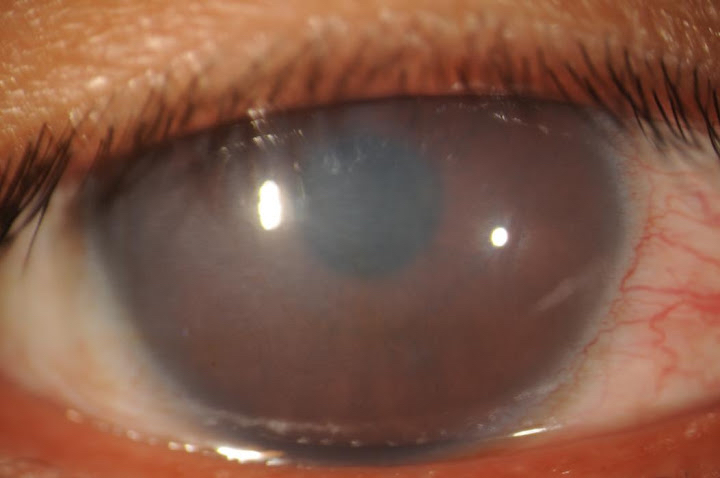
[1]
Patel SP, Parker MD. SLC4A11 and the Pathophysiology of Congenital Hereditary Endothelial Dystrophy. BioMed research international. 2015:2015():475392. doi: 10.1155/2015/475392. Epub 2015 Sep 16
[PubMed PMID: 26451371]
[2]
Yang F, Hong J, Xiao G, Feng Y, Peng R, Wang M, Qu H. Descemet Stripping Endothelial Keratoplasty in Pediatric Patients with Congenital Hereditary Endothelial Dystrophy. American journal of ophthalmology. 2020 Jan:209():132-140. doi: 10.1016/j.ajo.2019.08.010. Epub 2019 Aug 26
[PubMed PMID: 31465754]
[3]
Desir J, Abramowicz M. Congenital hereditary endothelial dystrophy with progressive sensorineural deafness (Harboyan syndrome). Orphanet journal of rare diseases. 2008 Oct 15:3():28. doi: 10.1186/1750-1172-3-28. Epub 2008 Oct 15
[PubMed PMID: 18922146]
[4]
Ashar JN, Ramappa M, Vaddavalli PK. Paired-eye comparison of Descemet's stripping endothelial keratoplasty and penetrating keratoplasty in children with congenital hereditary endothelial dystrophy. The British journal of ophthalmology. 2013 Oct:97(10):1247-9. doi: 10.1136/bjophthalmol-2012-302602. Epub 2013 Apr 23
[PubMed PMID: 23613513]
[5]
Schmedt T, Silva MM, Ziaei A, Jurkunas U. Molecular bases of corneal endothelial dystrophies. Experimental eye research. 2012 Feb:95(1):24-34. doi: 10.1016/j.exer.2011.08.002. Epub 2011 Aug 10
[PubMed PMID: 21855542]
[6]
Aldave AJ, Han J, Frausto RF. Genetics of the corneal endothelial dystrophies: an evidence-based review. Clinical genetics. 2013 Aug:84(2):109-19. doi: 10.1111/cge.12191. Epub 2013 Jun 10
[PubMed PMID: 23662738]
[7]
Paliwal P, Sharma A, Tandon R, Sharma N, Titiyal JS, Sen S, Nag TC, Vajpayee RB. Congenital hereditary endothelial dystrophy - mutation analysis of SLC4A11 and genotype-phenotype correlation in a North Indian patient cohort. Molecular vision. 2010 Dec 31:16():2955-63
[PubMed PMID: 21203343]
[8]
Lopez IA, Rosenblatt MI, Kim C, Galbraith GC, Jones SM, Kao L, Newman D, Liu W, Yeh S, Pushkin A, Abuladze N, Kurtz I. Slc4a11 gene disruption in mice: cellular targets of sensorineuronal abnormalities. The Journal of biological chemistry. 2009 Sep 25:284(39):26882-96. doi: 10.1074/jbc.M109.008102. Epub 2009 Jul 8
[PubMed PMID: 19586905]
[9]
Jalimarada SS,Ogando DG,Vithana EN,Bonanno JA, Ion transport function of SLC4A11 in corneal endothelium. Investigative ophthalmology
[PubMed PMID: 23745003]
[10]
Chaurasia S, Ramappa M, Annapurna M, Kannabiran C. Coexistence of Congenital Hereditary Endothelial Dystrophy and Fuchs Endothelial Corneal Dystrophy Associated With SLC4A11 Mutations in Affected Families. Cornea. 2020 Mar:39(3):354-357. doi: 10.1097/ICO.0000000000002183. Epub
[PubMed PMID: 31714402]
[11]
Alka K, Casey JR. Molecular phenotype of SLC4A11 missense mutants: Setting the stage for personalized medicine in corneal dystrophies. Human mutation. 2018 May:39(5):676-690. doi: 10.1002/humu.23401. Epub 2018 Feb 2
[PubMed PMID: 29327391]
Level 3 (low-level) evidence
[12]
Siddiqui S, Zenteno JC, Rice A, Chacón-Camacho O, Naylor SG, Rivera-de la Parra D, Spokes DM, James N, Toomes C, Inglehearn CF, Ali M. Congenital hereditary endothelial dystrophy caused by SLC4A11 mutations progresses to Harboyan syndrome. Cornea. 2014 Mar:33(3):247-51. doi: 10.1097/ICO.0000000000000041. Epub
[PubMed PMID: 24351571]
[13]
Moazzeni H,Javadi MA,Asgari D,Khani M,Emami M,Moghadam A,Panahi-Bazaz MR,Hosseini Tehrani M,Karimian F,Hosseini B,Nekuie Moghadam T,Hassanpour H,Akbari MT,Elahi E, Observation of nine previously reported and 10 non-reported {i}SLC4A11{/i} mutations among 20 Iranian CHED probands and identification of an {i}MPDZ{/i} mutation as possible cause of CHED and FECD in one family. The British journal of ophthalmology. 2019 Aug 16;
[PubMed PMID: 31420327]
[14]
Sultana A, Garg P, Ramamurthy B, Vemuganti GK, Kannabiran C. Mutational spectrum of the SLC4A11 gene in autosomal recessive congenital hereditary endothelial dystrophy. Molecular vision. 2007 Jul 26:13():1327-32
[PubMed PMID: 17679935]
[15]
Kumawat BL, Gupta R, Sharma A, Sen S, Gupta S, Tandon R. Delayed onset of congenital hereditary endothelial dystrophy due to compound heterozygous SLC4A11 mutations. Indian journal of ophthalmology. 2016 Jul:64(7):492-5. doi: 10.4103/0301-4738.190100. Epub
[PubMed PMID: 27609159]
[16]
Bermejo E, Martínez-Frías ML. Congenital eye malformations: clinical-epidemiological analysis of 1,124,654 consecutive births in Spain. American journal of medical genetics. 1998 Feb 17:75(5):497-504
[PubMed PMID: 9489793]
Level 2 (mid-level) evidence
[17]
Hemadevi B,Veitia RA,Srinivasan M,Arunkumar J,Prajna NV,Lesaffre C,Sundaresan P, Identification of mutations in the SLC4A11 gene in patients with recessive congenital hereditary endothelial dystrophy. Archives of ophthalmology (Chicago, Ill. : 1960). 2008 May;
[PubMed PMID: 18474783]
[18]
AlArrayedh H, Collum L, Murphy CC. Outcomes of penetrating keratoplasty in congenital hereditary endothelial dystrophy. The British journal of ophthalmology. 2018 Jan:102(1):19-25. doi: 10.1136/bjophthalmol-2016-309565. Epub 2017 May 6
[PubMed PMID: 28478395]
[19]
Mohamed MD, McKibbin M, Jafri H, Rasheed Y, Woods CG, Inglehearn CF. A new pedigree with recessive mapping to CHED2 locus on 20p13. The British journal of ophthalmology. 2001 Jun:85(6):758-9
[PubMed PMID: 11439918]
[20]
Alka K, Casey JR. Ophthalmic Nonsteroidal Anti-Inflammatory Drugs as a Therapy for Corneal Dystrophies Caused by SLC4A11 Mutation. Investigative ophthalmology & visual science. 2018 Aug 1:59(10):4258-4267. doi: 10.1167/iovs.18-24301. Epub
[PubMed PMID: 30140924]
Level 3 (low-level) evidence
[21]
Khan AO,Aldahmesh MA,Alkuraya F, Congenital hereditary endothelial dystrophy, not glaucoma, in a child with iris colobomas. Journal of AAPOS : the official publication of the American Association for Pediatric Ophthalmology and Strabismus. 2016 Aug;
[PubMed PMID: 27373217]
[22]
Judisch GF, Maumenee IH. Clinical differentiation of recessive congenital hereditary endothelial dystrophy and dominant hereditary endothelial dystrophy. American journal of ophthalmology. 1978 May:85(5 Pt 1):606-12
[PubMed PMID: 306759]
[23]
Nischal KK. Genetics of Congenital Corneal Opacification--Impact on Diagnosis and Treatment. Cornea. 2015 Oct:34 Suppl 10():S24-34. doi: 10.1097/ICO.0000000000000552. Epub
[PubMed PMID: 26352876]
[24]
Ehlers N, Módis L, Møller-Pedersen T. A morphological and functional study of Congenital Hereditary Endothelial Dystrophy. Acta ophthalmologica Scandinavica. 1998 Jun:76(3):314-8
[PubMed PMID: 9686844]
[25]
Khan AO,Al-Shehah A,Ghadhfan FE, High measured intraocular pressure in children with recessive congenital hereditary endothelial dystrophy. Journal of pediatric ophthalmology and strabismus. 2010 Jan-Feb;
[PubMed PMID: 20128551]
[26]
Al-Ghamdi A, Al-Rajhi A, Wagoner MD. Primary pediatric keratoplasty: indications, graft survival, and visual outcome. Journal of AAPOS : the official publication of the American Association for Pediatric Ophthalmology and Strabismus. 2007 Feb:11(1):41-7
[PubMed PMID: 17307682]
[27]
Ashar JN, Madhavi Latha K, Vaddavalli PK. Descemet's stripping endothelial keratoplasty (DSEK) for children with congenital hereditary endothelial dystrophy: surgical challenges and 1-year outcomes. Graefe's archive for clinical and experimental ophthalmology = Albrecht von Graefes Archiv fur klinische und experimentelle Ophthalmologie. 2012 Sep:250(9):1341-5. doi: 10.1007/s00417-012-2014-8. Epub 2012 Apr 19
[PubMed PMID: 22527319]
[28]
Hermina Strungaru M, Ali A, Rootman D, Mireskandari K. Endothelial keratoplasty for posterior polymorphous corneal dystrophy in a 4-month-old infant. American journal of ophthalmology case reports. 2017 Sep:7():23-26. doi: 10.1016/j.ajoc.2017.05.001. Epub 2017 May 4
[PubMed PMID: 29260073]
Level 3 (low-level) evidence
[29]
Gonnermann J, Klamann MK, Maier AK, Bertelmann E, Schroeter J, von Au K, Joussen AM, Torun N. Descemet membrane endothelial keratoplasty in a child with corneal endothelial dysfunction in Kearns-Sayre syndrome. Cornea. 2014 Nov:33(11):1232-4. doi: 10.1097/ICO.0000000000000252. Epub
[PubMed PMID: 25211357]
[30]
Huang PT. Penetrating keratoplasty in infants and children. Journal of AAPOS : the official publication of the American Association for Pediatric Ophthalmology and Strabismus. 2007 Feb:11(1):5-6
[PubMed PMID: 17307675]
[31]
Gulias-Cañizo R, Gonzalez-Salinas R, Hernandez-Zimbron LF, Hernandez-Quintela E, Sanchez-Huerta V. Indications and outcomes of pediatric keratoplasty in a tertiary eye care center: A retrospective review. Medicine. 2017 Nov:96(45):e8587. doi: 10.1097/MD.0000000000008587. Epub
[PubMed PMID: 29137083]
Level 2 (mid-level) evidence
[32]
Busin M, Beltz J, Scorcia V. Descemet-stripping automated endothelial keratoplasty for congenital hereditary endothelial dystrophy. Archives of ophthalmology (Chicago, Ill. : 1960). 2011 Sep:129(9):1140-6. doi: 10.1001/archophthalmol.2011.114. Epub 2011 May 9
[PubMed PMID: 21555597]
[33]
Kaur M, Titiyal JS, Gagrani M, Shaikh F, Agarwal T, Sinha R, Sharma N. Repeat keratoplasty in failed Descemet stripping automated endothelial keratoplasty. Indian journal of ophthalmology. 2019 Oct:67(10):1586-1592. doi: 10.4103/ijo.IJO_1729_18. Epub
[PubMed PMID: 31546486]
[34]
Mittal V, Mittal R. Challenges in pediatric endothelial keratoplasty. Indian journal of ophthalmology. 2014 Feb:62(2):251-4. doi: 10.4103/0301-4738.128638. Epub
[PubMed PMID: 24618494]
[35]
Maier P, Reinhard T, Cursiefen C. Descemet stripping endothelial keratoplasty--rapid recovery of visual acuity. Deutsches Arzteblatt international. 2013 May:110(21):365-71. doi: 10.3238/arztebl.2013.0365. Epub 2013 May 24
[PubMed PMID: 23795211]
[36]
Melles GR, Ong TS, Ververs B, van der Wees J. Descemet membrane endothelial keratoplasty (DMEK). Cornea. 2006 Sep:25(8):987-90
[PubMed PMID: 17102683]
[37]
Anshu A, Price MO, Price FW Jr. Risk of corneal transplant rejection significantly reduced with Descemet's membrane endothelial keratoplasty. Ophthalmology. 2012 Mar:119(3):536-40. doi: 10.1016/j.ophtha.2011.09.019. Epub 2012 Jan 3
[PubMed PMID: 22218143]
[38]
Chiu AM, Mandziuk JJ, Loganathan SK, Alka K, Casey JR. High Throughput Assay Identifies Glafenine as a Corrector for the Folding Defect in Corneal Dystrophy-Causing Mutants of SLC4A11. Investigative ophthalmology & visual science. 2015 Dec:56(13):7739-53. doi: 10.1167/iovs.15-17802. Epub
[PubMed PMID: 26641551]
Level 3 (low-level) evidence
[39]
Miao S, Lin Q, Liu Y, Song YW, Zhang YN, Pan ZQ. Clinicopathologic Features and Treatment Characteristics of Congenital Corneal Opacity Infants and Children Aged 3 Years or Less: A Retrospective Single Institution Analysis. Medical principles and practice : international journal of the Kuwait University, Health Science Centre. 2020:29(1):18-24. doi: 10.1159/000501763. Epub 2019 Jun 28
[PubMed PMID: 31247621]
Level 2 (mid-level) evidence
[40]
Nischal KK. A new approach to the classification of neonatal corneal opacities. Current opinion in ophthalmology. 2012 Sep:23(5):344-54. doi: 10.1097/ICU.0b013e328356893d. Epub
[PubMed PMID: 22871880]
Level 3 (low-level) evidence
[41]
Kurji K, Damji K. Ophthaproblem. Can you identify this condition? Primary congenital glaucoma. Canadian family physician Medecin de famille canadien. 2012 Apr:58(4):409, 412-3
[PubMed PMID: 22499817]
[42]
Tomatsu S, Pitz S, Hampel U. Ophthalmological Findings in Mucopolysaccharidoses. Journal of clinical medicine. 2019 Sep 14:8(9):. doi: 10.3390/jcm8091467. Epub 2019 Sep 14
[PubMed PMID: 31540112]
[43]
Bose S, Yeo DCM, Wijetilleka S. Using two smartphones to look for corneal cystine crystals. Digital journal of ophthalmology : DJO. 2019 Jan:25(1):12-15. doi: 10.5693/djo.02.2019.02.003. Epub 2019 Mar 29
[PubMed PMID: 31080371]
[44]
Bhandari R, Ferri S, Whittaker B, Liu M, Lazzaro DR. Peters anomaly: review of the literature. Cornea. 2011 Aug:30(8):939-44. doi: 10.1097/ICO.0b013e31820156a9. Epub
[PubMed PMID: 21448066]
[45]
Davidson AE, Liskova P, Evans CJ, Dudakova L, Nosková L, Pontikos N, Hartmannová H, Hodaňová K, Stránecký V, Kozmík Z, Levis HJ, Idigo N, Sasai N, Maher GJ, Bellingham J, Veli N, Ebenezer ND, Cheetham ME, Daniels JT, Thaung CM, Jirsova K, Plagnol V, Filipec M, Kmoch S, Tuft SJ, Hardcastle AJ. Autosomal-Dominant Corneal Endothelial Dystrophies CHED1 and PPCD1 Are Allelic Disorders Caused by Non-coding Mutations in the Promoter of OVOL2. American journal of human genetics. 2016 Jan 7:98(1):75-89. doi: 10.1016/j.ajhg.2015.11.018. Epub 2015 Dec 31
[PubMed PMID: 26749309]
[46]
Panahi-Bazaz M, Sharifipour F, Malekahmadi M. Modified Descemet's Stripping Automated Endothelial Keratoplasty for Congenital Hereditary Endothelial Dystrophy. Journal of ophthalmic & vision research. 2014 Oct-Dec:9(4):522-5. doi: 10.4103/2008-322X.150836. Epub
[PubMed PMID: 25709783]
[47]
Mohebbi M, Nabavi A, Fadakar K, Hashemi H. Outcomes of Descemet-Stripping Automated Endothelial Keratoplasty in Congenital Hereditary Endothelial Dystrophy. Eye & contact lens. 2020 Jan:46(1):57-62. doi: 10.1097/ICL.0000000000000604. Epub
[PubMed PMID: 31008826]