Continuing Education Activity
Alcoholic cardiomyopathy (ACM) is a cardiac disease caused by chronic alcohol consumption. It is characterized by ventricular dilation and impairment in cardiac function. ACM represents one of the leading causes of non-ischemic dilated cardiomyopathy. The major risk factor for developing ACM is chronic alcohol use; however, there is no cutoff value for the amount of alcohol consumption that would lead to the development of ACM. This activity describes the pathophysiology of ACM, its causes, presentation and the role of the interprofessional team in its management.
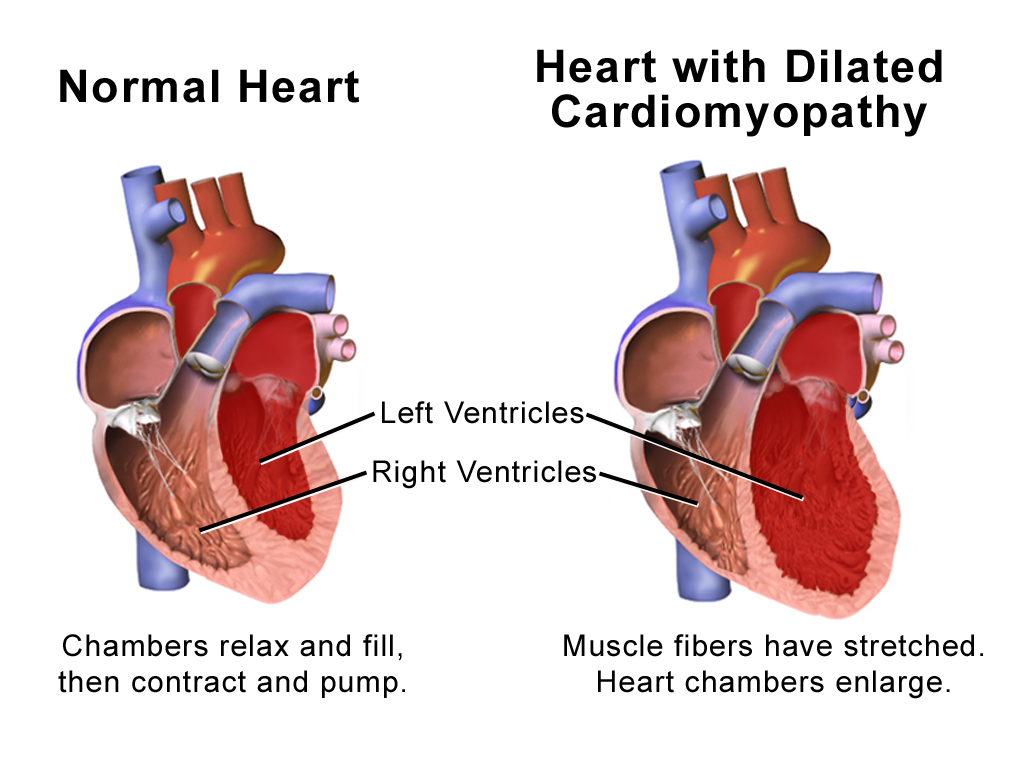
ACM is characterized by increased left ventricular mass, dilatation of the left ventricle, and heart failure (both systolic and diastolic). This activity examines when this condition should be considered on differential diagnosis. This activity highlights the role of the interprofessional team in caring for patients with this condition.
Objectives:
- Review the evaluation of a patient with ACM.
- Summarize the treatment options for ACM.
- Describe the pathophysiology of ACM.
- Explain the importance of improving care coordination among interprofessional team members to improve outcomes for patients affected by ACM.
Introduction
Alcohol-induced toxicity leads to non-ischemic dilated cardiomyopathy characterized by loss of contractile function and dilatation of myocardial ventricles. These findings are coupled with a clinical history of heavy alcohol use in the absence of coronary artery disease as a supportive etiology.[1]
Alcohol use is an important cause for non-ischemic cardiomyopathy and accounts for 10% of all cases of dilated cardiomyopathies.[2]
The major risk factor for developing ACM is chronic alcohol abuse; however, there is no specific cutoff value for the amount of alcohol consumption that would lead to the development of ACM.[3][4][5]
Etiology
Alcohol and its metabolites are cardiotoxic. Myocardial depression secondary to alcohol is initially reversible however prolonged sustained alcohol use leads to irreversible dysfunction.
Dilated cardiomyopathy secondary to alcohol use does not have a pre-defined exposure time. Daily alcohol consumption of 80 g per day or more for more than 5 years significantly increases the risk, however not all chronic alcohol users will develop Alcohol-induced cardiomyopathy.
There is neither a certain amount of alcohol known to be toxic to myocardial cells nor is there a specified period of exposure time to develop ACM. Alcohol consumption of 80 g per day for at least 5 years significantly increases the risk of developing ACM but, not all chronic alcohol abusers develop ACM.[6][7]
Drinking habits and echocardiogram evaluation were studied in (CARDIA) in 2368 adults in a long follow up. Increased alcohol intake had a direct inverse adverse association with ventricular structure. Increase alcohol consumption had greater indexed left ventricular mass and increased left ventricular end-diastolic volume after 20 years. Alcohol consumption did not show any association with systolic dysfunction. [8]
Epidemiology
Alcoholic cardiomyopathy is a leading cause of non-ischemic dilated cardiomyopathy in United States.
Incidence of alcoholic cardiomyopathy ranges from 1-2% of all heavy alcohol users. It is estimated, approximately 21-36% of all non-ischemic cardiomyopathies are attributed to alcohol. The prevalance of alcoholic cardiomyopathy in addiction units is estimated around 21-32 %. Overall data with regards to alcohol induced cardiomyopathy is insuffienct and does not illustrate significant available data.
Most common age population for ACM is males from age 30-55 with significant history of alcohol use for more than 10 years. Females constitute roughly 14 % of cases of alcohol induced cardiomyopathy however lifetime exposure required for women to develop alcohol induced cardiomyopathy is less compared to men.
Mortality rates are higher for males compared to females and more in blacks compared to white population[9][10]
Interactions between genetic and non-genetic factors, exposure to other cardiotoxic substances, or mineral deficiencies such as thiamine play an important role in determining progression of alcoholic cardiomyopathy
Pathophysiology
Alcohol-induced cardiotoxicity can be characterized by acute and chronic.
Acute can be defined as large volume acute consumption of alcohol promotes myocardial inflammation leading to increased troponin concentration in serum, tachyarrhythmias including atrial fibrillation and rarely ventricular fibrillation.
Chronic alcohol consumption can cause multi-organ damage including myocardial dysfunction. There are no specific targeted histological or immunological biomarkers for the diagnosis of alcohol-induced cardiomyopathy. The key diagnostic element is the absence of coronary artery disease. Various pathophysiological mechanisms have been postulated in the development of cardiomyopathy however one key factor undergoing active research is the role of genetic mutation and susceptibility to develop cardiomyopathy.
The postulated mechanism includes mitochondria damage, oxidative stress injury, apoptosis, modification of actin and myosin structure, and alteration of calcium homeostasis. Alcohol consumption causes an increase in mitochondrial fragmentation. Studies have shown an increase in reactive oxygen species (ROS) level in myocytes following alcohol consumption and thus causes oxidation of lipids, proteins, and DNA leading to cardiac dysfunction. These changes are related to both direct alcohol toxicity on cardiac cells and the indirect toxicity of major alcohol metabolites such as acetaldehyde.
The pathogenesis of ACM also involves interaction between genetically related factors, such as HLA subtypes or the alcohol dehydrogenase enzyme allele as well as non-genetic factors including thiamine deficiency and exposure of various substances that are directly toxic to cardiac cells. These structural and intracellular alterations cause activation in compensatory mechanisms in response to cardiac dysfunction such as the renin-angiotensin-aldosterone system, increase sympathetic signal firing and an increase in brain natriuretic peptide release. These mechanisms are responsible for an increase in preload, left ventricular dilation, and decreased cardiac output which all contribute to the clinical manifestation of ACM.[11][12]
A study by Ware et al. evaluated cardiomyopathy associated genetics in the pathophysiology of dilated cardiomyopathy and determined titin truncating variants (TTNtv) gene to be prevalent in the genetic disposition of dilated cardiomyopathy with worse left ventricular ejection fraction.[13]
Other deficiencies including nutritional such as thiamine or other toxic materials ingested may lead to additional concomitant complications.
Histopathology
There are no specific histological changes in cardiomyocytes. Alcohol may induce changes at the cellular level.
Certain microscopic features may suggest damage secondary to alcohol causing cardiomyopathy. Commonly seen cellular structural alterations include changes in the mitochondrial reticulum, cluster formation of mitochondria and disappearance of inter-mitochondrial junctions. In later stages, minor mitochondria and septic mitochondria can be seen.
Enzymatic activity changes which are seen in the idiopathic cardiomyopathy including decreased activity of oxygen reduction mitochondrial enzymes, increased fatty acid uptake and increased lysosomal/microsomal enzyme activity can be seen.
History and Physical
Alcoholic cardiomyopathy can present with signs and symptoms of congestive heart failure. Patients may present with dilated cardiomyopathy with systolic dysfunction. Symptoms include gradual onset worsening shortness of breath, orthopnea/paroxysmal nocturnal dyspnea. Palpitations and syncopal episodes can occur due to tachyarrhythmias seen in alcoholic cardiomyopathy.
Diastolic dysfunction is the earliest sign of ACM and is usually seen in approximately 30% of patients with a history of chronic alcohol abuse with no evidence of systolic dysfunction nor left ventricle hypertrophy.
The most important factor includes a significant history of chronic alcohol use
On physical examination, patients present with non-specific signs of congestive heart failure such as anorexia, generalized cachexia, muscular atrophy, weakness, peripheral edema, third spacing, hepatomegaly, and jugular venous distention. ACM can also cause tachyarrhythmia (most common is atrial fibrillation). Apical impulse can be displaced causing a down and out apex. S3 gallop sound along with apical pansystolic murmur due to mitral regurgitation is often heard.
Other etiologies associated with chronic alcohol use may be associated with it and patients may also show signs of liver disease, folate deficiency, increase the risk for bleeding, malnutrition, peripheral neuropathy, and neurological conditions such as Wernicke-Korsakoff syndrome.
Evaluation
The diagnosis of alcoholic cardiomyopathy is non-specific.
The key to diagnosis is a personal history of chronic heavy alcohol use and the absence of other etiologies.
In patients exhibiting chronic alcohol use, other causes of dilated cardiomyopathy need workup. Investigative work up such as mean corpuscular volume (MCV), gamma-glutamyl-transpeptidase (GGT), elevated transaminases (AST, ALT) and elevated INR usually are seen in liver injury can be helpful as supportive evidence of alcohol use.[14][15].
Other supportive workups can include:
Electrocardiography:
Many changes can be observed including premature atrial or ventricular contractions, supraventricular tachycardias, atrioventricular blocks, bundle branch blocks, QT prolongation, non-specific ST and T wave changes and abnormal Q waves.
Chest X-Ray:
Enlarged cardiac silhouette, pulmonary vascular congestion, and pleural effusion
Alcoholic cardiomyopathy can be diagnosed by the following criteria
- Dilated cardiomyopathy is seen on 2D echocardiography
- LV end-diastolic dimension greater than 2 standard deviations above normal.Left ventricle ejection fraction less than 50%.Exclusion of hypertensive, valvular and ischemic heart diseases. Hence patients with suspected alcoholic cardiomyopathy should undergo coronary angiogram along with echocardiography.
Treatment / Management
Treatment for alcoholic cardiomyopathy is directed towards source control.
The mainstay for treatment is complete abstinence from alcohol. Management should include providing patient resources and counseling.
Symptomatic management for patients with secondary heart failure and manage associated complications is warranted.
Some studies have suggested that even moderation of alcohol consumption similar outcomes as compared to abstinence.
Pharmacologic therapy should include goal-directed heart failure therapy as used in idiopathic dilated cardiomyopathy with reduced ejection fraction. This includes a combination of beta-blockers, an angiotensin-converting enzyme inhibitor, diuretics, aldosterone receptor antagonist and angiotensin blocker-neprilysin inhibitor (if LVEF is less than or equal to 40%). The use of carvedilol, trimetazidine with other conventional heart failure drugs have been proven to be beneficial in some studies.
Some promising new treatments are targeting the pathways that are involved in the pathogenesis of ACM such as myocyte hypertrophy, cell necrosis and fibrosis, and oxidative stress; however, these are still under investigation.[16][17]
Differential Diagnosis
- Constrictive cardiomyopathy
- Hypertrophic cardiomyopathy
- Idiopathic dilated cardiomyopathy
- Cirrhotic cardiomyopathy
- Takatsubo cardiomyopathy
- Drug-induced cardiomyopathy
- Beriberi
Prognosis
Long-term survival is directly associated with both the amount of alcohol use and the duration of this usage.
Alcohol-induced dilated cardiomyopathy has a better prognosis than ischemia-induced cardiomyopathy. Atrial fibrillation, QRS widening of > 120 ms and absence of beta-blockers are associated with poor outcomes.
Patients who continue to drink have a poor prognosis. Data suggests patients with successful quitting of alcohol have improved overall outcomes with a reduced number of inpatient admissions and improvement in diameter size on echocardiogram.
Complications for those who continue to drink alcohol may include progressive heart failure, arrhythmias, and cardioembolic phenomenon. Data reveal that depending on the alcohol consumed, mortality rates of 40-80% are seen within 10 years. [5][18]
Complications
- Heart failure
- Cachexia
- Arrhythmias
- Cardioembolism
- Death
Enhancing Healthcare Team Outcomes
Alcoholic cardiomyopathy is best managed with an interprofessional approach with the involvement of primary care physician and cardiology.
The mainstay of management is providing support, resources including but not limited to alcoholic anonymous and encouragement for alcohol abstinence and address underlying stressors if any which requires assistance from nursing staff and pharmacy. These patients may also benefit from a dietary consult to assess nutrition.