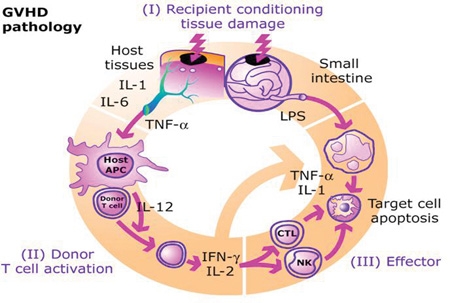
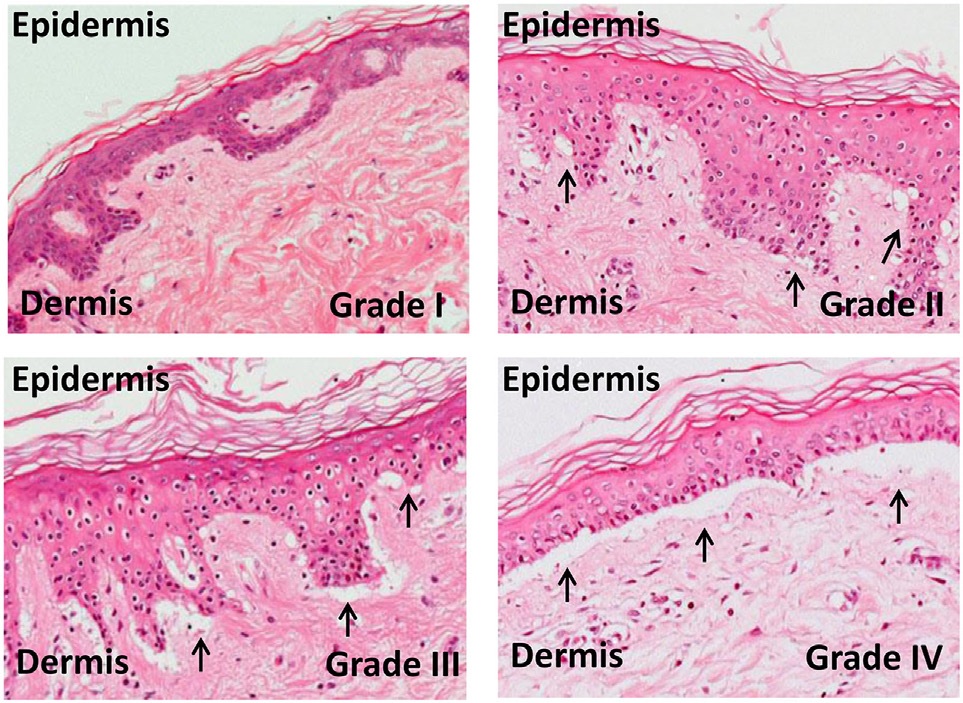
[1]
Socié G, Ritz J. Current issues in chronic graft-versus-host disease. Blood. 2014 Jul 17:124(3):374-84. doi: 10.1182/blood-2014-01-514752. Epub 2014 Jun 9
[PubMed PMID: 24914139]
[2]
Antin JH, Kim HT, Cutler C, Ho VT, Lee SJ, Miklos DB, Hochberg EP, Wu CJ, Alyea EP, Soiffer RJ. Sirolimus, tacrolimus, and low-dose methotrexate for graft-versus-host disease prophylaxis in mismatched related donor or unrelated donor transplantation. Blood. 2003 Sep 1:102(5):1601-5
[PubMed PMID: 12730113]
[3]
Shimabukuro-Vornhagen A, Liebig T, von Bergwelt-Baildon M. Statins inhibit human APC function: implications for the treatment of GVHD. Blood. 2008 Aug 15:112(4):1544-5. doi: 10.1182/blood-2008-04-149609. Epub
[PubMed PMID: 18684883]
[4]
Mielcarek M, Martin PJ, Leisenring W, Flowers ME, Maloney DG, Sandmaier BM, Maris MB, Storb R. Graft-versus-host disease after nonmyeloablative versus conventional hematopoietic stem cell transplantation. Blood. 2003 Jul 15:102(2):756-62
[PubMed PMID: 12663454]
[5]
Ferrara JL, Levine JE, Reddy P, Holler E. Graft-versus-host disease. Lancet (London, England). 2009 May 2:373(9674):1550-61. doi: 10.1016/S0140-6736(09)60237-3. Epub 2009 Mar 11
[PubMed PMID: 19282026]
[6]
Jamil MO, Mineishi S. State-of-the-art acute and chronic GVHD treatment. International journal of hematology. 2015 May:101(5):452-66. doi: 10.1007/s12185-015-1785-1. Epub 2015 Apr 12
[PubMed PMID: 25864189]
[7]
Atkinson K, Horowitz MM, Gale RP, van Bekkum DW, Gluckman E, Good RA, Jacobsen N, Kolb HJ, Rimm AA, Ringdén O. Risk factors for chronic graft-versus-host disease after HLA-identical sibling bone marrow transplantation. Blood. 1990 Jun 15:75(12):2459-64
[PubMed PMID: 2350582]
[8]
MacMillan ML, Robin M, Harris AC, DeFor TE, Martin PJ, Alousi A, Ho VT, Bolaños-Meade J, Ferrara JL, Jones R, Arora M, Blazar BR, Holtan SG, Jacobsohn D, Pasquini M, Socie G, Antin JH, Levine JE, Weisdorf DJ. A refined risk score for acute graft-versus-host disease that predicts response to initial therapy, survival, and transplant-related mortality. Biology of blood and marrow transplantation : journal of the American Society for Blood and Marrow Transplantation. 2015 Apr:21(4):761-7. doi: 10.1016/j.bbmt.2015.01.001. Epub 2015 Jan 10
[PubMed PMID: 25585275]
[9]
Gale RP, Bortin MM, van Bekkum DW, Biggs JC, Dicke KA, Gluckman E, Good RA, Hoffmann RG, Kay HE, Kersey JH. Risk factors for acute graft-versus-host disease. British journal of haematology. 1987 Dec:67(4):397-406
[PubMed PMID: 3322360]
[10]
Nassereddine S, Rafei H, Elbahesh E, Tabbara I. Acute Graft Versus Host Disease: A Comprehensive Review. Anticancer research. 2017 Apr:37(4):1547-1555
[PubMed PMID: 28373413]
Level 3 (low-level) evidence
[11]
Salomao M, Dorritie K, Mapara MY, Sepulveda A. Histopathology of Graft-vs-Host Disease of Gastrointestinal Tract and Liver: An Update. American journal of clinical pathology. 2016 May:145(5):591-603. doi: 10.1093/ajcp/aqw050. Epub
[PubMed PMID: 27247365]
[12]
Ghimire S, Weber D, Mavin E, Wang XN, Dickinson AM, Holler E. Pathophysiology of GvHD and Other HSCT-Related Major Complications. Frontiers in immunology. 2017:8():79. doi: 10.3389/fimmu.2017.00079. Epub 2017 Mar 20
[PubMed PMID: 28373870]
[13]
Lee SJ, Vogelsang G, Flowers ME. Chronic graft-versus-host disease. Biology of blood and marrow transplantation : journal of the American Society for Blood and Marrow Transplantation. 2003 Apr:9(4):215-33
[PubMed PMID: 12720215]
[14]
Elad S, Zadik Y, Zeevi I, Miyazaki A, de Figueiredo MA, Or R. Oral cancer in patients after hematopoietic stem-cell transplantation: long-term follow-up suggests an increased risk for recurrence. Transplantation. 2010 Dec 15:90(11):1243-4. doi: 10.1097/TP.0b013e3181f9caaa. Epub
[PubMed PMID: 21119507]
[15]
Gálvez K, Muñoz P, Vera V, Arce C. Vitiligo type cutaneous manifestation of chronic graft-versus-host disease. Case report. Revista chilena de pediatria. 2018 Feb:89(1):113-117. doi: 10.4067/S0370-41062018000100113. Epub
[PubMed PMID: 29664513]
Level 3 (low-level) evidence
[16]
Gandelman JS, Zic J, Dewan AK, Lee SJ, Flowers M, Cutler C, Pidala J, Chen H, Jagasia MH, Tkaczyk ER. The Anatomic Distribution of Skin Involvement in Patients with Incident Chronic Graft-versus-Host Disease. Biology of blood and marrow transplantation : journal of the American Society for Blood and Marrow Transplantation. 2019 Feb:25(2):279-286. doi: 10.1016/j.bbmt.2018.09.007. Epub 2018 Sep 13
[PubMed PMID: 30219700]
[17]
Munir SZ, Aylward J. A Review of Ocular Graft-Versus-Host Disease. Optometry and vision science : official publication of the American Academy of Optometry. 2017 May:94(5):545-555. doi: 10.1097/OPX.0000000000001071. Epub
[PubMed PMID: 28422804]
[19]
Firoz BF, Lee SJ, Nghiem P, Qureshi AA. Role of skin biopsy to confirm suspected acute graft-vs-host disease: results of decision analysis. Archives of dermatology. 2006 Feb:142(2):175-82
[PubMed PMID: 16490845]
[20]
Vander Lugt MT, Braun TM, Hanash S, Ritz J, Ho VT, Antin JH, Zhang Q, Wong CH, Wang H, Chin A, Gomez A, Harris AC, Levine JE, Choi SW, Couriel D, Reddy P, Ferrara JL, Paczesny S. ST2 as a marker for risk of therapy-resistant graft-versus-host disease and death. The New England journal of medicine. 2013 Aug 8:369(6):529-39. doi: 10.1056/NEJMoa1213299. Epub
[PubMed PMID: 23924003]
[21]
Ferrara JL, Harris AC, Greenson JK, Braun TM, Holler E, Teshima T, Levine JE, Choi SW, Huber E, Landfried K, Akashi K, Vander Lugt M, Reddy P, Chin A, Zhang Q, Hanash S, Paczesny S. Regenerating islet-derived 3-alpha is a biomarker of gastrointestinal graft-versus-host disease. Blood. 2011 Dec 15:118(25):6702-8. doi: 10.1182/blood-2011-08-375006. Epub 2011 Oct 6
[PubMed PMID: 21979939]
[22]
Korngold R, Marini JC, de Baca ME, Murphy GF, Giles-Komar J. Role of tumor necrosis factor-alpha in graft-versus-host disease and graft-versus-leukemia responses. Biology of blood and marrow transplantation : journal of the American Society for Blood and Marrow Transplantation. 2003 May:9(5):292-303
[PubMed PMID: 12766879]
[23]
Matsukuma KE, Wei D, Sun K, Ramsamooj R, Chen M. Diagnosis and differential diagnosis of hepatic graft versus host disease (GVHD). Journal of gastrointestinal oncology. 2016 Apr:7(Suppl 1):S21-31. doi: 10.3978/j.issn.2078-6891.2015.036. Epub
[PubMed PMID: 27034810]
[24]
Naymagon S, Naymagon L, Wong SY, Ko HM, Renteria A, Levine J, Colombel JF, Ferrara J. Acute graft-versus-host disease of the gut: considerations for the gastroenterologist. Nature reviews. Gastroenterology & hepatology. 2017 Dec:14(12):711-726. doi: 10.1038/nrgastro.2017.126. Epub 2017 Sep 27
[PubMed PMID: 28951581]
[25]
Glucksberg H, Storb R, Fefer A, Buckner CD, Neiman PE, Clift RA, Lerner KG, Thomas ED. Clinical manifestations of graft-versus-host disease in human recipients of marrow from HL-A-matched sibling donors. Transplantation. 1974 Oct:18(4):295-304
[PubMed PMID: 4153799]
[26]
Rowlings PA, Przepiorka D, Klein JP, Gale RP, Passweg JR, Henslee-Downey PJ, Cahn JY, Calderwood S, Gratwohl A, Socié G, Abecasis MM, Sobocinski KA, Zhang MJ, Horowitz MM. IBMTR Severity Index for grading acute graft-versus-host disease: retrospective comparison with Glucksberg grade. British journal of haematology. 1997 Jun:97(4):855-64
[PubMed PMID: 9217189]
Level 2 (mid-level) evidence
[27]
Cho C, Hsu M, Barba P, Maloy MA, Avecilla ST, Barker JN, Castro-Malaspina H, Giralt SA, Jakubowski AA, Koehne G, Meagher RC, O'Reilly RJ, Papadopoulos EB, Ponce DM, Tamari R, van den Brink MRM, Young JW, Devlin SM, Perales MA. Long-term prognosis for 1-year relapse-free survivors of CD34+ cell-selected allogeneic hematopoietic stem cell transplantation: a landmark analysis. Bone marrow transplantation. 2017 Dec:52(12):1629-1636. doi: 10.1038/bmt.2017.197. Epub 2017 Oct 9
[PubMed PMID: 28991247]
[28]
Cahn JY, Klein JP, Lee SJ, Milpied N, Blaise D, Antin JH, Leblond V, Ifrah N, Jouet JP, Loberiza F, Ringden O, Barrett AJ, Horowitz MM, Socié G, Société Française de Greffe de Moëlle et Thérapie Cellulaire, Dana Farber Cancer Institute, International Bone Marrow Transplant Registry. Prospective evaluation of 2 acute graft-versus-host (GVHD) grading systems: a joint Société Française de Greffe de Moëlle et Thérapie Cellulaire (SFGM-TC), Dana Farber Cancer Institute (DFCI), and International Bone Marrow Transplant Registry (IBMTR) prospective study. Blood. 2005 Aug 15:106(4):1495-500
[PubMed PMID: 15878974]
[29]
Müller JA, Zirafi O, Roan NR, Lee SJ, Münch J. Evaluation of EPI-X4 as a urinary peptide biomarker for diagnosis and prognosis of late acute GvHD. Bone marrow transplantation. 2016 Aug:51(8):1137-9. doi: 10.1038/bmt.2016.65. Epub 2016 Apr 4
[PubMed PMID: 27042833]
[30]
Bergeron A, Cheng GS. Bronchiolitis Obliterans Syndrome and Other Late Pulmonary Complications After Allogeneic Hematopoietic Stem Cell Transplantation. Clinics in chest medicine. 2017 Dec:38(4):607-621. doi: 10.1016/j.ccm.2017.07.003. Epub 2017 Sep 19
[PubMed PMID: 29128013]
[31]
van der Meij BS, de Graaf P, Wierdsma NJ, Langius JA, Janssen JJ, van Leeuwen PA, Visser OJ. Nutritional support in patients with GVHD of the digestive tract: state of the art. Bone marrow transplantation. 2013 Apr:48(4):474-82. doi: 10.1038/bmt.2012.124. Epub 2012 Jul 9
[PubMed PMID: 22773121]
[32]
Mays JW, Fassil H, Edwards DA, Pavletic SZ, Bassim CW. Oral chronic graft-versus-host disease: current pathogenesis, therapy, and research. Oral diseases. 2013 May:19(4):327-46. doi: 10.1111/odi.12028. Epub 2012 Oct 28
[PubMed PMID: 23107104]
[33]
Elhage A, Sligar C, Cuthbertson P, Watson D, Sluyter R. Insights into mechanisms of graft-versus-host disease through humanised mouse models. Bioscience reports. 2022 Sep 30:42(9):. doi: 10.1042/BSR20211986. Epub
[PubMed PMID: 35993192]
[34]
Várady G,Cserepes J,Németh A,Szabó E,Sarkadi B, Cell surface membrane proteins as personalized biomarkers: where we stand and where we are headed. Biomarkers in medicine. 2013 Oct;
[PubMed PMID: 24044572]