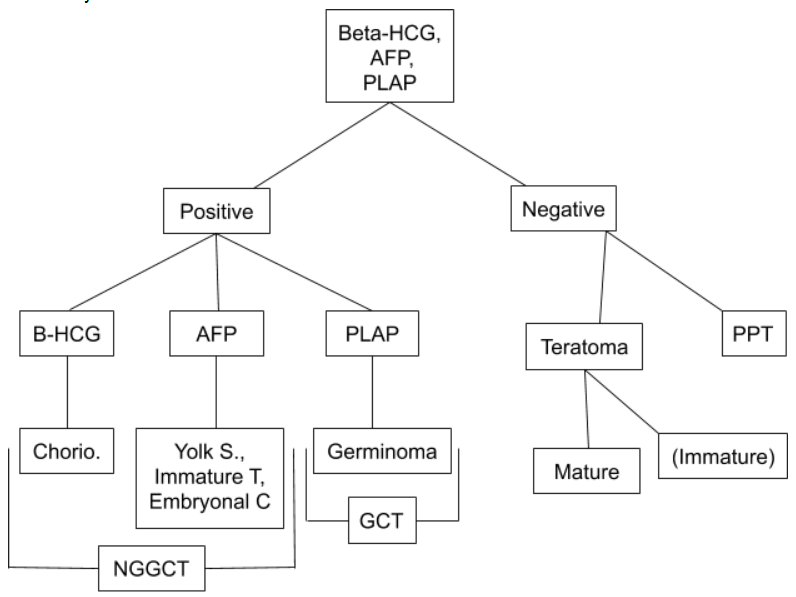
[1]
Smith AB, Rushing EJ, Smirniotopoulos JG. From the archives of the AFIP: lesions of the pineal region: radiologic-pathologic correlation. Radiographics : a review publication of the Radiological Society of North America, Inc. 2010 Nov:30(7):2001-20. doi: 10.1148/rg.307105131. Epub
[PubMed PMID: 21057132]
[2]
Feingold KR, Anawalt B, Blackman MR, Boyce A, Chrousos G, Corpas E, de Herder WW, Dhatariya K, Dungan K, Hofland J, Kalra S, Kaltsas G, Kapoor N, Koch C, Kopp P, Korbonits M, Kovacs CS, Kuohung W, Laferrère B, Levy M, McGee EA, McLachlan R, New M, Purnell J, Sahay R, Shah AS, Singer F, Sperling MA, Stratakis CA, Trence DL, Wilson DP, Arendt J, Aulinas A. Physiology of the Pineal Gland and Melatonin. Endotext. 2000:():
[PubMed PMID: 31841296]
[3]
Yamamoto I. Pineal region tumor: surgical anatomy and approach. Journal of neuro-oncology. 2001 Sep:54(3):263-75
[PubMed PMID: 11767292]
[4]
Louis DN, Perry A, Reifenberger G, von Deimling A, Figarella-Branger D, Cavenee WK, Ohgaki H, Wiestler OD, Kleihues P, Ellison DW. The 2016 World Health Organization Classification of Tumors of the Central Nervous System: a summary. Acta neuropathologica. 2016 Jun:131(6):803-20. doi: 10.1007/s00401-016-1545-1. Epub 2016 May 9
[PubMed PMID: 27157931]
[5]
Osborn AG, Salzman KL, Thurnher MM, Rees JH, Castillo M. The new World Health Organization Classification of Central Nervous System Tumors: what can the neuroradiologist really say? AJNR. American journal of neuroradiology. 2012 May:33(5):795-802. doi: 10.3174/ajnr.A2583. Epub 2011 Aug 11
[PubMed PMID: 21835942]
[6]
Nishioka H, Inoshita N. New WHO classification of pituitary adenomas (4th edition): assessment of pituitary transcription factors and the prognostic histological factors. Brain tumor pathology. 2018 Apr:35(2):57-61. doi: 10.1007/s10014-017-0307-7. Epub 2018 Jan 9
[PubMed PMID: 29318396]
[7]
Dumrongpisutikul N, Intrapiromkul J, Yousem DM. Distinguishing between germinomas and pineal cell tumors on MR imaging. AJNR. American journal of neuroradiology. 2012 Mar:33(3):550-5. doi: 10.3174/ajnr.A2806. Epub 2011 Dec 15
[PubMed PMID: 22173760]
[8]
Rodjan F, de Graaf P, Moll AC, Imhof SM, Verbeke JI, Sanchez E, Castelijns JA. Brain abnormalities on MR imaging in patients with retinoblastoma. AJNR. American journal of neuroradiology. 2010 Sep:31(8):1385-9. doi: 10.3174/ajnr.A2102. Epub 2010 Apr 22
[PubMed PMID: 20413604]
[9]
de Jong MC, Kors WA, de Graaf P, Castelijns JA, Kivelä T, Moll AC. Trilateral retinoblastoma: a systematic review and meta-analysis. The Lancet. Oncology. 2014 Sep:15(10):1157-67. doi: 10.1016/S1470-2045(14)70336-5. Epub 2014 Aug 7
[PubMed PMID: 25126964]
Level 1 (high-level) evidence
[10]
Kilgore DP, Strother CM, Starshak RJ, Haughton VM. Pineal germinoma: MR imaging. Radiology. 1986 Feb:158(2):435-8
[PubMed PMID: 3941869]
[11]
Bazot M, Cortez A, Sananes S, Buy JN. Imaging of pure primary ovarian choriocarcinoma. AJR. American journal of roentgenology. 2004 Jun:182(6):1603-4
[PubMed PMID: 15150023]
[12]
Allen SD, Lim AK, Seckl MJ, Blunt DM, Mitchell AW. Radiology of gestational trophoblastic neoplasia. Clinical radiology. 2006 Apr:61(4):301-13
[PubMed PMID: 16546459]
[13]
Takeuchi M, Matsuzaki K, Uehara H, Yoshida S, Nishitani H, Shimazu H. Pathologies of the uterine endometrial cavity: usual and unusual manifestations and pitfalls on magnetic resonance imaging. European radiology. 2005 Nov:15(11):2244-55
[PubMed PMID: 16228215]
[14]
Horowitz MB, Hall WA. Central nervous system germinomas. A review. Archives of neurology. 1991 Jun:48(6):652-7
[PubMed PMID: 2039390]
[15]
Smirniotopoulos JG, Rushing EJ, Mena H. Pineal region masses: differential diagnosis. Radiographics : a review publication of the Radiological Society of North America, Inc. 1992 May:12(3):577-96
[PubMed PMID: 1609147]
[16]
Tan HW, Ty A, Goh SG, Wong MC, Hong A, Chuah KL. Pineal yolk sac tumour with a solid pattern: a case report in a Chinese adult man with Down's syndrome. Journal of clinical pathology. 2004 Aug:57(8):882-4
[PubMed PMID: 15280413]
Level 3 (low-level) evidence
[17]
Nakashima T, Nishimura Y, Sakai N, Yamada H, Hara A. Germinoma in cerebral hemisphere associated with Down syndrome. Child's nervous system : ChNS : official journal of the International Society for Pediatric Neurosurgery. 1997 Oct:13(10):563-6
[PubMed PMID: 9403208]
[18]
Chik K, Li C, Shing MM, Leung T, Yuen PM. Intracranial germ cell tumors in children with and without Down syndrome. Journal of pediatric hematology/oncology. 1999 Mar-Apr:21(2):149-51
[PubMed PMID: 10206462]
[19]
Sandow BA, Dory CE, Aguiar MA, Abuhamad AZ. Best cases from the AFIP: congenital intracranial teratoma. Radiographics : a review publication of the Radiological Society of North America, Inc. 2004 Jul-Aug:24(4):1165-70
[PubMed PMID: 15256635]
Level 3 (low-level) evidence
[20]
Buetow PC, Smirniotopoulos JG, Done S. Congenital brain tumors: a review of 45 cases. AJR. American journal of roentgenology. 1990 Sep:155(3):587-93
[PubMed PMID: 2167004]
Level 3 (low-level) evidence
[21]
Sumida M, Barkovich AJ, Newton TH. Development of the pineal gland: measurement with MR. AJNR. American journal of neuroradiology. 1996 Feb:17(2):233-6
[PubMed PMID: 8938291]
[22]
Ueno T, Tanaka YO, Nagata M, Tsunoda H, Anno I, Ishikawa S, Kawai K, Itai Y. Spectrum of germ cell tumors: from head to toe. Radiographics : a review publication of the Radiological Society of North America, Inc. 2004 Mar-Apr:24(2):387-404
[PubMed PMID: 15026588]
[23]
Gempt J, Ringel F, Oexle K, Delbridge C, Förschler A, Schlegel J, Meyer B, Schmidt-Graf F. Familial pineocytoma. Acta neurochirurgica. 2012 Aug:154(8):1413-6. doi: 10.1007/s00701-012-1402-5. Epub 2012 Jun 15
[PubMed PMID: 22699425]
[24]
Kaido T, Sasaoka Y, Hashimoto H, Taira K. De novo germinoma in the brain in association with Klinefelter's syndrome: case report and review of the literature. Surgical neurology. 2003 Dec:60(6):553-8; discussion 559
[PubMed PMID: 14670679]
Level 3 (low-level) evidence
[25]
Hashimoto T, Sasagawa I, Ishigooka M, Kubota Y, Nakada T, Fujita T, Nakai O. Down's syndrome associated with intracranial germinoma and testicular embryonal carcinoma. Urologia internationalis. 1995:55(2):120-2
[PubMed PMID: 8533196]
[26]
Wong TT, Ho DM, Chang TK, Yang DD, Lee LS. Familial neurofibromatosis 1 with germinoma involving the basal ganglion and thalamus. Child's nervous system : ChNS : official journal of the International Society for Pediatric Neurosurgery. 1995 Aug:11(8):456-8
[PubMed PMID: 7585682]
[27]
Fakhran S, Escott EJ. Pineocytoma mimicking a pineal cyst on imaging: true diagnostic dilemma or a case of incomplete imaging? AJNR. American journal of neuroradiology. 2008 Jan:29(1):159-63
[PubMed PMID: 17925371]
Level 3 (low-level) evidence
[28]
Chang AH, Fuller GN, Debnam JM, Karis JP, Coons SW, Ross JS, Dean BL. MR imaging of papillary tumor of the pineal region. AJNR. American journal of neuroradiology. 2008 Jan:29(1):187-9
[PubMed PMID: 17925365]
[29]
Beker-Acay M, Turamanlar O, Horata E, Unlu E, Fidan N, Oruc S. Assessment of Pineal Gland Volume and Calcification in Healthy Subjects: Is it Related to Aging? Journal of the Belgian Society of Radiology. 2016 Feb 1:100(1):13. doi: 10.5334/jbr-btr.892. Epub 2016 Feb 1
[PubMed PMID: 30038974]
[30]
Labidi M, Lavoie P, Lapointe G, Obaid S, Weil AG, Bojanowski MW, Turmel A. Predicting success of endoscopic third ventriculostomy: validation of the ETV Success Score in a mixed population of adult and pediatric patients. Journal of neurosurgery. 2015 Dec:123(6):1447-55. doi: 10.3171/2014.12.JNS141240. Epub 2015 Jul 24
[PubMed PMID: 26207604]
Level 1 (high-level) evidence
[31]
Kulwin C, Matsushima K, Malekpour M, Cohen-Gadol AA. Lateral supracerebellar infratentorial approach for microsurgical resection of large midline pineal region tumors: techniques to expand the operative corridor. Journal of neurosurgery. 2016 Jan:124(1):269-76. doi: 10.3171/2015.2.JNS142088. Epub 2015 Aug 14
[PubMed PMID: 26275000]
[32]
Rey-Dios R, Cohen-Gadol AA. A surgical technique to expand the operative corridor for supracerebellar infratentorial approaches: technical note. Acta neurochirurgica. 2013 Oct:155(10):1895-900. doi: 10.1007/s00701-013-1844-4. Epub 2013 Aug 28
[PubMed PMID: 23982230]
[33]
Moshel YA, Parker EC, Kelly PJ. Occipital transtentorial approach to the precentral cerebellar fissure and posterior incisural space. Neurosurgery. 2009 Sep:65(3):554-64; discussion 564. doi: 10.1227/01.NEU.0000350898.68212.AB. Epub
[PubMed PMID: 19687701]
[34]
Mootha SL, Barkovich AJ, Grumbach MM, Edwards MS, Gitelman SE, Kaplan SL, Conte FA. Idiopathic hypothalamic diabetes insipidus, pituitary stalk thickening, and the occult intracranial germinoma in children and adolescents. The Journal of clinical endocrinology and metabolism. 1997 May:82(5):1362-7
[PubMed PMID: 9141516]