac arrest: Systematic review and meta-analysis. International journal of cardiology. 2016 Oct 15:221():780-8. doi: 10.1016/j.ijcard.2016.07.138. Epub 2016 Jul 9
[PubMed PMID: 27434349]
Level 1 (high-level) evidence
[12]
Bardy GH, Lee KL, Mark DB, Poole JE, Packer DL, Boineau R, Domanski M, Troutman C, Anderson J, Johnson G, McNulty SE, Clapp-Channing N, Davidson-Ray LD, Fraulo ES, Fishbein DP, Luceri RM, Ip JH, Sudden Cardiac Death in Heart Failure Trial (SCD-HeFT) Investigators. Amiodarone or an implantable cardioverter-defibrillator for congestive heart failure. The New England journal of medicine. 2005 Jan 20:352(3):225-37
[PubMed PMID: 15659722]
[13]
Levantesi G, Scarano M, Marfisi R, Borrelli G, Rutjes AW, Silletta MG, Tognoni G, Marchioli R. Meta-analysis of effect of statin treatment on risk of sudden death. The American journal of cardiology. 2007 Dec 1:100(11):1644-50
[PubMed PMID: 18036362]
Level 1 (high-level) evidence
[14]
Holmberg M, Holmberg S, Herlitz J. Incidence, duration and survival of ventricular fibrillation in out-of-hospital cardiac arrest patients in sweden. Resuscitation. 2000 Mar:44(1):7-17
[PubMed PMID: 10699695]
[15]
Demidova MM, Smith JG, Höijer CJ, Holmqvist F, Erlinge D, Platonov PG. Prognostic impact of early ventricular fibrillation in patients with ST-elevation myocardial infarction treated with primary PCI. European heart journal. Acute cardiovascular care. 2012 Dec:1(4):302-11. doi: 10.1177/2048872612463553. Epub
[PubMed PMID: 24062921]