Definition/Introduction
The cornea is a smooth, clear structure at the front of the eye. It functions to (1) shield the eye from foreign substances and (2) help control visual focus.[1][2] To focus light, the cornea must be clear; therefore, it has no blood vessels to impede light refraction. Tears and the aqueous humor of the eye nourish it (fluid in the anterior part of the eye between the cornea and the pupil and iris).[1]
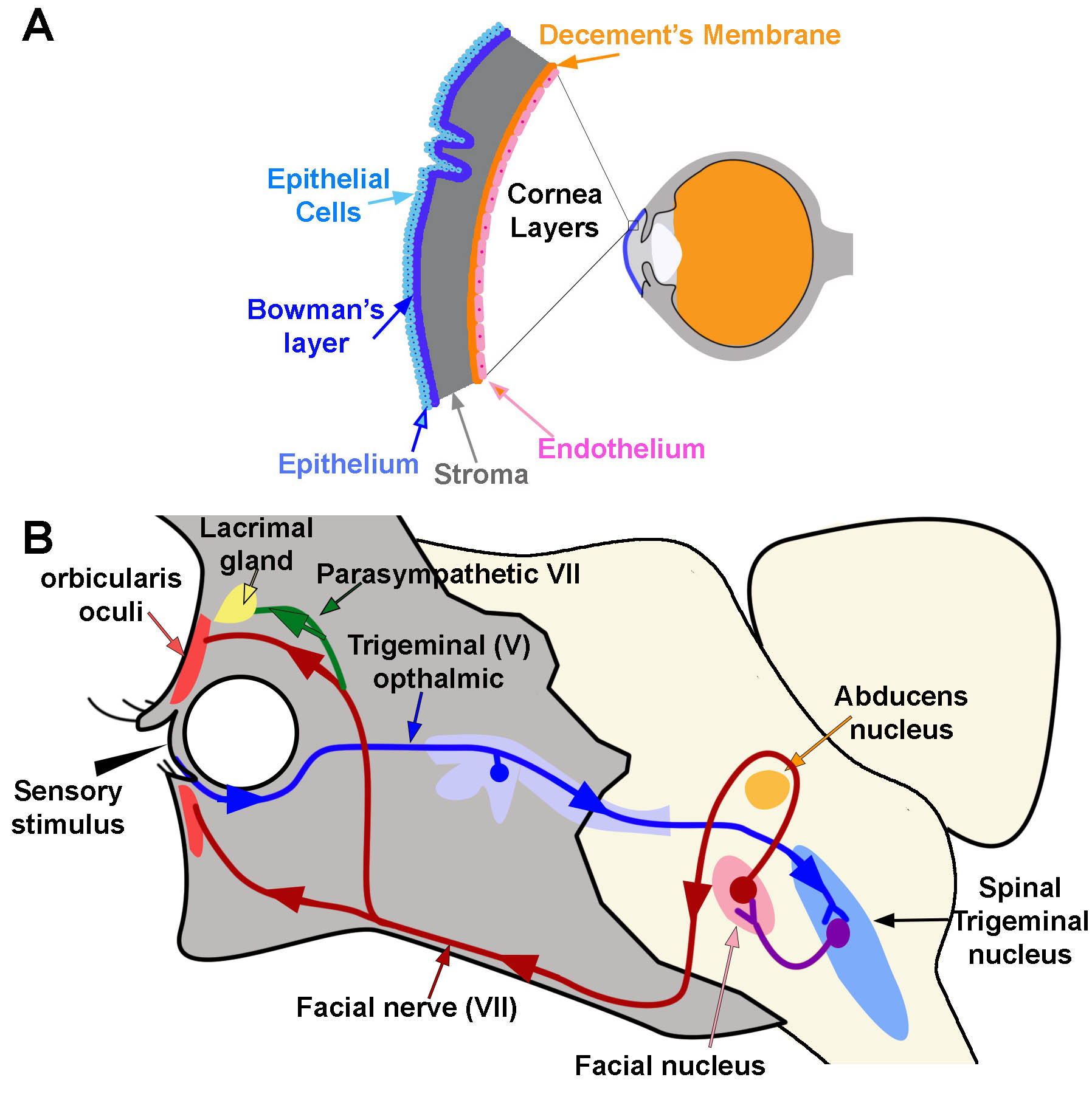
The cornea is composed of 5 layers: epithelium, Bowman’s layer, stroma, Descemet’s membrane, and endothelium (Figure 1A).[1] The epithelium is the outermost layer of the cornea (light blue, Figure 1A). It provides a smooth surface that absorbs both oxygen from the environment and nutrients from tears to supply the other layers of the cornea. It also functions as a barrier to foreign substances. Bowman’s layer (deep to the epithelium) is comprised of collagen to provide strength and structure. If injured, this layer can scar and impede vision (dark blue, Figure 1A). The stroma is the thickest layer of the cornea. Most of this layer is water with small amounts of collagen (gray, Figure 1A). Deep to the stroma, another strong layer of collagen, Descemet’s membrane, provides a secondary layer against injuries and infections. This layer is created by the deeper endothelial cells and can regenerate if injured (orange, Figure 1A). The deepest layer is the endothelium (pink, Figure 1A). The endothelium serves as a pump to remove excess fluid out of the stroma. The fluid typically leaks from inside the eye into the stroma. Without the endothelial pump, the stroma swells. Because endothelium cells do not regenerate if damaged, damage can cause blindness. The only therapy for endothelial damage is a corneal transplant.[1]
The cornea of the human eye is the most densely innervated part of the body, 300 to 600 times more sensitive than skin.[3] It receives sensory innervation from the long ciliary branches of the ophthalmic division of the trigeminal nerve (V).[2] Branches from these nerves form nerve plexuses running parallel to the surface of the cornea between the epithelium and Bowman’s layers.[4][5] Sensory nerve fibers within the cornea include myelinated A-beta and A-gamma nerves and unmyelinated C fibers.[6][2]
Corneal Reflex
The cornea is the first substance irritants or foreign objects will touch when they come in contact with the eye. Contact with the cornea initiates 2 reflexes: blink reflex (corneal reflex) and tear production. The corneal blink reflex is caused by a loop between the trigeminal sensory nerves and the facial motor (VII) nerve innervation of the orbicularis oculi muscles. The reflex activates when a sensory stimulus contacts either free nerve endings or mechanoreceptors within the epithelium of the cornea. Sensory information transmits through the ophthalmic division of the trigeminal nerve (V) to synapse within the spinal trigeminal nucleus in the brainstem (blue pathway, Figure 1B). The contacted nerve within the spinal trigeminal nucleus then projects to the facial nucleus and synapses with the facial nerve (purple pathway, Figure 1B). The facial nerve exits the facial nucleus, wraps around the abducens nucleus, and exits the skull at the stylomastoid foramen. After exiting the skull, the facial nerve travels medially over the surface of the face to activate the orbicularis oculi muscle. Contraction of this muscle causes a blink movement (i.e., eye closure; red pathway; Figure 1B).
There are 2 stages to the blink reflex, early and late. A-beta fibers activate the initial movement of the eyelid (early response) on the ipsilateral side. Other fiber types activate the late stage. The late-stage reflex stimulates facial nerves bilaterally so that both eyes blink. Input from the secondary motor system (e.g., interpositus nucleus of the cerebellum, red nucleus, and reticular activating system) can modulate this late stage reflex.[7][8][9][10][11][12][13][6]
The motor output of each blink can vary in non-pathological conditions. Many factors can influence the blink response. One factor is the duration and intensity of the sensory stimulus.[9] Secondary factors include a variety of brainstem and cortical circuits that synapse directly or indirectly with the trigeminal spinal and facial nuclei. These other circuits modulate the strength of blink activity based on conditioning.[14][15]
The blink response causes the eyelid to close. This system works together with a secondary circuit pathway through the oculomotor nucleus to re-open the eyelid. This secondary pathway is activated by the same touch pathway used by the blink response (Trigeminal V1). To facilitate this response, V1 nerves have secondary inputs within the trigeminal spinal nucleus onto nerves that project to the oculomotor nucleus. These spinal nucleus/oculomotor neurons synapse onto oculomotor neurons that then activate the levator palpebrae. The oculomotor pathway initiates the eyelid to re-open after it closes in the blink response. It works in conjunction with the facial motor pathway.[9]
Tear Production
Touching the cornea can also activate circuits that initiate tear production. This reflex circuit follows the same pathway as the blink reflex except that the parasympathetic nerves within the facial VII nerve innervates the lacrimal and meibomian “tarsal” glands instead of muscle (green circuit; Figure 1B).[16] The lacrimal gland produces tears. The Meibomian gland produces an oily film that helps to prevent evaporation of tears over the eye.[17] To reach these glands, parasympathetic fibers travel through the zygomatic branch of the facial VII nerve. They synapse in the pterygopalatine ganglion and then continue as the lacrimal nerve.
Tears have several functions.[16]
- Form smooth optical surface
- Keep cornea surface moist
- Wash away debris or irritants
- Have antibacterial properties that help to prevent infection
The corneal blink reflex is not the only reflex that can trigger tear production. Certain visual and taste reflexes can also stimulate these glands. Bright lights trigger visual reflexes. Specific chemicals that may be specifically noxious if they entered the eye (e.g., peppery, spicy, or hot foods) activate taste reflexes. Both systems contact parasympathetics that generate tear production. [16]