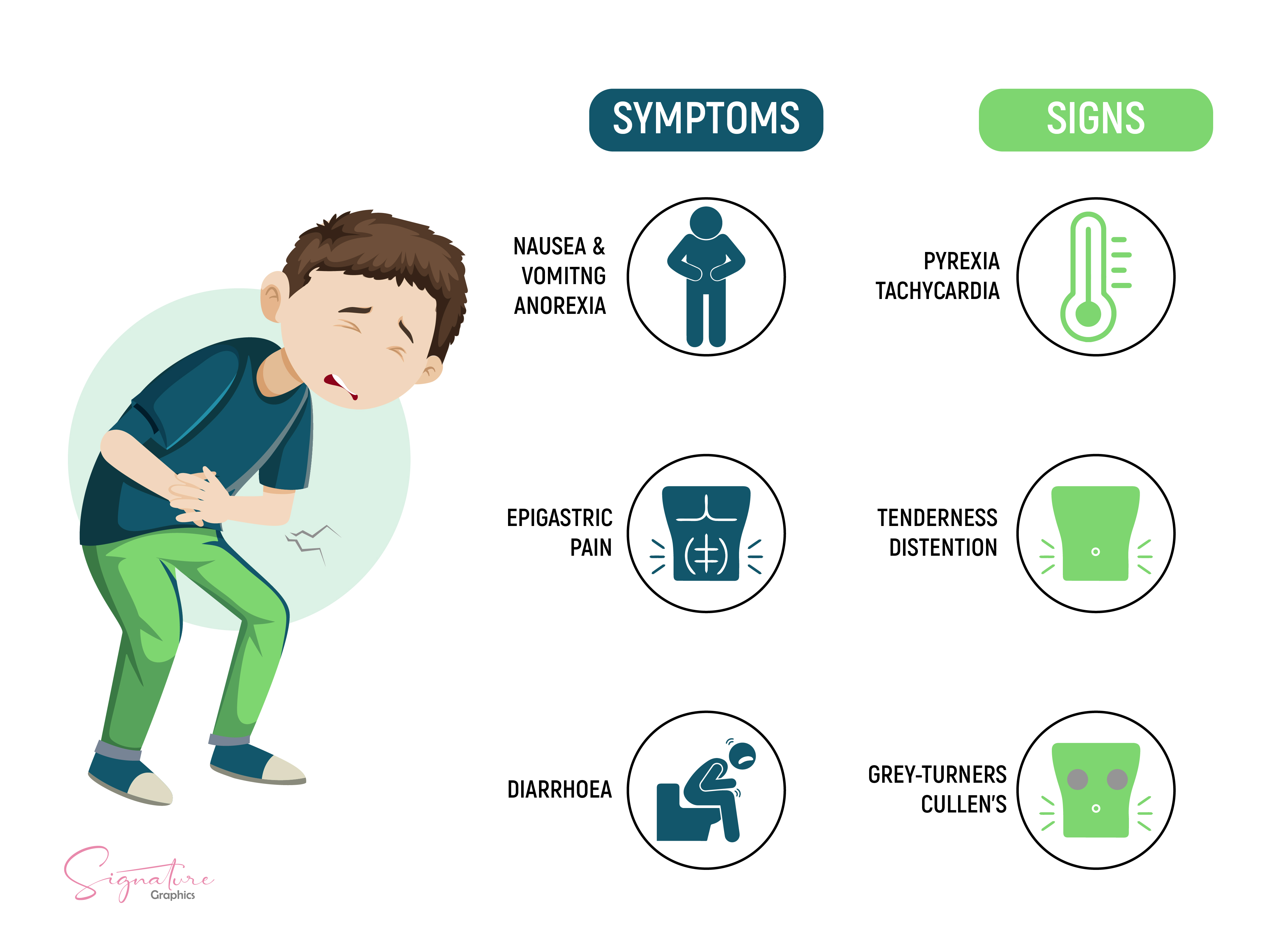
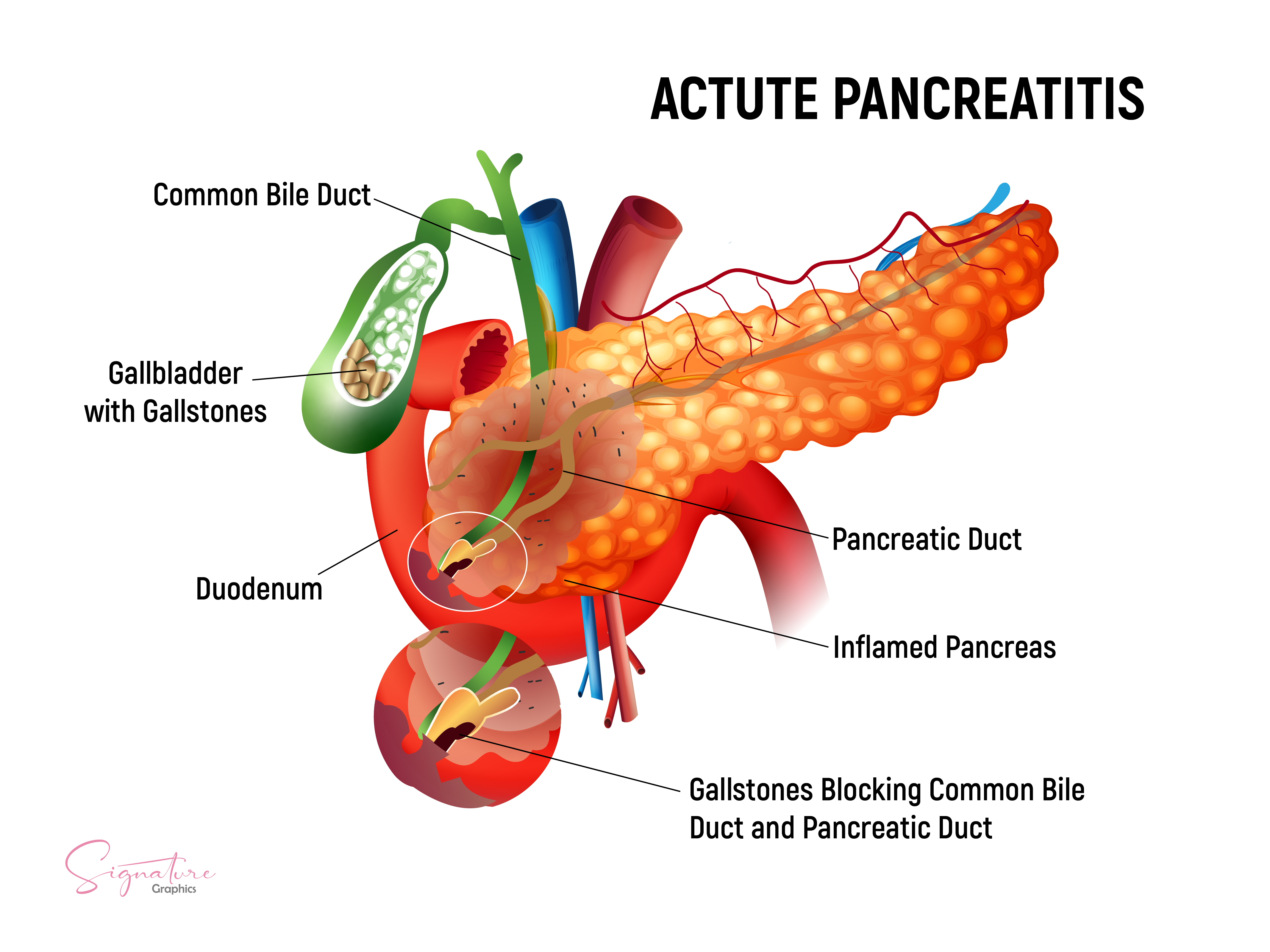
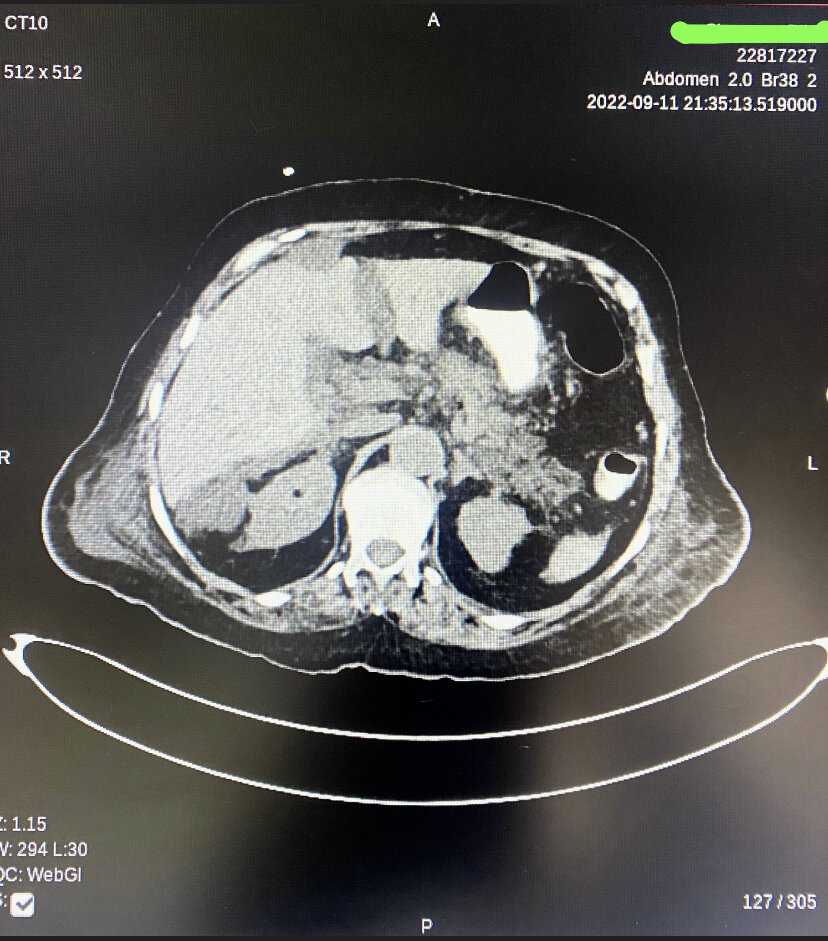
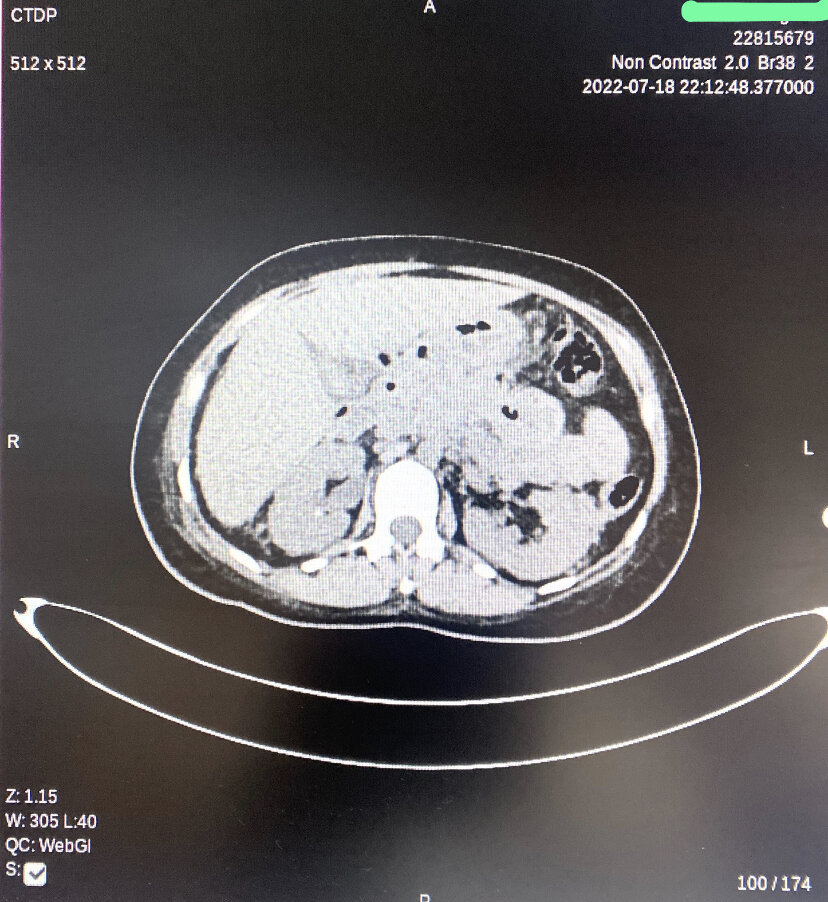
[1]
Werge M,Novovic S,Schmidt PN,Gluud LL, Infection increases mortality in necrotizing pancreatitis: A systematic review and meta-analysis. Pancreatology : official journal of the International Association of Pancreatology (IAP) ... [et al.]. 2016 Sep-Oct
[PubMed PMID: 27449605]
Level 1 (high-level) evidence
[2]
Valverde-López F,Wilcox CM,Redondo-Cerezo E, Evaluation and management of acute pancreatitis in Spain. Gastroenterologia y hepatologia. 2018 Dec
[PubMed PMID: 30149943]
[3]
Kahaleh M, Management of pancreatitis and pancreatic: fluid collections. Revista de gastroenterologia del Peru : organo oficial de la Sociedad de Gastroenterologia del Peru. 2018 Apr-Jun
[PubMed PMID: 30118464]
[4]
Bazerbachi F,Haffar S,Hussain MT,Vargas EJ,Watt KD,Murad MH,Chari S,Abu Dayyeh BK, Systematic review of acute pancreatitis associated with interferon-α or pegylated interferon-α: Possible or definitive causation? Pancreatology : official journal of the International Association of Pancreatology (IAP) ... [et al.]. 2018 Oct
[PubMed PMID: 30061072]
Level 1 (high-level) evidence
[5]
Ortiz Morales CM,Girela Baena EL,Olalla Muñoz JR,Parlorio de Andrés E,López Corbalán JA, Radiology of acute pancreatitis today: the Atlanta classification and the current role of imaging in its diagnosis and treatment. Radiologia. 2019 Nov - Dec
[PubMed PMID: 31153603]
[7]
Sepúlveda EVF,Guerrero-Lozano R, Acute pancreatitis and recurrent acute pancreatitis: an exploration of clinical and etiologic factors and outcomes. Jornal de pediatria. 2018 Jul 31
[PubMed PMID: 30075118]
[9]
de Pretis N,Amodio A,Frulloni L, Hypertriglyceridemic pancreatitis: Epidemiology, pathophysiology and clinical management. United European gastroenterology journal. 2018 Jun
[PubMed PMID: 30083325]
[10]
Kirkegård J,Cronin-Fenton D,Heide-Jørgensen U,Mortensen FV, Acute Pancreatitis and Pancreatic Cancer Risk: A Nationwide Matched-Cohort Study in Denmark. Gastroenterology. 2018 May
[PubMed PMID: 29432727]
[11]
Johnstone C, Pathophysiology and nursing management of acute pancreatitis. Nursing standard (Royal College of Nursing (Great Britain) : 1987). 2018 Jun 28
[PubMed PMID: 29952150]
[12]
Constantinoiu S,Cochior D, Severe Acute Pancreatitis - Determinant Factors and Current Therapeutic Conduct. Chirurgia (Bucharest, Romania : 1990). 2018 May-Jun
[PubMed PMID: 29981669]
[13]
Choi HW,Park HJ,Choi SY,Do JH,Yoon NY,Ko A,Lee ES, Early Prediction of the Severity of Acute Pancreatitis Using Radiologic and Clinical Scoring Systems With Classification Tree Analysis. AJR. American journal of roentgenology. 2018 Nov
[PubMed PMID: 30160978]
[14]
Mandalia A,Wamsteker EJ,DiMagno MJ, Recent advances in understanding and managing acute pancreatitis. F1000Research. 2018
[PubMed PMID: 30026919]
Level 3 (low-level) evidence
[15]
Smeets XJNM,Litjens G,Gijsbers K,Prokop M,Drenth JPH,Hermans J,van Geenen EJM, The Accuracy of Pancreatic Perfusion Computed Tomography and Angiography in Predicting Necrotizing Pancreatitis: A Systematic Review. Pancreas. 2018 Jul
[PubMed PMID: 29894416]
Level 1 (high-level) evidence
[16]
Barrie J,Jamdar S,Smith N,McPherson SJ,Siriwardena AK,O'Reilly DA, Mis-use of antibiotics in acute pancreatitis: Insights from the United Kingdom's National Confidential Enquiry into patient outcome and death (NCEPOD) survey of acute pancreatitis. Pancreatology : official journal of the International Association of Pancreatology (IAP) ... [et al.]. 2018 May 24
[PubMed PMID: 30075909]
Level 3 (low-level) evidence
[17]
Reynolds PT,Brady EK,Chawla S, The utility of early cross-sectional imaging to evaluate suspected acute mild pancreatitis. Annals of gastroenterology. 2018 Sep-Oct
[PubMed PMID: 30174401]
Level 2 (mid-level) evidence
[18]
Sellers ZM,Abu-El-Haija M,Husain SZ,Morinville V, New Management Guidelines for Both Children and Adults With Acute Pancreatitis. Gastroenterology. 2018 Jul
[PubMed PMID: 29890113]
[19]
de-Madaria E,Buxbaum JL,Maisonneuve P,García García de Paredes A,Zapater P,Guilabert L,Vaillo-Rocamora A,Rodríguez-Gandía MÁ,Donate-Ortega J,Lozada-Hernández EE,Collazo Moreno AJR,Lira-Aguilar A,Llovet LP,Mehta R,Tandel R,Navarro P,Sánchez-Pardo AM,Sánchez-Marin C,Cobreros M,Fernández-Cabrera I,Casals-Seoane F,Casas Deza D,Lauret-Braña E,Martí-Marqués E,Camacho-Montaño LM,Ubieto V,Ganuza M,Bolado F,ERICA Consortium., Aggressive or Moderate Fluid Resuscitation in Acute Pancreatitis. The New England journal of medicine. 2022 Sep 15;
[PubMed PMID: 36103415]
[20]
Gardner TB, Fluid Resuscitation in Acute Pancreatitis - Going over the WATERFALL. The New England journal of medicine. 2022 Sep 15
[PubMed PMID: 36103418]
[21]
Petrov MS, Yadav D. Global epidemiology and holistic prevention of pancreatitis. Nature reviews. Gastroenterology & hepatology. 2019 Mar:16(3):175-184. doi: 10.1038/s41575-018-0087-5. Epub
[PubMed PMID: 30482911]
[22]
Samarasekera E,Mahammed S,Carlisle S,Charnley R,Guideline Committee., Pancreatitis: summary of NICE guidance. BMJ (Clinical research ed.). 2018 Sep 5
[PubMed PMID: 30185473]
[23]
Crockett SD, Wani S, Gardner TB, Falck-Ytter Y, Barkun AN, American Gastroenterological Association Institute Clinical Guidelines Committee. American Gastroenterological Association Institute Guideline on Initial Management of Acute Pancreatitis. Gastroenterology. 2018 Mar:154(4):1096-1101. doi: 10.1053/j.gastro.2018.01.032. Epub 2018 Feb 3
[PubMed PMID: 29409760]
[24]
Leppäniemi A,Tolonen M,Tarasconi A,Segovia-Lohse H,Gamberini E,Kirkpatrick AW,Ball CG,Parry N,Sartelli M,Wolbrink D,van Goor H,Baiocchi G,Ansaloni L,Biffl W,Coccolini F,Di Saverio S,Kluger Y,Moore E,Catena F, 2019 WSES guidelines for the management of severe acute pancreatitis. World journal of emergency surgery : WJES. 2019
[PubMed PMID: 31210778]
[25]
Cai W,Liu F,Wen Y,Han C,Prasad M,Xia Q,Singh VK,Sutton R,Huang W, Pain Management in Acute Pancreatitis: A Systematic Review and Meta-Analysis of Randomised Controlled Trials. Frontiers in medicine. 2021
[PubMed PMID: 34977084]
Level 1 (high-level) evidence
[26]
Basurto Ona X,Rigau Comas D,Urrútia G, Opioids for acute pancreatitis pain. The Cochrane database of systematic reviews. 2013 Jul 26
[PubMed PMID: 23888429]
Level 1 (high-level) evidence
[27]
Stigliano S,Sternby H,de Madaria E,Capurso G,Petrov MS, Early management of acute pancreatitis: A review of the best evidence. Digestive and liver disease : official journal of the Italian Society of Gastroenterology and the Italian Association for the Study of the Liver. 2017 Jun
[PubMed PMID: 28262458]
[28]
Arvanitakis M,Dumonceau JM,Albert J,Badaoui A,Bali MA,Barthet M,Besselink M,Deviere J,Oliveira Ferreira A,Gyökeres T,Hritz I,Hucl T,Milashka M,Papanikolaou IS,Poley JW,Seewald S,Vanbiervliet G,van Lienden K,van Santvoort H,Voermans R,Delhaye M,van Hooft J, Endoscopic management of acute necrotizing pancreatitis: European Society of Gastrointestinal Endoscopy (ESGE) evidence-based multidisciplinary guidelines. Endoscopy. 2018 May
[PubMed PMID: 29631305]
[29]
Abu-El-Haija M,Kumar S,Quiros JA,Balakrishnan K,Barth B,Bitton S,Eisses JF,Foglio EJ,Fox V,Francis D,Freeman AJ,Gonska T,Grover AS,Husain SZ,Kumar R,Lapsia S,Lin T,Liu QY,Maqbool A,Sellers ZM,Szabo F,Uc A,Werlin SL,Morinville VD, Management of Acute Pancreatitis in the Pediatric Population: A Clinical Report From the North American Society for Pediatric Gastroenterology, Hepatology and Nutrition Pancreas Committee. Journal of pediatric gastroenterology and nutrition. 2018 Jan
[PubMed PMID: 29280782]
[30]
Working Group IAP/APA Acute Pancreatitis Guidelines., IAP/APA evidence-based guidelines for the management of acute pancreatitis. Pancreatology : official journal of the International Association of Pancreatology (IAP) ... [et al.]. 2013 Jul-Aug
[PubMed PMID: 24054878]
Level 1 (high-level) evidence
[31]
Mofidi R,Duff MD,Wigmore SJ,Madhavan KK,Garden OJ,Parks RW, Association between early systemic inflammatory response, severity of multiorgan dysfunction and death in acute pancreatitis. The British journal of surgery. 2006 Jun;
[PubMed PMID: 16671062]
[32]
Buter A,Imrie CW,Carter CR,Evans S,McKay CJ, Dynamic nature of early organ dysfunction determines outcome in acute pancreatitis. The British journal of surgery. 2002 Mar;
[PubMed PMID: 11872053]
[33]
Singh VK,Wu BU,Bollen TL,Repas K,Maurer R,Mortele KJ,Banks PA, Early systemic inflammatory response syndrome is associated with severe acute pancreatitis. Clinical gastroenterology and hepatology : the official clinical practice journal of the American Gastroenterological Association. 2009 Nov
[PubMed PMID: 19686869]
[34]
Gao W,Yang HX,Ma CE, The Value of BISAP Score for Predicting Mortality and Severity in Acute Pancreatitis: A Systematic Review and Meta-Analysis. PloS one. 2015
[PubMed PMID: 26091293]
Level 1 (high-level) evidence
[35]
Mortele KJ,Wiesner W,Intriere L,Shankar S,Zou KH,Kalantari BN,Perez A,vanSonnenberg E,Ros PR,Banks PA,Silverman SG, A modified CT severity index for evaluating acute pancreatitis: improved correlation with patient outcome. AJR. American journal of roentgenology. 2004 Nov
[PubMed PMID: 15505289]
[36]
Gódi S,Erőss B,Gyömbér Z,Szentesi A,Farkas N,Párniczky A,Sarlós P,Bajor J,Czimmer J,Mikó A,Márta K,Hágendorn R,Márton Z,Verzár Z,Czakó L,Szepes Z,Vincze Á,Hegyi P, Centralized care for acute pancreatitis significantly improves outcomes. Journal of gastrointestinal and liver diseases : JGLD. 2018 Jun
[PubMed PMID: 29922760]
[37]
Branquinho D,Ramos-Andrade D,Elvas L,Amaro P,Ferreira M,Sofia C, Drug-Induced Acute Pancreatitis and Pseudoaneurysms: An Ominous Combination. GE Portuguese journal of gastroenterology. 2016 Nov-Dec
[PubMed PMID: 28868485]
[38]
Srinivasan G,Venkatakrishnan L,Sambandam S,Singh G,Kaur M,Janarthan K,John BJ, Current concepts in the management of acute pancreatitis. Journal of family medicine and primary care. 2016 Oct-Dec
[PubMed PMID: 28348985]
[39]
Jin T,Jiang K,Deng L,Guo J,Wu Y,Wang Z,Shi N,Zhang X,Lin Z,Asrani V,Jones P,Mittal A,Phillips A,Sutton R,Huang W,Yang X,Xia Q,Windsor JA, Response and outcome from fluid resuscitation in acute pancreatitis: a prospective cohort study. HPB : the official journal of the International Hepato Pancreato Biliary Association. 2018 Nov
[PubMed PMID: 30170979]
[40]
Amas Gómez L,Zubia Olaskoaga F, Results of the modification of an acute pancreatitis management protocol in Intensive Care medicine. Medicina intensiva. 2019 Dec
[PubMed PMID: 30072142]
[41]
Garret C,Péron M,Reignier J,Le Thuaut A,Lascarrou JB,Douane F,Lerhun M,Archambeaud I,Brulé N,Bretonnière C,Zambon O,Nicolet L,Regenet N,Guitton C,Coron E, Risk factors and outcomes of infected pancreatic necrosis: Retrospective cohort of 148 patients admitted to the ICU for acute pancreatitis. United European gastroenterology journal. 2018 Jul
[PubMed PMID: 30023069]
Level 2 (mid-level) evidence