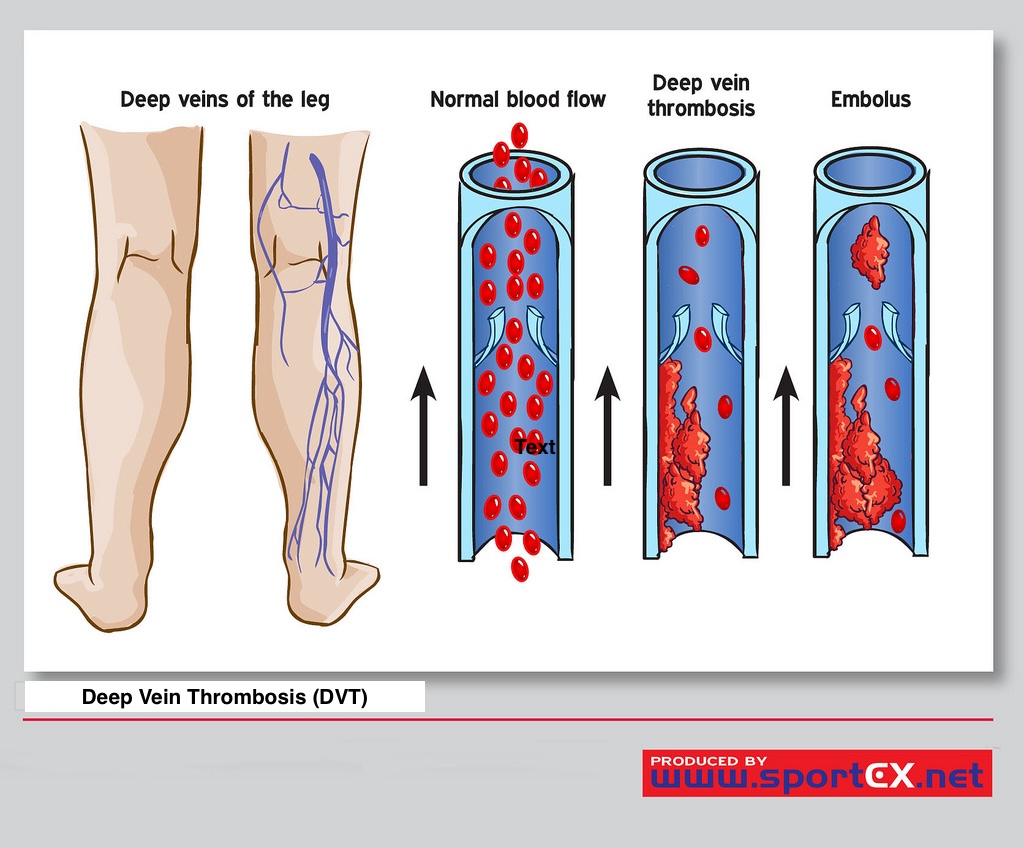
[1]
Huang Y,Ge H,Wang X,Zhang X, Association Between Blood Lipid Levels and Lower Extremity Deep Venous Thrombosis: A Population-Based Cohort Study. Clinical and applied thrombosis/hemostasis : official journal of the International Academy of Clinical and Applied Thrombosis/Hemostasis. 2022 Jan-Dec
[PubMed PMID: 36189865]
[2]
Chen R,Feng R,Jiang S,Chang G,Hu Z,Yao C,Jia B,Wang S,Wang S, Stent patency rates and prognostic factors of endovascular intervention for iliofemoral vein occlusion in post-thrombotic syndrome. BMC surgery. 2022 Jul 12;
[PubMed PMID: 35831845]
[3]
Albricker ACL,Freire CMV,Santos SND,Alcantara ML,Saleh MH,Cantisano AL,Teodoro JAR,Porto CLL,Amaral SID,Veloso OCG,Petisco ACGP,Barros FS,Barros MVL,Souza AJ,Sobreira ML,Miranda RB,Moraes D,Verrastro CGY,Mançano AD,Lima RSL,Muglia VF,Matushita CS,Lopes RW,Coutinho AMN,Pianta DB,Santos AASMDD,Naves BL,Vieira MLC,Rochitte CE, Joint Guideline on Venous Thromboembolism - 2022. Arquivos brasileiros de cardiologia. 2022 Apr;
[PubMed PMID: 35508060]
[5]
Parker K,Thachil J, The use of direct oral anticoagulants in chronic kidney disease. British journal of haematology. 2018 Oct
[PubMed PMID: 30183070]
[6]
Naringrekar H,Sun J,Ko C,Rodgers SK, It's Not All Deep Vein Thrombosis: Sonography of the Painful Lower Extremity With Multimodality Correlation. Journal of ultrasound in medicine : official journal of the American Institute of Ultrasound in Medicine. 2019 Apr
[PubMed PMID: 30171620]
[7]
Seifi A,Dengler B,Martinez P,Godoy DA, Pulmonary embolism in severe traumatic brain injury. Journal of clinical neuroscience : official journal of the Neurosurgical Society of Australasia. 2018 Aug 25
[PubMed PMID: 30154000]
[8]
Belcaro G,Cornelli U,Dugall M,Hosoi M,Cotellese R,Feragalli B, Long-haul flights, edema, and thrombotic events: prevention with stockings and Pycnogenol® supplementation (LONFLIT Registry Study). Minerva cardioangiologica. 2018 Apr
[PubMed PMID: 29512362]
[9]
Liu Z,Tao X,Chen Y,Fan Z,Li Y, Bed rest versus early ambulation with standard anticoagulation in the management of deep vein thrombosis: a meta-analysis. PloS one. 2015
[PubMed PMID: 25860350]
Level 1 (high-level) evidence
[10]
Prins MH,Lensing AW,Brighton TA,Lyons RM,Rehm J,Trajanovic M,Davidson BL,Beyer-Westendorf J,Pap ÁF,Berkowitz SD,Cohen AT,Kovacs MJ,Wells PS,Prandoni P, Oral rivaroxaban versus enoxaparin with vitamin K antagonist for the treatment of symptomatic venous thromboembolism in patients with cancer (EINSTEIN-DVT and EINSTEIN-PE): a pooled subgroup analysis of two randomised controlled trials. The Lancet. Haematology. 2014 Oct
[PubMed PMID: 27030066]
Level 1 (high-level) evidence
[11]
Ruskin KJ, Deep vein thrombosis and venous thromboembolism in trauma. Current opinion in anaesthesiology. 2018 Apr
[PubMed PMID: 29334497]
Level 3 (low-level) evidence
[12]
Mechanick JI,Apovian C,Brethauer S,Garvey WT,Joffe AM,Kim J,Kushner RF,Lindquist R,Pessah-Pollack R,Seger J,Urman RD,Adams S,Cleek JB,Correa R,Figaro MK,Flanders K,Grams J,Hurley DL,Kothari S,Seger MV,Still CD, Clinical practice guidelines for the perioperative nutrition, metabolic, and nonsurgical support of patients undergoing bariatric procedures - 2019 update: cosponsored by American Association of Clinical Endocrinologists/American College of Endocrinology, The Obesity Society, American Society for Metabolic & Bariatric Surgery, Obesity Medicine Association, and American Society of Anesthesiologists. Surgery for obesity and related diseases : official journal of the American Society for Bariatric Surgery. 2020 Feb
[PubMed PMID: 31917200]
Level 1 (high-level) evidence
[13]
Vayá A,Suescun M, Hemorheological parameters as independent predictors of venous thromboembolism. Clinical hemorheology and microcirculation. 2013
[PubMed PMID: 22954636]
[15]
Wypasek E, Undas A. Protein C and protein S deficiency - practical diagnostic issues. Advances in clinical and experimental medicine : official organ Wroclaw Medical University. 2013 Jul-Aug:22(4):459-67
[PubMed PMID: 23986205]
Level 3 (low-level) evidence
[16]
Lee SY,Niikura T,Iwakura T,Sakai Y,Kuroda R,Kurosaka M, Thrombin-antithrombin III complex tests. Journal of orthopaedic surgery (Hong Kong). 2017 Jan 1;
[PubMed PMID: 28418276]
[17]
Mori S,Ogata F,Tsunoda R, Risk of venous thromboembolism associated with Janus kinase inhibitors for rheumatoid arthritis: case presentation and literature review. Clinical rheumatology. 2021 Nov
[PubMed PMID: 34554329]
Level 3 (low-level) evidence
[18]
Edens C,Rodrigues BC,Lacerda MI,Dos Santos FC,De Jesús GR,De Jesús NR,Levy RA,Leatherwood C,Mandel J,Bermas B, Challenging cases in rheumatic pregnancies. Rheumatology (Oxford, England). 2018 Jul 1
[PubMed PMID: 30137591]
Level 3 (low-level) evidence
[20]
Goktay AY, Senturk C. Endovascular Treatment of Thrombosis and Embolism. Advances in experimental medicine and biology. 2017:906():195-213
[PubMed PMID: 27664152]
Level 3 (low-level) evidence
[21]
Zhang W,Liu X,Cheng H,Yang Z,Zhang G, Risk factors and treatment of venous thromboembolism in perioperative patients with ovarian cancer in China. Medicine. 2018 Aug
[PubMed PMID: 30075594]
[22]
Hansen AT,Juul S,Knudsen UB,Hvas AM, Low risk of venous thromboembolism following early pregnancy loss in pregnancies conceived by IVF. Human reproduction (Oxford, England). 2018 Aug 20
[PubMed PMID: 30137318]
[23]
Sharif S,Eventov M,Kearon C,Parpia S,Li M,Jiang R,Sneath P,Fuentes CO,Marriott C,de Wit K, Comparison of the age-adjusted and clinical probability-adjusted D-dimer to exclude pulmonary embolism in the ED. The American journal of emergency medicine. 2019 May
[PubMed PMID: 30077494]
[24]
Delluc A,Le Mao R,Tromeur C,Chambry N,Rault-Nagel H,Bressollette L,Mottier D,Couturaud F,Lacut K, Incidence of upper-extremity deep vein thrombosis in western France: a community-based study. Haematologica. 2019 Jan
[PubMed PMID: 30076179]
[25]
Carroll BJ,Piazza G, Hypercoagulable states in arterial and venous thrombosis: When, how, and who to test? Vascular medicine (London, England). 2018 Aug
[PubMed PMID: 30045685]
[26]
Sun ML,Wang XH,Huang J,Wang J,Wang Y, [Comparative study on deep venous thrombosis onset in hospitalized patients with different underlying diseases]. Zhonghua nei ke za zhi. 2018 Jun 1
[PubMed PMID: 29925128]
Level 2 (mid-level) evidence
[29]
Rodriguez V, Thrombosis Complications in Pediatric Acute Lymphoblastic Leukemia: Risk Factors, Management, and Prevention: Is There Any Role for Pharmacologic Prophylaxis? Frontiers in pediatrics. 2022;
[PubMed PMID: 35359904]
[30]
Min SK,Kim YH,Joh JH,Kang JM,Park UJ,Kim HK,Chang JH,Park SJ,Kim JY,Bae JI,Choi SY,Kim CW,Park SI,Yim NY,Jeon YS,Yoon HK,Park KH, Diagnosis and Treatment of Lower Extremity Deep Vein Thrombosis: Korean Practice Guidelines. Vascular specialist international. 2016 Sep
[PubMed PMID: 27699156]
Level 1 (high-level) evidence
[31]
Lurie F, Passman M, Meisner M, Dalsing M, Masuda E, Welch H, Bush RL, Blebea J, Carpentier PH, De Maeseneer M, Gasparis A, Labropoulos N, Marston WA, Rafetto J, Santiago F, Shortell C, Uhl JF, Urbanek T, van Rij A, Eklof B, Gloviczki P, Kistner R, Lawrence P, Moneta G, Padberg F, Perrin M, Wakefield T. The 2020 update of the CEAP classification system and reporting standards. Journal of vascular surgery. Venous and lymphatic disorders. 2020 May:8(3):342-352. doi: 10.1016/j.jvsv.2019.12.075. Epub 2020 Feb 27
[PubMed PMID: 32113854]
[32]
Bawa H,Weick JW,Dirschl DR,Luu HH, Trends in Deep Vein Thrombosis Prophylaxis and Deep Vein Thrombosis Rates After Total Hip and Knee Arthroplasty. The Journal of the American Academy of Orthopaedic Surgeons. 2018 Aug 27
[PubMed PMID: 30153117]
[33]
Denny N,Musale S,Edlin H,Serracino-Inglott F,Thachil J, Chronic deep vein thrombosis. Acute medicine. 2018
[PubMed PMID: 30129947]
[34]
Rahaghi FN,Minhas JK,Heresi GA, Diagnosis of Deep Venous Thrombosis and Pulmonary Embolism: New Imaging Tools and Modalities. Clinics in chest medicine. 2018 Sep
[PubMed PMID: 30122174]
[36]
Lewis TC,Cortes J,Altshuler D,Papadopoulos J, Venous Thromboembolism Prophylaxis: A Narrative Review With a Focus on the High-Risk Critically Ill Patient. Journal of intensive care medicine. 2018 Aug 30
[PubMed PMID: 30165770]
Level 3 (low-level) evidence
[37]
Oh DK,Song JM,Park DW,Oh SY,Ryu JS,Lee J,Lee SD,Lee JS, The effect of a multidisciplinary team on the implementation rates of major diagnostic and therapeutic procedures of chronic thromboembolic pulmonary hypertension. Heart & lung : the journal of critical care. 2019 Jan
[PubMed PMID: 30115494]
[38]
Ten Cate V,Prins MH, Secondary prophylaxis decision-making in venous thromboembolism: interviews on clinical practice in thirteen countries. Research and practice in thrombosis and haemostasis. 2017 Jul
[PubMed PMID: 30046672]
Level 3 (low-level) evidence
[39]
Young AM,Marshall A,Thirlwall J,Chapman O,Lokare A,Hill C,Hale D,Dunn JA,Lyman GH,Hutchinson C,MacCallum P,Kakkar A,Hobbs FDR,Petrou S,Dale J,Poole CJ,Maraveyas A,Levine M, Comparison of an Oral Factor Xa Inhibitor With Low Molecular Weight Heparin in Patients With Cancer With Venous Thromboembolism: Results of a Randomized Trial (SELECT-D). Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 2018 Jul 10
[PubMed PMID: 29746227]
Level 1 (high-level) evidence
[40]
Boon GJAM,Van Dam LF,Klok FA,Huisman MV, Management and treatment of deep vein thrombosis in special populations. Expert review of hematology. 2018 Sep
[PubMed PMID: 30016119]
[41]
Ha NB,Regal RE, Anticoagulation in Patients With Cirrhosis: Caught Between a Rock-Liver and a Hard Place. The Annals of pharmacotherapy. 2016 May;
[PubMed PMID: 26861989]
[42]
Kruger PC,Eikelboom JW,Douketis JD,Hankey GJ, Deep vein thrombosis: update on diagnosis and management. The Medical journal of Australia. 2019 Jun
[PubMed PMID: 31155730]
[44]
Key NS, Khorana AA, Kuderer NM, Bohlke K, Lee AYY, Arcelus JI, Wong SL, Balaban EP, Flowers CR, Francis CW, Gates LE, Kakkar AK, Levine MN, Liebman HA, Tempero MA, Lyman GH, Falanga A. Venous Thromboembolism Prophylaxis and Treatment in Patients With Cancer: ASCO Clinical Practice Guideline Update. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 2020 Feb 10:38(5):496-520. doi: 10.1200/JCO.19.01461. Epub 2019 Aug 5
[PubMed PMID: 31381464]
Level 1 (high-level) evidence
[45]
Li YK,Guo CG,Cheung KS,Liu KSH,Leung WK, Interruption of Antithrombotic Therapies and Risk of Postcolonoscopy Thromboembolic Events: A Real-World Cohort Study. Clinical gastroenterology and hepatology : the official clinical practice journal of the American Gastroenterological Association. 2022 Sep 24;
[PubMed PMID: 36167228]
[46]
Cheung KS, Leung WK. Gastrointestinal bleeding in patients on novel oral anticoagulants: Risk, prevention and management. World journal of gastroenterology. 2017 Mar 21:23(11):1954-1963. doi: 10.3748/wjg.v23.i11.1954. Epub
[PubMed PMID: 28373761]
[47]
Nepal G,Kharel S,Bhagat R,Ka Shing Y,Ariel Coghlan M,Poudyal P,Ojha R,Sunder Shrestha G, Safety and efficacy of Direct Oral Anticoagulants in cerebral venous thrombosis: A meta-analysis. Acta neurologica Scandinavica. 2022 Jan
[PubMed PMID: 34287841]
Level 1 (high-level) evidence
[48]
Cohen AT,Hamilton M,Mitchell SA,Phatak H,Liu X,Bird A,Tushabe D,Batson S, Comparison of the Novel Oral Anticoagulants Apixaban, Dabigatran, Edoxaban, and Rivaroxaban in the Initial and Long-Term Treatment and Prevention of Venous Thromboembolism: Systematic Review and Network Meta-Analysis. PloS one. 2015
[PubMed PMID: 26716830]
Level 1 (high-level) evidence
[49]
Cohen AT,Hamilton M,Bird A,Mitchell SA,Li S,Horblyuk R,Batson S, Comparison of the Non-VKA Oral Anticoagulants Apixaban, Dabigatran, and Rivaroxaban in the Extended Treatment and Prevention of Venous Thromboembolism: Systematic Review and Network Meta-Analysis. PloS one. 2016;
[PubMed PMID: 27487187]
Level 1 (high-level) evidence
[50]
Rollins BM,Silva MA,Donovan JL,Kanaan AO, Evaluation of oral anticoagulants for the extended treatment of venous thromboembolism using a mixed-treatment comparison, meta-analytic approach. Clinical therapeutics. 2014 Oct 1
[PubMed PMID: 25092394]
[51]
Almutairi AR,Zhou L,Gellad WF,Lee JK,Slack MK,Martin JR,Lo-Ciganic WH, Effectiveness and Safety of Non-vitamin K Antagonist Oral Anticoagulants for Atrial Fibrillation and Venous Thromboembolism: A Systematic Review and Meta-analyses. Clinical therapeutics. 2017 Jul
[PubMed PMID: 28668628]
Level 1 (high-level) evidence
[52]
Root CW,Dudzinski DM,Zakhary B,Friedman OA,Sista AK,Horowitz JM, Multidisciplinary approach to the management of pulmonary embolism patients: the pulmonary embolism response team (PERT). Journal of multidisciplinary healthcare. 2018
[PubMed PMID: 29670358]
[53]
McCaughan GJB,Favaloro EJ,Pasalic L,Curnow J, Anticoagulation at the extremes of body weight: choices and dosing. Expert review of hematology. 2018 Oct
[PubMed PMID: 30148651]
[54]
Fallaha MA,Radha S,Patel S, Safety and efficacy of a new thromboprophylaxis regiment for total knee and total hip replacement: a retrospective cohort study in 265 patients. Patient safety in surgery. 2018
[PubMed PMID: 30123323]
Level 2 (mid-level) evidence