Continuing Education Activity
Central toxic keratopathy (CTK) was initially described as a complication of LASIK surgery, and the vast majority of published reports are associated with excimer laser ablation. This activity discusses the etiology, pathophysiology, and debated management by an interprofessional team of central toxic keratopathy (CTK) and its relation to diffuse lamellar keratitis (DLK). Additionally, it reviews physical exam findings and the prognosis of patients with CTK.
Objectives:
- Outline the typical clinical picture associated with central toxic keratopathy (CTK).
- Summarize the underlying pathophysiologic changes in CTK.
- Explain the various treatment regimens for CTK. Summarize the differential diagnosis in a patient with CTK.
- Review the role of the interprofessional team in the care of central toxic keratopathy.
Introduction
Excimer laser ablation is an increasingly popular option for the correction of mild to moderate refractive errors in the United States. It is generally regarded as safe, with an overall annual complication rate of less than 0.8% since 2010.[1] Central toxic keratopathy (CTK), first described in 1998, has numerous alternative descriptive names including stage IV diffuse lamellar keratitis, central lamellar keratitis, central flap necrosis, central necrosis lamellar inflammation, and keratinocyte-induced corneal micro edema (KME).[2][3][4][5][6][7] This rare clinical syndrome classically presents as a central or paracentral, amorphous corneal opacification with associated striae, stromal loss, and hyperopic shifts that usually present within nine days of excimer laser ablation (i.e., laser-assisted keratomileusis (LASIK) or photorefractive keratopathy [PRK]).[5][8][9] It is believed to be non-inflammatory, but its cause is still unknown. Though initially thought of as a progression, or severe form, of diffuse lamellar keratitis (DLK)[3], there is significant support that CTK is a distinct and separate entity from DLK.[8][9] In addition to cases in other refractive surgeries, there are proposals that CTK exists as an entity outside of the refractive surgery literature: there have been reports of CTK associated with contact lens use with and without recent mechanical debridement, idiopathically, as well as potentially with selective laser trabeculoplasty or topical anesthetic use.[10][11][12][13][14][15][16][17]
Etiology
The etiology of CTK is still unelucidated. However, there are several hypotheses, including a reaction to a component of surgical gloves, meibomian gland secretions, povidone-iodine, or marking pen ink.[8][9][18][19] Also, since the opacity occurs centrally, in an area most affected by the laser, it has been hypothesized that the ultraviolet laser energy may have a pathophysiologic, perhaps toxin-activating, the role of the substances as mentioned earlier.[9] In addition to cases that do not involve excimer laser ablation, there are four published cases where the opacification occurs in the periphery (termed peripheral toxic keratopathy), making this hypothesis less likely, although some activating source may be responsible.[20] In the absence of excimer laser therapy, some authors have proposed possible options, including inflammatory cytokines and metalloproteinases, and antigen-antibody immunologic responses that may be the underlying cause.[11][14]
As the etiology of CTK is only speculative, there are no proven risk factors that lead to its development. However, CTK's initial description as a complication of LASIK surgery, and the vast majority of published reports are associated with excimer laser ablation. Therefore, patients undergoing refractive laser correction seem to be at the greatest risk. Additionally, since a majority of cases of CTK correlate with preceding DLK (95% in the initial study by Sonmez et al.), or persistent, and therefore coexistent, (64%), risk factors associated with DLK may also have an association with CTK.[9][21] These risk factors, however, are unlikely to be applicable for patients who present with CTK and no history of recent refractive surgery.
Epidemiology
To the author's knowledge, there has yet been no study completed to assess epidemiologic data for cases of CTK. However, in LASIK treated eyes, one study reported that out of 522 eyes, four developed CTK (0.77%), and in another study of 17100 eyes, nine developed CTK (0.05%).[18][5]
Pathophysiology
Pathophysiologic changes in CTK are a topic of interest, as the mechanisms that cause this rare syndrome remain incompletely established. However, some aspects of the underlying physiologic mechanisms are understood, and there have been many proposed theories.
One principal aspect of the disease is central corneal thinning or flattening, up to over 100 micrometers in one case.[22] This degeneration is most prominent within the first week and is followed by a period of regeneration that resolves the overall hyperopic shift, to a degree.[23] Research has shown that there is stromal loss, and corneal stretching has been a proposed alternative or secondary process.[7][8][23]
The loss of stromal tissue is likely due to one of two possible mechanisms; either there could be apoptosis of keratocytes, enzymatic degradation of the stromal matrix, or a combination of the two. A lack of associated inflammation has made keratocyte death by apoptosis the predominant theory, and the striae and opacity that characterize the disease may be the gross visualization of this microscopic process.[8][9][12][23][24] Apoptosis leads to the loss of tissue, causing a corneal flattening that is predominantly found in the anterior tangential curvature of the cornea, while the posterior tangential curvature shows minimal changes. This change indicates that as the stromal cells undergo apoptosis, it is the anterior surface that may fall to fill the gap left behind.[23] Following the initial stromal loss, the steepening corrective phase occurs over subsequent weeks to months and is most active shortly after the thinning resolves.[23] This thickening process is less studied and may occur as part of epithelial wound healing and stromal remodeling. Optical coherence tomography (OCT) imaging demonstrating increased reflectivity may indicate that there may be a disorganized cellular matrix associated with this process.[8]
Additional theories to account for the fluctuations in corneal curvature include the idea that keratinocyte injury and subsequent cytokine release cause changes in interstitial fluid pressure and corneal micro-edema. Fluid accumulating more prominently in the mid-periphery as opposed to the central cornea (due to its more rigid collagen matrix composition) causes changes in tissue tension. Thus the stromal bed flattens as a result of stretching.[7]
An additional hypothesis proposes an immunologic process may also be present, as confocal imaging has shown potential immune complexes within the stroma.[25]
A final theory revolves around a DLK based inflammatory model inciting CTK. It proposes that the release of collagenase and various proteases from damaged keratinocytes may cause the observed stromal thinning.[26] However, the understanding that CTK is not predicated on preceding DLK makes this theory less likely.[9]
History and Physical
Patients who develop CTK may initially present with DLK and associated inflammation after having undergone excimer laser ablation. Thus, patients may complain of nonspecific findings such as pain, photophobia, halos, floaters, a foreign body sensation, and/or blurry vision, which is common to this pathology. However, DLK is not necessary for diagnosis.[9] On follow up for photorefractive procedures, those first diagnosed with DLK may be identified within days of the procedure. Patients with DLK should be given appropriate treatment and monitored for subsequent or concurrent CTK.[8][9]
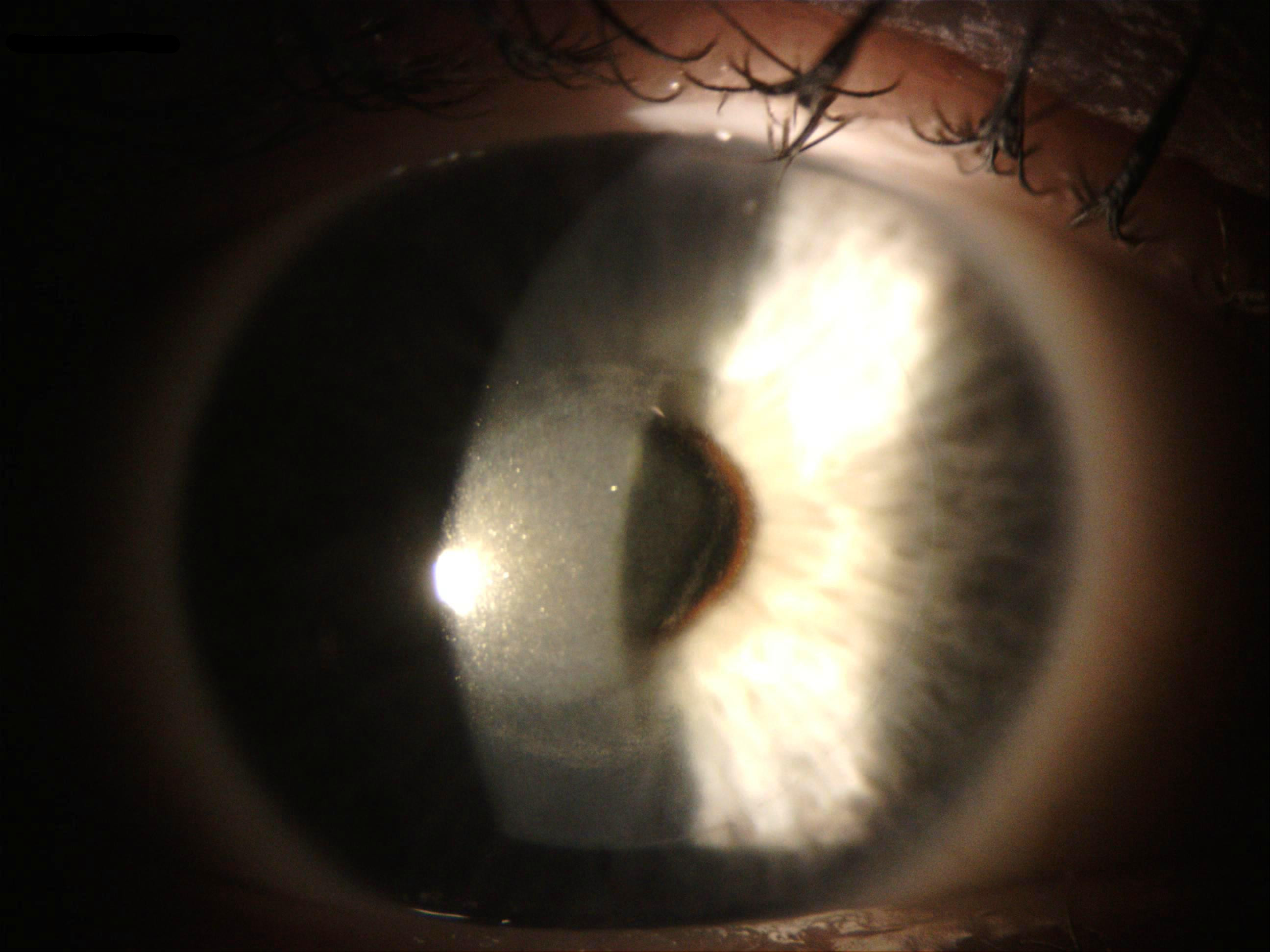
On the slit lamp exam, the prominent finding in CTK is a non-inflammatory central or pericentral amorphous corneal opacity involving the stroma. This condition leads to a decreased visual acuity in the affected eye(s), generally within nine days of surgery. The cloudiness (distinct from the hazy cornea that is occasionally associated with PRK surgery) reportedly persists for 2 to 18 months before resolving spontaneously. Though most opacities largely clear within a year, residual stromal thinning and decreases in uncorrected visual acuity may remain.[8][9]
A complete ocular exam with OCT in cases of CTK may also show an inverse dome-shaped appearance of the opacity with backscattering within the central cornea and increased homogenous internal reflectivity that extends from the anterior to the posterior stroma.[8][11] Keratometry may reveal anterior corneal flattening and thinning, leading to hyperopic shift. Anterior corneal flattening (by three to six diopters) occurs most prominently within the first month and is reversed (by one to three diopters) over time.[8][23] Additionally, in vivo, confocal microscopy may show activated keratocytes and folds in Bowman’s layer with no obvious accumulation of inflammatory cells.[27][28]
Evaluation
There is no confirmatory imaging or diagnostic test available for CTK, and the diagnosis is clinical. A triad of findings including a non-inflammatory central corneal opacity with or without striae, corneal thinning, and a hyperopic change in visual acuity can lead clinicians to make a diagnosis. In patients who have recently undergone excimer laser ablation and/or presented with DLK in the preceding days, the diagnosis may be more definite. When considering cases without a history of excimer laser ablation, a high index of clinical suspicion is necessary to identify and diagnose these patients correctly.
Treatment / Management
Whereas steroid-based treatment was the initial recommendation, the newer understanding that CTK is a distinct syndrome that is likely non-inflammatory in nature has caused a dramatic shift in management. Though topical steroids do have an important use in the treatment of DLK stages I-III which may precede, or coexist with CTK, steroids may worsen disease progression in patients with CTK.[8] Steroids may inhibit keratocytes and fibroblasts from rebuilding the cellular matrix; thus, once topical steroid treatment stops, keratocytes and fibroblasts are better able to proliferate and reconstitute the corneal thickness.[23][29] Sonmez et al. recommend the use of steroids in DLK only until the opacification is present, and there are no visible leukocytes. After this point, the use of steroids in CTK likely has no benefit and exposes the patient to unnecessary side effects of medication.[9] Stromal loss may be most predominant within the first four weeks as a result of steroid therapy.[23] With an understanding that the opacity in CTK largely clears without intervention, close follow up and regular monitoring without invasive intervention is typically the suggested approach.[2][3][8][9]
While there is no standard of care for the treatment of CTK, other medical management options may be beneficial. As keratinocytes regenerate and produce stromal extracellular matrix, metalloproteases may slow healing and contribute to the development of CTK.[11] Therefore, the inhibition of matrix metalloproteases may prove advantageous in promoting increased corneal thickness and reducing visual acuity changes. In this regard, doxycycline, a known metalloproteinase inhibitor, may help to prevent corneal matrix degeneration and promote proliferative effects.[30][31] Similarly, ascorbic acid (vitamin C) can promote extracellular matrix deposition and stem cell proliferation, aiding in the corneal wound healing process.[32] Conversely, mitomycin-C, frequently used off-label in excimer laser procedures to reduce postoperative haze from PRK, has been shown to reduce keratocyte proliferation and new collagen deposition and may exacerbate the stromal loss in CTK.[33] Finally, potential underlying corneal micro edema may warrant the use of hyperosmotic agents to reduce corneal refractive changes, as these have been of some benefit in a few cases.[7]
Since the opacity is not limited to the interface and expands down into the stroma, in addition to the fact that stomal loss is a prominent aspect of the disease, invasive flap lift, and irrigation are generally not recommended for fear of further tissue loss and thinning. However, some authors have proposed that surgical interventions do have a place in management[34], and report incidental findings in which invasive procedures seem to speed up recovery.[35] However, more invasive procedures have not demonstrably shown to improve outcomes. In a report of three patients who developed CTK within four days of each other (two of which received flap lift with irrigation and one that opted for medical management), Tu and Aslanides reported that flap lift and irrigation in early CTK resulted in an arrest of the development of the opacity in one patient. Both patients who received more invasive procedures fared better than their patient who did not. They additionally reported that the stroma of the flap in their patients was less affected than the residual posterior stroma and state that, “flap lift and irrigation during early phases of CTK need not result in significant stromal loss and can be of benefit.”[34] Flap lift and management of striae, if present, has also been suggested as a possible therapy to provide better visual acuity.[7] Finally, reports exist of flap amputation to correct hyperopic defects associated with CTK.[36]
Regardless of the decision to manage solely by medical means, or to implement a surgical intervention as well, patients should be followed closely with serial topography, tomography, confocal microscopy, and OCT imaging to better monitor corneal thickness and changes in visual acuity. Additionally, monitoring patients in this way may contribute to a greater understanding of the underlying pathophysiology and long-term effects that may result from CTK.
Differential Diagnosis
The differential diagnosis for CTK is varied and based on conditions that may present with central or paracentral opacifications or changes in visual acuity. As stated above, the previous thinking was that CTK was a severe form of DLK. While we now better understand the difference between the two pathologies, distinguishing CTK from DLK by the localization, response to steroids, or hyperopic changes is important as the two may coexist. Additionally, unlike DLK, in which the opacification remains contained within the interface, the amorphous opaqueness of CTK can be found expanding into the corneal flap anteriorly, and residual stromal bed posteriorly.[8][9]
Infectious keratitis may also result in a dense central opacity with infiltrate. However, CTK does not result in an infiltrate due to the lack of inflammation and also lacks the characteristic off-yellow dullness of inflammatory lesions. Furthermore, infectious keratitis may present with other signs that are not associated with CTK, such as a progressive spread, conjunctival hyperemia, purulent discharge, inflammatory cells, and an anterior chamber reaction.[8][9]
Corneal cross-linking (CXL) and photorefractive keratectomy (PRK) are also associated with postsurgical haze, which can be confused with CTK. Post-procedural haze differs in its time course and generally develops more than a month after surgery, whereas CTK most often presents within nine days.[8][11]
In patients with previous LASIK surgery, epithelial ingrowth may occur, resulting in opacification. These opacifications are distinguishable from CTK due to their delayed onset and peripheral location around the flap edges.[8] Finally, other diagnoses that may be possibilities in the differential diagnosis include interface fluid or debris, an acute steroid response, increased intraocular pressure based haze, superficial punctate keratitis, and trauma.[7][8] Each merits consideration when making the diagnosis of CTK.
Prognosis
The prognosis for patients with CTK may be fair to good, as the central opacity eventually clears to a degree, and stromal thickness increases with time. However, residual hyperopic defects often remain, which may be disappointing for patients whose initial desire to undergoing excimer laser ablation was uncorrected emmetropia. However, patients may undergo post-surgical enhancement after the opacity remits to reduce residual striae and correct persistent hyperopia without the recurrence of CTK.[9]
Complications
Undiagnosed or poorly managed cases of CTK may result in worse outcomes for patients. As stated earlier in the text, continued steroid treatment may also delay or worsen the disease.[23] The most prominent persistent complications after the opacity mostly clears, are residual striae and hyperopia, which are addressable with refractive surgery.[9]
Deterrence and Patient Education
Since the etiology of CTK is uncertain, and reports exist of cases with no identifiable inciting factor, no definitive statements are possible about avoiding or decreasing the likelihood of developing CTK. However, the vast majority of cases are associated with excimer laser ablation and elective visual corrective surgery. Though these complications are rare, patients undergoing these surgeries should be made aware of the risks and potentially unfavorable outcomes associated with these procedures during the process of informed consent.
Pearls and Other Issues
- CTK is a distinct entity from DLK with a different pathophysiologic process with no apparent signs of inflammation.
- CTK generally manifests within nine days of excimer laser ablation, though there is documentation of other etiologies.
- Clinical signs include a central or paracentral amorphous opacity with striae, hyperopic shift, and stromal loss. The opacification may clear significantly without treatment, and the stroma will regenerate to a degree; however, hyperopia and striae may remain.
- The best treatment for CTK is subject to debate, and its general rarity makes well-controlled treatment studies difficult. However, most clinicians do not recommend invasive intervention. Steroids are universally understood to be of little or no benefit and are not warranted. Other corneal protective agents may be of use.
- After the opacity resolves to a degree, patients who had CTK may undergo enhancement procedures to correct hyperopia and clear striae without disease recurrence.
Enhancing Healthcare Team Outcomes
Although not explicitly stated, patients with CTK are most commonly identified in routine follow up after excimer laser ablative surgeries. In cases that present elsewhere (e.g., to their primary care physician for changes in vision and a corneal opacity), ophthalmic consultation is warranted. Clinicians should consider CTK as a potential cause in these cases even if a history of ocular surgery is not present in the recent past.