Introduction
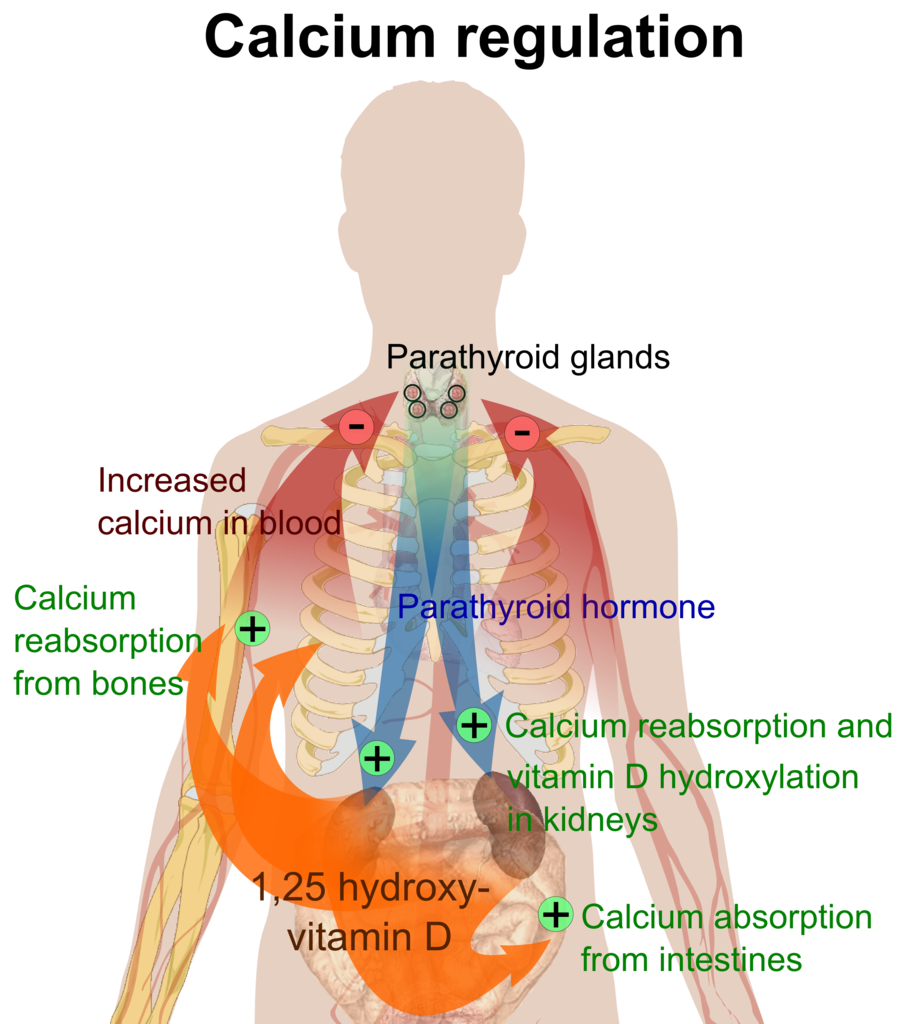
The parathyroid is comprised of 4 small glands embedded in the posterior aspect of the thyroid gland. Its main function is the production and secretion of parathyroid hormone (PTH), a polypeptide hormone responsible for maintaining serum calcium homeostasis. The levels of PTH and serum calcium are inversely proportional. At low serum calcium levels, PTH, in conjunction with vitamin D, works at many sites in the body to mobilize calcium stores and increase calcium absorption and reabsorption. Both calcium and vitamin D provide negative feedback to the parathyroid glands; as calcium and vitamin D levels increase, they bind receptors at the parathyroid glands and inhibit the production and release of PTH.[1][2][3]
Cellular Level
The parathyroid glands contain two main cell types: chief cells and oxyphil cells.[4][5]
Chief Cells
The chief cells are the functional cells of the parathyroid gland, responsible for both synthesizing and secreting parathyroid hormone. Regulation of PTH production and release is dependent on serum calcium levels. The G protein-coupled transmembrane receptor, calcium-sensing receptor (CaSR), on the surface of chief cells responds to low serum calcium and activates translation and secretion of PTH.
Oxyphil Cells
Also known as oxyntic cells, these cells have no recognized endocrine function. The proportion of oxyphil cells in the parathyroid glands increases with age.
Development
The 4 parathyroid glands develop from the endoderm of the 3 and 4 pharyngeal pouches around the 6 weeks of gestation. Recent studies have posited that there may be some ectodermal and neural crest contribution to the glands’ formation as well. Interestingly, the inferior parathyroid glands develop from the 3 pharyngeal pouches, while the superior glands develop from the 4 pharyngeal pouches. This is due to the additional formation of the thymus by these pharyngeal pouches that migrate caudally later in gestation. The parathyroid glands are functional during gestation, acting to control calcium balance in the fetus.
Function
The parathyroid glands’ function is to maintain serum calcium homeostasis through the synthesis and release of PTH. At the bone, PTH inhibits osteoblast activity and stimulates osteoclast activity leading to bone breakdown and calcium release. PTH increases calcium reabsorption at the kidneys and blocks phosphate reabsorption from the tubules. PTH also acts on the kidneys to stimulate the formation of vitamin D. Vitamin D is an essential component of calcium and phosphate homeostasis, yielding its effects on the kidneys and gastrointestinal (GI) system.
Skeletal System
PTH works at the cellular level by indirectly stimulating osteoclasts to break down bone. PTH binds to cell receptors on osteoblasts stimulating the release of Receptor Activator of Nuclear factor Kappa-B Ligand (RANKL). RANKL binds to its receptor on osteoclast precursors, stimulating them to fuse into mature osteoclasts, thereby increasing calcium resorption from bone.
Renal System
PTH has 2 main roles in the kidney. It works in the thick ascending loop of Henle, distal convoluted tubule (DCT), and collecting ducts to increase reabsorption of calcium by up-regulating TRPV5, a calcium transporter on the tubular epithelium. PTH also binds to sites in the proximal tubule that block phosphate reabsorption. Therefore, the net effect of PTH is to decrease calcium excretion and increase phosphate excretion in the urine. Additionally, PTH works at the proximal tubule of the kidneys to up-regulate the translation of alpha-1-hydroxylase, the enzyme responsible for generating the biologically active form of vitamin D (1,25-dihydroxy-vitamin D or calcitriol). Vitamin D binds to receptors in the bone that function similarly to PTH, increasing serum calcium. In the kidney, vitamin D increases both calcium and phosphate reabsorption.
Gastrointestinal System
Calcium absorption occurs in the small intestines, with 70% to 80% absorbed in the ileum. Although PTH has no direct effects on the small intestine, the downstream effects of PTH on vitamin D synthesis occur at this level. Vitamin D increases calcium and phosphate absorption from the gut. Vitamin D receptors occur along the entirety of the gut epithelium, and studies have shown that both paracellular and transcellular absorption of calcium increase when stimulated. Vitamin D increases transcription and translation of calcium transport proteins in the epithelium, including TRPV6, calbindin, and ATP-dependent calcium pumps.
Related Testing
Total Serum Calcium
Total serum calcium is the amount of calcium in the blood, including free or ionized calcium, complexed calcium (calcium bound to bicarbonate or citrate), and protein-bound calcium. Normal levels range from 8.5 to 10.2 mg/dL. This value is affected by the amount of protein, usually albumin, available for binding and must be corrected in hypoalbuminemia. For every 1 gm decrease in serum albumin, we expect the total serum calcium to decrease by approximately 0.8 mg. The official equation is:
Corrected Calcium = [Ca + 0.02 x (40 - Albumin)]
Ionized Serum Calcium
Ionized calcium, or free calcium, is the biologically active form of calcium necessary for many functions, including cell signaling, neurotransmission, and muscle contraction. Normal levels range from 4.64 to 5.28 mg/dL. Blood pH can affect ionized calcium levels by changing the binding affinity of albumin for positively charged anions. At low pH, an abundance of hydrogen ions effectively competes with free calcium for binding sites on negatively charged albumin, thus increasing free calcium in the serum.
Parathyroid Hormone
PTH levels can be measured through immunoassays of blood samples. Normal levels range from 10 to 65 pg/mL.
Parathyroid-related peptide (PTHrP)
PTHrP is a peptide produced by nearly all tissues in the body at very low levels. The physiologic function of PTHrP is unknown, but it appears to serve mostly autocrine and paracrine functions, accounting for its low plasma level. Normal levels are expected to be below 2.0 pmol/L. PTHrP is overproduced in a wide variety of carcinomas but is most notably associated with squamous cell lung cancer, squamous cell cancers of the head and neck, and breast cancer. Levels of PTHrP greater than the reference range warrant a thorough search for related cancers.
Sestamibi Parathyroid Scintigraphy
A sestamibi parathyroid scan is ordered in patients who are believed to have parathyroid adenomas or hyperplasia. The radioisotope technetium-99m is injected intravenously and taken up by active oxyphil cells in the parathyroid tissue. A gamma camera is used to take images of the neck and will show the locations and sizes of the parathyroid glands. Repeat images are taken after a washout period of 2 to 4 hours, and glands still showing activity are considered hyperactive. Studies have shown a variable sensitivity of this test ranging from 39% to 90%.
Vitamin D
Most assays measuring vitamin D test the biologically inactive form, 25-(OH)-Vitamin D, as it is the major form of vitamin D in circulation. The active metabolite, 1,25-OH-vitamin D, has a circulating half-life of 4 to 6 hours, making it difficult to interpret accurately. Vitamin D deficiency is defined by a 25-(OH)-Vitamin D level less than 20 ng/mL and insufficiency as a level between 21 to 29 ng/mL. Normal vitamin D levels range from 30 to 100 ng/mL.
Clinical Significance
Hyperparathyroidism
Hyperparathyroidism can lead to dangerously elevated levels of calcium in the blood. The expression “moans, stones, bones, and psychic overtones” is used to describe the symptoms that are commonly seen with hypercalcemia, including nausea, vomiting, constipation, kidney stones, bone pain, and psychosis or altered mental status.[6][7]
It is important to consider the etiology of hyperparathyroidism when forming your differential diagnosis. Primary hyperparathyroidism is a result of overproduction and release of PTH. This can be a result of a parathyroid adenoma, hyperplasia, or carcinoma. In these instances, a single gland or multiple parathyroid glands function above the physiological set point due to a primary disease. PTH and serum calcium will be elevated, while serum phosphate will be reduced.
Secondary hyperparathyroidism is a physiologic response to a chronic hypocalcemic state. This can be caused by chronic kidney disease (CKD) or vitamin D deficiency. In CKD, the damaged kidneys fail to produce enough 1-alpha-hydroxylase, so they are unable to convert vitamin D into its active form. Additionally, the kidneys are unable to excrete phosphate, resulting in higher blood phosphate levels properly. Phosphate acts as a positive stimulus at the parathyroid glands, increasing PTH release. PTH and phosphate levels will be high when tested, while calcium and 1,25(OH)-vitamin D levels will be low.
Tertiary hyperparathyroidism results from uncorrected, long-term hyperparathyroidism, usually seen in end-stage renal disease. The chronic low calcium state in these patients leads to hyperplasia of the parathyroid glands. The parathyroid glands cannot respond to serum calcium levels and instead act autonomously, producing and secreting PTH. PTH levels will be extremely high, while calcium and phosphate will also be elevated.
In patients presenting with acute hypercalcemia, it is important first to correct the electrolyte disturbance as it can be life-threatening. Fluid administration is first-line treatment, and the addition of calcium-lowering medications such as diuretics, bisphosphonates or calcitonin can be discussed. Once the patient is stabilized, physicians should look for the etiology and treat the underlying disease.
Hypoparathyroidism
Decreased function of the parathyroid glands, called hypoparathyroidism, can lead to low serum calcium levels. As calcium is necessary for muscle and nerve functions, the main symptoms of hypocalcemia deal with these 2 systems.[8][8]
Patients commonly report perioral numbness or tingling, but it can also be felt in the hands and feet. Weakness, paresthesias, muscle cramps or tetany, and seizures can all result from severe hypocalcemia. On physical exam of the hypocalcemic patient, physicians can elicit tetany in 2 ways. Tapping on the facial nerve (the area anterior to the tragus) will elicit spasms of the facial muscles on the ipsilateral side, known as Chvostek’s sign. Trousseau’s sign is the induction of carpopedal spasms after inflating the blood pressure cuff above systolic blood pressure.
The most common etiology of hypoparathyroidism is iatrogenic, with damage to one or multiple parathyroid glands occurring during surgery. Thyroid, parathyroid, laryngeal, and pharyngeal surgeries commonly risk damage to the parathyroid glands that can be temporary or permanent. Other causes of hypoparathyroidism include congenital parathyroid aplasia, as in DiGeorge syndrome, autoimmune destruction, infiltrative processes like hemochromatosis, magnesium deficiency, or genetic mutations in PTH receptors.