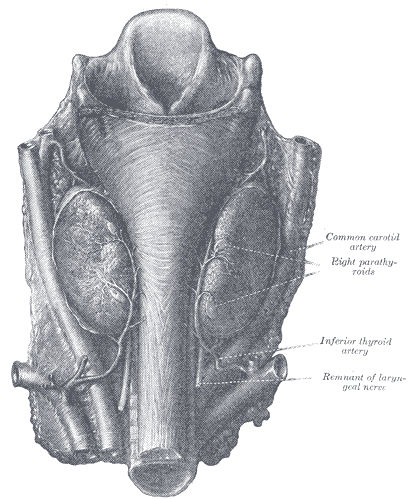
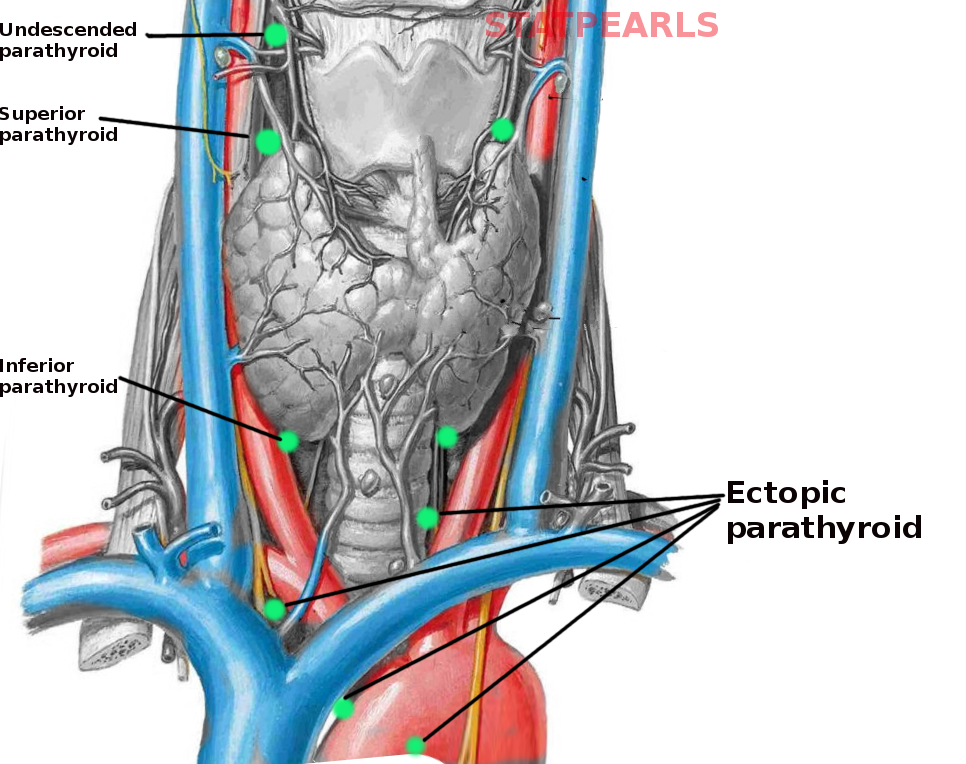
[1]
Caron NR, Sturgeon C, Clark OH. Persistent and recurrent hyperparathyroidism. Current treatment options in oncology. 2004 Aug:5(4):335-45
[PubMed PMID: 15233910]
[2]
Fraser WD. Hyperparathyroidism. Lancet (London, England). 2009 Jul 11:374(9684):145-58. doi: 10.1016/S0140-6736(09)60507-9. Epub
[PubMed PMID: 19595349]
[3]
Taterra D, Wong LM, Vikse J, Sanna B, Pękala P, Walocha J, Cirocchi R, Tomaszewski K, Henry BM. The prevalence and anatomy of parathyroid glands: a meta-analysis with implications for parathyroid surgery. Langenbeck's archives of surgery. 2019 Feb:404(1):63-70. doi: 10.1007/s00423-019-01751-8. Epub 2019 Feb 14
[PubMed PMID: 30762091]
Level 1 (high-level) evidence
[4]
Culhane KJ, Belina ME, Sims JN, Cai Y, Liu Y, Wang PSP, Yan ECY. Parathyroid Hormone Senses Extracellular Calcium To Modulate Endocrine Signaling upon Binding to the Family B GPCR Parathyroid Hormone 1 Receptor. ACS chemical biology. 2018 Aug 17:13(8):2347-2358. doi: 10.1021/acschembio.8b00568. Epub 2018 Jul 11
[PubMed PMID: 29952553]
[5]
Schappacher-Tilp G, Cherif A, Fuertinger DH, Bushinsky D, Kotanko P. A mathematical model of parathyroid gland biology. Physiological reports. 2019 Apr:7(7):e14045. doi: 10.14814/phy2.14045. Epub
[PubMed PMID: 30927339]
[6]
Choi H, Magyar CE, Nervina JM, Tetradis S. Different duration of parathyroid hormone exposure distinctively regulates primary response genes Nurr1 and RANKL in osteoblasts. PloS one. 2018:13(12):e0208514. doi: 10.1371/journal.pone.0208514. Epub 2018 Dec 21
[PubMed PMID: 30576321]
[7]
Patrinos A, Zarokosta M, Piperos T, Chrysikos D, Kakaviatos D, Theodoropoulos P, Kalles V, Tsiaoussis J, Noussios G, Mariolis-Sapsako T. WITHDRAWN: An anatomic aberration and a surgical challenge: Mediastinal parathyroid adenoma anterior the pericardium. A case report. International journal of surgery case reports. 2019:57():106-109. doi: 10.1016/j.ijscr.2019.03.014. Epub 2019 Mar 21
[PubMed PMID: 30943449]
Level 3 (low-level) evidence
[8]
Lane MJ, Desser TS, Weigel RJ, Jeffrey RB Jr. Use of color and power Doppler sonography to identify feeding arteries associated with parathyroid adenomas. AJR. American journal of roentgenology. 1998 Sep:171(3):819-23
[PubMed PMID: 9725323]
[9]
Joliat GR, Guarnero V, Demartines N, Schweizer V, Matter M. Recurrent laryngeal nerve injury after thyroid and parathyroid surgery: Incidence and postoperative evolution assessment. Medicine. 2017 Apr:96(17):e6674. doi: 10.1097/MD.0000000000006674. Epub
[PubMed PMID: 28445266]
[10]
Miura D. Ectopic parathyroid tumor in the sternohyoid muscles: supernumerary gland in a patient with MEN type 1. Journal of bone and mineral research : the official journal of the American Society for Bone and Mineral Research. 2005 Aug:20(8):1478-9
[PubMed PMID: 16007345]
[11]
Park HL, Yoo IR, Kim SH, Lee S. Multiple endocrine neoplasia type 1 with anterior mediastinal parathyroid adenoma: successful localization using Tc-99m sestamibi SPECT/CT. Annals of surgical treatment and research. 2016 Dec:91(6):323-326
[PubMed PMID: 27904855]