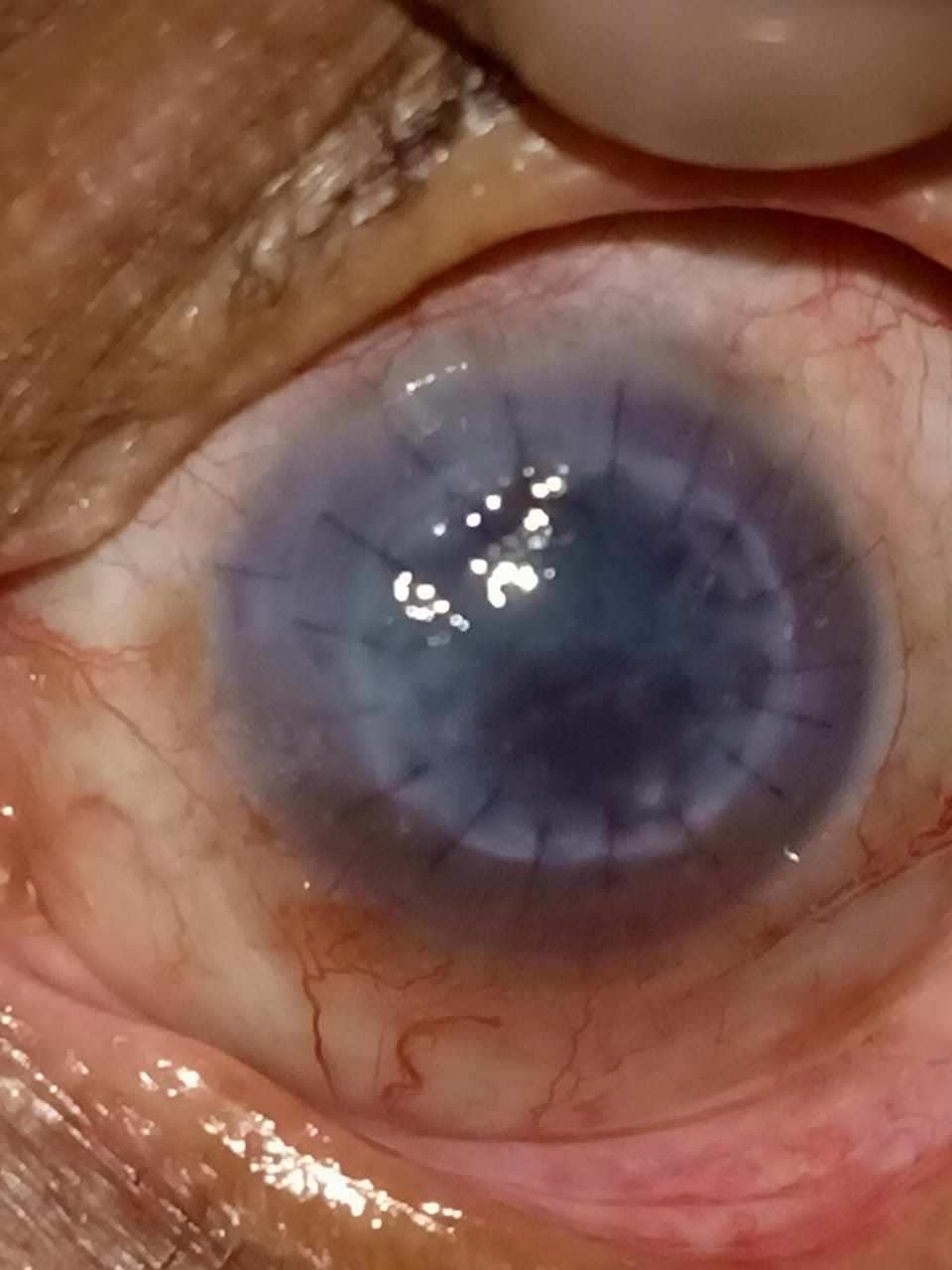
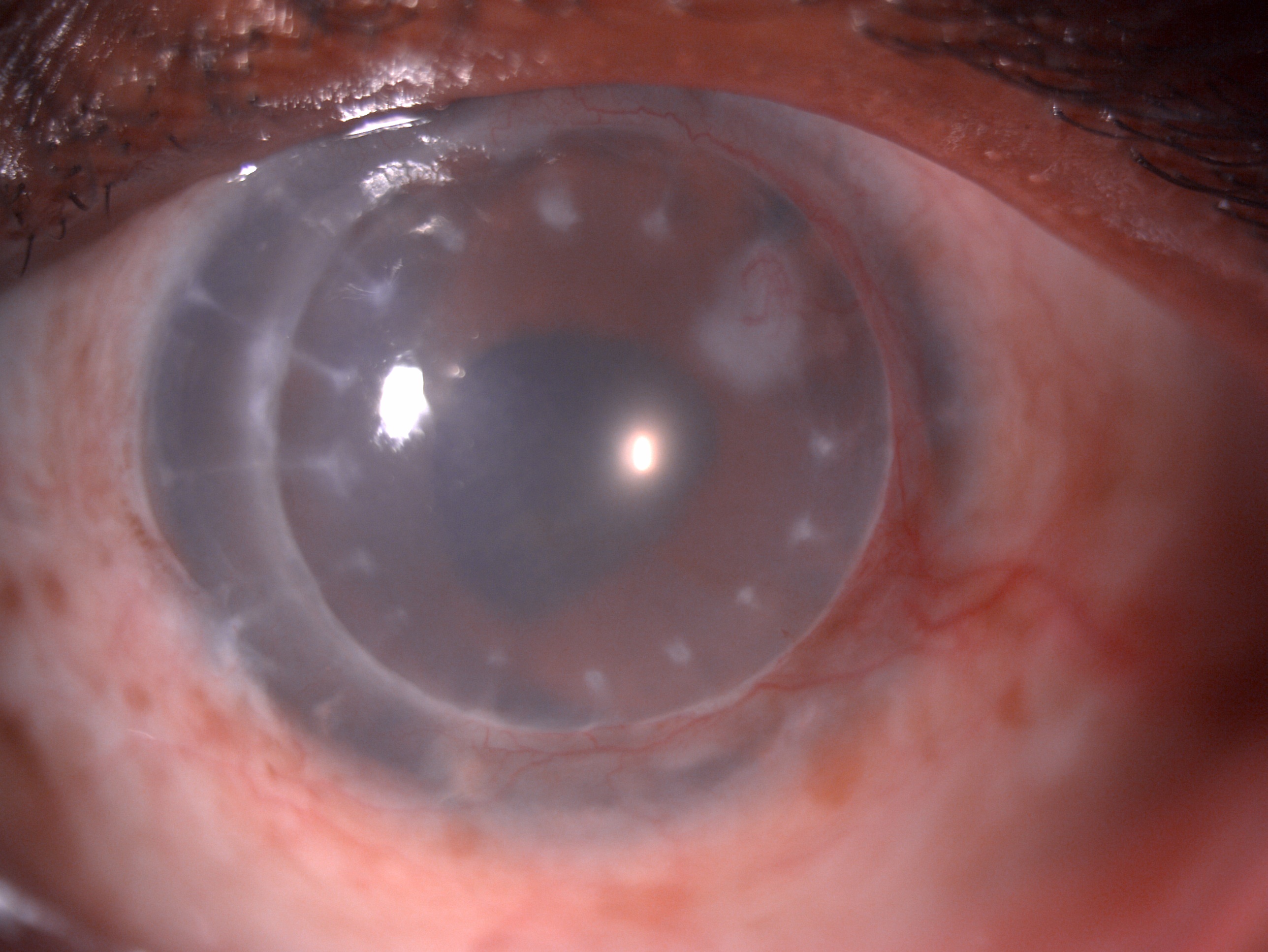
[1]
Niederkorn JY,Larkin DF, Immune privilege of corneal allografts. Ocular immunology and inflammation. 2010 Jun
[PubMed PMID: 20482389]
[3]
Catapano J,Fung SSM,Halliday W,Jobst C,Cheyne D,Ho ES,Zuker RM,Borschel GH,Ali A, Treatment of neurotrophic keratopathy with minimally invasive corneal neurotisation: long-term clinical outcomes and evidence of corneal reinnervation. The British journal of ophthalmology. 2019 Feb 15;
[PubMed PMID: 30770356]
Level 2 (mid-level) evidence
[4]
Hussen MS,Belete GT, Knowledge and Attitude toward Eye Donation among Adults, Northwest Ethiopia: A Community-based, Cross-sectional Study. Middle East African journal of ophthalmology. 2018 Jul-Dec;
[PubMed PMID: 30765949]
Level 2 (mid-level) evidence
[5]
Sharifi R,Yang Y,Adibnia Y,Dohlman CH,Chodosh J,Gonzalez-Andrades M, Finding an Optimal Corneal Xenograft Using Comparative Analysis of Corneal Matrix Proteins Across Species. Scientific reports. 2019 Feb 12;
[PubMed PMID: 30755666]
Level 2 (mid-level) evidence
[6]
Tanaka TS,Hood CT,Kriegel MF,Niziol L,Soong HK, Long-term outcomes of penetrating keratoplasty for corneal complications of herpes zoster ophthalmicus. The British journal of ophthalmology. 2019 Feb 7;
[PubMed PMID: 30733209]
[7]
Di Zazzo A,Kheirkhah A,Abud TB,Goyal S,Dana R, Management of high-risk corneal transplantation. Survey of ophthalmology. 2017 Nov - Dec
[PubMed PMID: 28012874]
Level 3 (low-level) evidence
[8]
Barraquer RI,Pareja-Aricò L,Gómez-Benlloch A,Michael R, Risk factors for graft failure after penetrating keratoplasty. Medicine. 2019 Apr;
[PubMed PMID: 31027083]
[9]
Gracitelli CPB,Ferrar PV,Pereira CAP,Hirai FE,Freitas D, A case of recurrent keratitis caused by Paecilomyces lilacinus and treated by voriconazole. Arquivos brasileiros de oftalmologia. 2019 Mar-Apr;
[PubMed PMID: 30726410]
Level 3 (low-level) evidence
[10]
Gadhvi KA,Romano V,Fernández-Vega Cueto L,Aiello F,Day AC,Allan BD, Deep anterior lamellar keratoplasty for keratoconus: multi-surgeon results. American journal of ophthalmology. 2019 Feb 2;
[PubMed PMID: 30721690]
[11]
Armitage WJ, Goodchild C, Griffin MD, Gunn DJ, Hjortdal J, Lohan P, Murphy CC, Pleyer U, Ritter T, Tole DM, Vabres B. High-risk Corneal Transplantation: Recent Developments and Future Possibilities. Transplantation. 2019 Dec:103(12):2468-2478. doi: 10.1097/TP.0000000000002938. Epub
[PubMed PMID: 31765363]
[12]
Jonas JB,Rank RM,Budde WM, Immunologic graft reactions after allogenic penetrating keratoplasty. American journal of ophthalmology. 2002 Apr
[PubMed PMID: 11931775]
[13]
Amouzegar A,Chauhan SK,Dana R, Alloimmunity and Tolerance in Corneal Transplantation. Journal of immunology (Baltimore, Md. : 1950). 2016 May 15
[PubMed PMID: 27183635]
[14]
Qazi Y,Hamrah P, Corneal Allograft Rejection: Immunopathogenesis to Therapeutics. Journal of clinical & cellular immunology. 2013 Nov 20
[PubMed PMID: 24634796]
[15]
Gain P, Jullienne R, He Z, Aldossary M, Acquart S, Cognasse F, Thuret G. Global Survey of Corneal Transplantation and Eye Banking. JAMA ophthalmology. 2016 Feb:134(2):167-73. doi: 10.1001/jamaophthalmol.2015.4776. Epub
[PubMed PMID: 26633035]
Level 3 (low-level) evidence
[16]
Liu J,Li L,Li X, Effectiveness of Cryopreserved Amniotic Membrane Transplantation in Corneal Ulceration: A Meta-Analysis. Cornea. 2019 Jan 29;
[PubMed PMID: 30702468]
Level 1 (high-level) evidence
[17]
Campbell JDM,Ahmad S,Agrawal A,Bienek C,Atkinson A,Mcgowan NWA,Kaye S,Mantry S,Ramaesh K,Glover A,Pelly J,MacRury C,MacDonald M,Hargreaves E,Barry J,Drain J,Cuthbertson B,Nerurkar L,Downing I,Fraser AR,Turner ML,Dhillon B, Allogeneic Ex Vivo Expanded Corneal Epithelial Stem Cell Transplantation: A Randomized Controlled Clinical Trial. Stem cells translational medicine. 2019 Jan 28;
[PubMed PMID: 30688407]
Level 1 (high-level) evidence
[18]
Lin PA,Tseng SH,Huang YH, Corneal Transplantation From Donors With Hepatitis B: Preliminary Results. The American journal of gastroenterology. 2019 Feb;
[PubMed PMID: 30676375]
[19]
Panda A, Vanathi M, Kumar A, Dash Y, Priya S. Corneal graft rejection. Survey of ophthalmology. 2007 Jul-Aug:52(4):375-96
[PubMed PMID: 17574064]
Level 3 (low-level) evidence
[20]
Alldredge OC,Krachmer JH, Clinical types of corneal transplant rejection. Their manifestations, frequency, preoperative correlates, and treatment. Archives of ophthalmology (Chicago, Ill. : 1960). 1981 Apr
[PubMed PMID: 7013739]
[21]
Krachmer JH,Alldredge OC, Subepithelial infiltrates: a probable sign of corneal transplant rejection. Archives of ophthalmology (Chicago, Ill. : 1960). 1978 Dec
[PubMed PMID: 363109]
[22]
Bartels MC,Otten HG,van Gelderen BE,Van der Lelij A, Influence of HLA-A, HLA-B, and HLA-DR matching on rejection of random corneal grafts using corneal tissue for retrospective DNA HLA typing. The British journal of ophthalmology. 2001 Nov
[PubMed PMID: 11673303]
Level 2 (mid-level) evidence
[23]
Watson SL,Tuft SJ,Dart JK, Patterns of rejection after deep lamellar keratoplasty. Ophthalmology. 2006 Apr;
[PubMed PMID: 16581417]
[24]
Büyüktepe TÇ,Yalçındağ N, Cytomegalovirus Endotheliitis After Penetrating Keratoplasty. Turkish journal of ophthalmology. 2020 Oct 30;
[PubMed PMID: 33342198]
[25]
Ho Wang Yin G,Sampo M,Soare S,Hoffart L, [Visual acuity, pachymetry and corneal density after 5% sodium chloride treatment in corneal edema after surgery]. Journal francais d'ophtalmologie. 2015 Dec
[PubMed PMID: 26547229]
[26]
Niederkorn JY,Callanan D,Ross JR, Prevention of the induction of allospecific cytotoxic T lymphocyte and delayed-type hypersensitivity responses by ultraviolet irradiation of corneal allografts. Transplantation. 1990 Aug
[PubMed PMID: 2382295]
[28]
Christo CG,van Rooij J,Geerards AJ,Remeijer L,Beekhuis WH, Suture-related complications following keratoplasty: a 5-year retrospective study. Cornea. 2001 Nov;
[PubMed PMID: 11685058]
Level 2 (mid-level) evidence
[29]
Zhai LY,Zhang XR,Liu H,Ma Y,Xu HC, Observation of topical tacrolimus on high-risk penetrating keratoplasty patients: a randomized clinical trial study. Eye (London, England). 2020 Sep
[PubMed PMID: 31784702]
Level 1 (high-level) evidence
[30]
Bafna RK,Kalra N,Asif MI,Lata S,Rathod A,Balaji A,Sharma N, Suturing large therapeutic corneal grafts based on donor size: A simplified technique for the novice corneal surgeon. European journal of ophthalmology. 2021 May;
[PubMed PMID: 33228422]
[31]
Thompson JM,Truong AH,Stern HD,Djalilian A,Cortina MS,Tu EY,Johnson P,Verdier DD,Rafol L,Lubeck D,Spektor T,Jorgensen C,Rubenstein JB,Majmudar PA,Talati R,Basti S,Feder R,Sugar A,Mian SI,Balasubramanian N,Sandhu J,Gaynes BI,Bouchard CS, A Multicenter Study Evaluating the Risk Factors and Outcomes of Repeat Descemet Stripping Endothelial Keratoplasty. Cornea. 2019 Feb
[PubMed PMID: 30615600]
Level 2 (mid-level) evidence
[32]
Dandona L,Naduvilath TJ,Janarthanan M,Rao GN, Causes of corneal graft failure in India. Indian journal of ophthalmology. 1998 Sep
[PubMed PMID: 10085627]
[33]
Khodadoust AA,Silverstein AM, Transplantation and rejection of individual cell layers of the cornea. Investigative ophthalmology. 1969 Apr;
[PubMed PMID: 4887869]
[35]
Peeler JS,Niederkorn JY, Effect of Langerhans' cells on cytotoxic T lymphocyte responses to major and minor alloantigens expressed on heterotopic corneal allografts. Transplantation proceedings. 1987 Feb;
[PubMed PMID: 3547821]
[36]
Boisjoly HM,Bernard PM,Dubé I,Laughrea PA,Bazin R,Bernier J, Effect of factors unrelated to tissue matching on corneal transplant endothelial rejection. American journal of ophthalmology. 1989 Jun 15
[PubMed PMID: 2658619]
[38]
D'Amaro J,Volker-Dieben HJ,Kruit PJ,de Lange P,Schipper R, Influence of pretransplant sensitization on the survival of corneal allografts. Transplantation proceedings. 1991 Feb;
[PubMed PMID: 1990555]
[39]
Brahma A,Ennis F,Harper R,Ridgway A,Tullo A, Visual function after penetrating keratoplasty for keratoconus: a prospective longitudinal evaluation. The British journal of ophthalmology. 2000 Jan
[PubMed PMID: 10611101]
[40]
Dada T,Aggarwal A,Minudath KB,Vanathi M,Choudhary S,Gupta V,Sihota R,Panda A, Post-penetrating keratoplasty glaucoma. Indian journal of ophthalmology. 2008 Jul-Aug;
[PubMed PMID: 18579984]
[41]
Tuft SJ,Gregory W, Long-term refraction and keratometry after penetrating keratoplasty for keratoconus. Cornea. 1995 Nov
[PubMed PMID: 8575185]
[42]
Kwon RO,Price MO,Price FW Jr,Ambrósio R Jr,Belin MW, Pentacam characterization of corneas with Fuchs dystrophy treated with Descemet membrane endothelial keratoplasty. Journal of refractive surgery (Thorofare, N.J. : 1995). 2010 Dec
[PubMed PMID: 20166622]
[43]
Morris E,Kirwan JF,Sujatha S,Rostron CK, Corneal endothelial specular microscopy following deep lamellar keratoplasty with lyophilised tissue. Eye (London, England). 1998;
[PubMed PMID: 9850251]
[44]
Lim LS,Aung HT,Aung T,Tan DT, Corneal imaging with anterior segment optical coherence tomography for lamellar keratoplasty procedures. American journal of ophthalmology. 2008 Jan
[PubMed PMID: 18028862]
[45]
Chen M, Miki M, Lin S, Yung Choi S. Sodium Fluorescein Staining of the Cornea for the Diagnosis of Dry Eye: A Comparison of Three Eye Solutions. Medical hypothesis, discovery & innovation ophthalmology journal. 2017 Winter:6(4):105-109
[PubMed PMID: 29560363]
[46]
Dessì G,Lahuerta EF,Puce FG,Mendoza LH,Stefanini T,Rosenberg I,Del Prato A,Perinetti M,Villa A, Role of B-scan ocular ultrasound as an adjuvant for the clinical assessment of eyeball diseases: a pictorial essay. Journal of ultrasound. 2015 Sep
[PubMed PMID: 26261467]
[47]
Acar BT,Muftuoglu O,Acar S, Comparison of macular thickness measured by optical coherence tomography after deep anterior lamellar keratoplasty and penetrating keratoplasty. American journal of ophthalmology. 2011 Nov;
[PubMed PMID: 21794841]
[48]
Hill JC,Ivey A, Corticosteroids in corneal graft rejection: double versus single pulse therapy. Cornea. 1994 Sep
[PubMed PMID: 7995059]
[49]
Hill JC,Maske R,Watson PG, The use of a single pulse of intravenous methylprednisolone in the treatment of corneal graft rejection. A preliminary report. Eye (London, England). 1991
[PubMed PMID: 1743357]
[50]
Nguyen NX,Seitz B,Martus P,Langenbucher A,Cursiefen C, Long-term topical steroid treatment improves graft survival following normal-risk penetrating keratoplasty. American journal of ophthalmology. 2007 Aug
[PubMed PMID: 17659972]
[51]
Hill JC,Maske R,Watson P, Corticosteroids in corneal graft rejection. Oral versus single pulse therapy. Ophthalmology. 1991 Mar;
[PubMed PMID: 2023754]
[52]
Young AL,Rao SK,Cheng LL,Wong AK,Leung AT,Lam DS, Combined intravenous pulse methylprednisolone and oral cyclosporine A in the treatment of corneal graft rejection: 5-year experience. Eye (London, England). 2002 May;
[PubMed PMID: 12032722]
[53]
Coster DJ,Williams KA, Immunosuppression for corneal transplantation and treatment of graft rejection. Transplantation proceedings. 1989 Feb
[PubMed PMID: 2650437]
[54]
Poon A,Constantinou M,Lamoureux E,Taylor HR, Topical Cyclosporin A in the treatment of acute graft rejection: a randomized controlled trial. Clinical & experimental ophthalmology. 2008 Jul
[PubMed PMID: 18939344]
Level 1 (high-level) evidence
[55]
Ziaei M,Ziaei F,Manzouri B, Systemic cyclosporine and corneal transplantation. International ophthalmology. 2016 Feb
[PubMed PMID: 26463642]
[56]
Rath T, Tacrolimus in transplant rejection. Expert opinion on pharmacotherapy. 2013 Jan;
[PubMed PMID: 23228138]
Level 3 (low-level) evidence
[57]
Dong Y,Huang YF,Wang LQ,Chen B, [Experimental study on the effects of rapamycin in prevention of rat corneal allograft rejection]. [Zhonghua yan ke za zhi] Chinese journal of ophthalmology. 2005 Oct;
[PubMed PMID: 16271181]
[58]
Diehl R,Ferrara F,Müller C,Dreyer AY,McLeod DD,Fricke S,Boltze J, Immunosuppression for in vivo research: state-of-the-art protocols and experimental approaches. Cellular & molecular immunology. 2017 Feb
[PubMed PMID: 27721455]
[59]
Szaflik JP,Major J,Izdebska J,Lao M,Szaflik J, Systemic immunosuppression with mycophenolate mofetil to prevent corneal graft rejection after high-risk penetrating keratoplasty: a 2-year follow-up study. Graefe's archive for clinical and experimental ophthalmology = Albrecht von Graefes Archiv fur klinische und experimentelle Ophthalmologie. 2016 Feb
[PubMed PMID: 26553197]
[60]
Ma D,Mellon J,Niederkorn JY, Oral immunisation as a strategy for enhancing corneal allograft survival. The British journal of ophthalmology. 1997 Sep
[PubMed PMID: 9422933]
[61]
Rocher N,Behar-Cohen F,Pournaras JA,Naud MC,Jeanny JC,Jonet L,Bourges JL, Effects of rat anti-VEGF antibody in a rat model of corneal graft rejection by topical and subconjunctival routes. Molecular vision. 2011 Jan 11;
[PubMed PMID: 21245949]
[62]
Schäcke H,Berger M,Rehwinkel H,Asadullah K, Selective glucocorticoid receptor agonists (SEGRAs): novel ligands with an improved therapeutic index. Molecular and cellular endocrinology. 2007 Sep 15
[PubMed PMID: 17630119]
[63]
Gurnani B, Kaur K, Tripathy K. Is there a genetic link between Keratoconus and Fuch's endothelial corneal dystrophy? Medical hypotheses. 2021 Dec:157():110699. doi: 10.1016/j.mehy.2021.110699. Epub 2021 Oct 9
[PubMed PMID: 34666260]
[65]
Gurnani B, Christy J, Narayana S, Rajkumar P, Kaur K, Gubert J. Retrospective multifactorial analysis of Pythium keratitis and review of literature. Indian journal of ophthalmology. 2021 May:69(5):1095-1101. doi: 10.4103/ijo.IJO_1808_20. Epub
[PubMed PMID: 33913840]
Level 2 (mid-level) evidence
[66]
Sano Y,Ksander BR,Streilein JW, Fate of orthotopic corneal allografts in eyes that cannot support anterior chamber-associated immune deviation induction. Investigative ophthalmology & visual science. 1995 Oct
[PubMed PMID: 7558710]
[67]
Bersudsky V,Blum-Hareuveni T,Rehany U,Rumelt S, The profile of repeated corneal transplantation. Ophthalmology. 2001 Mar
[PubMed PMID: 11237899]
[68]
Liu D, Ahmet A, Ward L, Krishnamoorthy P, Mandelcorn ED, Leigh R, Brown JP, Cohen A, Kim H. A practical guide to the monitoring and management of the complications of systemic corticosteroid therapy. Allergy, asthma, and clinical immunology : official journal of the Canadian Society of Allergy and Clinical Immunology. 2013 Aug 15:9(1):30. doi: 10.1186/1710-1492-9-30. Epub 2013 Aug 15
[PubMed PMID: 23947590]
[69]
Vora GK,Ciolino JB, Corneal allograft reaction associated with nonocular inflammation. Digital journal of ophthalmology : DJO. 2014
[PubMed PMID: 25097462]
[70]
Howard CA, Fernandez-Vina MA, Appelbaum FR, Confer DL, Devine SM, Horowitz MM, Mendizabal A, Laport GG, Pasquini MC, Spellman SR. Recommendations for donor human leukocyte antigen assessment and matching for allogeneic stem cell transplantation: consensus opinion of the Blood and Marrow Transplant Clinical Trials Network (BMT CTN). Biology of blood and marrow transplantation : journal of the American Society for Blood and Marrow Transplantation. 2015 Jan:21(1):4-7. doi: 10.1016/j.bbmt.2014.09.017. Epub 2014 Sep 30
[PubMed PMID: 25278457]
Level 3 (low-level) evidence
[71]
Sugar J, The Collaborative Corneal Transplantation Studies. Archives of ophthalmology (Chicago, Ill. : 1960). 1992 Nov
[PubMed PMID: 1444902]
[72]
Vail A,Gore SM,Bradley BA,Easty DL,Rogers CA,Armitage WJ, Conclusions of the corneal transplant follow up study. Collaborating Surgeons. The British journal of ophthalmology. 1997 Aug
[PubMed PMID: 9349147]
[73]
Munkhbat B,Hagihara M,Sato T,Tsuchida F,Sato K,Shimazaki J,Tsubota K,Tsuji K, Association between HLA-DPB1 matching and 1-year rejection-free graft survival in high-risk corneal transplantation. Transplantation. 1997 Apr 15
[PubMed PMID: 9112356]