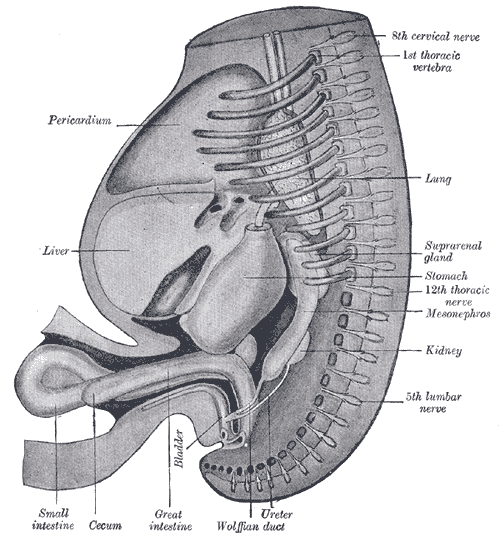
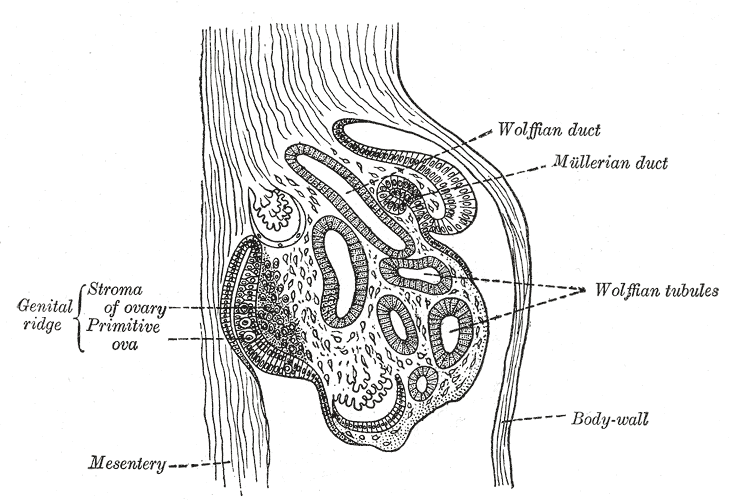
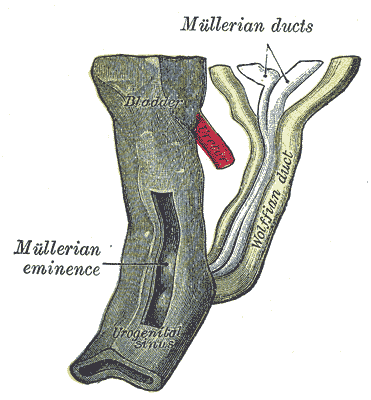
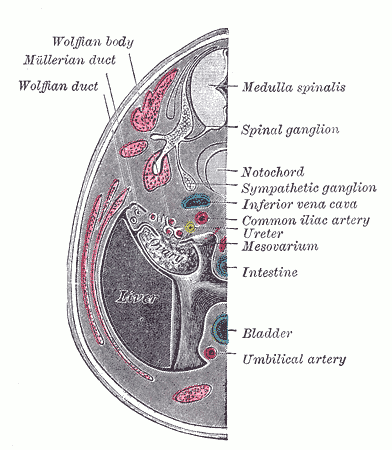
[2]
Sainio K, Hellstedt P, Kreidberg JA, Saxén L, Sariola H. Differential regulation of two sets of mesonephric tubules by WT-1. Development (Cambridge, England). 1997 Apr:124(7):1293-9
[PubMed PMID: 9118800]
[3]
Torres M, Gómez-Pardo E, Dressler GR, Gruss P. Pax-2 controls multiple steps of urogenital development. Development (Cambridge, England). 1995 Dec:121(12):4057-65
[PubMed PMID: 8575306]
[4]
Pedersen A, Skjong C, Shawlot W. Lim 1 is required for nephric duct extension and ureteric bud morphogenesis. Developmental biology. 2005 Dec 15:288(2):571-81
[PubMed PMID: 16216236]
[5]
Murashima A, Xu B, Hinton BT. Understanding normal and abnormal development of the Wolffian/epididymal duct by using transgenic mice. Asian journal of andrology. 2015 Sep-Oct:17(5):749-55. doi: 10.4103/1008-682X.155540. Epub
[PubMed PMID: 26112482]
Level 3 (low-level) evidence
[6]
Kreidberg JA, Sariola H, Loring JM, Maeda M, Pelletier J, Housman D, Jaenisch R. WT-1 is required for early kidney development. Cell. 1993 Aug 27:74(4):679-91
[PubMed PMID: 8395349]
[7]
Kobayashi H, Kawakami K, Asashima M, Nishinakamura R. Six1 and Six4 are essential for Gdnf expression in the metanephric mesenchyme and ureteric bud formation, while Six1 deficiency alone causes mesonephric-tubule defects. Mechanisms of development. 2007 Apr:124(4):290-303
[PubMed PMID: 17300925]
[8]
Miyamoto N, Yoshida M, Kuratani S, Matsuo I, Aizawa S. Defects of urogenital development in mice lacking Emx2. Development (Cambridge, England). 1997 May:124(9):1653-64
[PubMed PMID: 9165114]
[9]
Grote D, Souabni A, Busslinger M, Bouchard M. Pax 2/8-regulated Gata 3 expression is necessary for morphogenesis and guidance of the nephric duct in the developing kidney. Development (Cambridge, England). 2006 Jan:133(1):53-61
[PubMed PMID: 16319112]
[10]
Poladia DP,Kish K,Kutay B,Hains D,Kegg H,Zhao H,Bates CM, Role of fibroblast growth factor receptors 1 and 2 in the metanephric mesenchyme. Developmental biology. 2006 Mar 15;
[PubMed PMID: 16442091]
[11]
Okazawa M, Murashima A, Harada M, Nakagata N, Noguchi M, Morimoto M, Kimura T, Ornitz DM, Yamada G. Region-specific regulation of cell proliferation by FGF receptor signaling during the Wolffian duct development. Developmental biology. 2015 Apr 1:400(1):139-47. doi: 10.1016/j.ydbio.2015.01.023. Epub 2015 Feb 9
[PubMed PMID: 25678108]
[12]
Kume T, Deng K, Hogan BL. Murine forkhead/winged helix genes Foxc1 (Mf1) and Foxc2 (Mfh1) are required for the early organogenesis of the kidney and urinary tract. Development (Cambridge, England). 2000 Apr:127(7):1387-95
[PubMed PMID: 10704385]
[13]
Murashima A, Akita H, Okazawa M, Kishigami S, Nakagata N, Nishinakamura R, Yamada G. Midline-derived Shh regulates mesonephric tubule formation through the paraxial mesoderm. Developmental biology. 2014 Feb 1:386(1):216-26. doi: 10.1016/j.ydbio.2013.12.026. Epub 2013 Dec 24
[PubMed PMID: 24370450]
[14]
Mendelsohn C, Lohnes D, Décimo D, Lufkin T, LeMeur M, Chambon P, Mark M. Function of the retinoic acid receptors (RARs) during development (II). Multiple abnormalities at various stages of organogenesis in RAR double mutants. Development (Cambridge, England). 1994 Oct:120(10):2749-71
[PubMed PMID: 7607068]
[15]
Karner CM, Chirumamilla R, Aoki S, Igarashi P, Wallingford JB, Carroll TJ. Wnt9b signaling regulates planar cell polarity and kidney tubule morphogenesis. Nature genetics. 2009 Jul:41(7):793-9. doi: 10.1038/ng.400. Epub 2009 Jun 21
[PubMed PMID: 19543268]
[16]
Shima Y, Miyabayashi K, Haraguchi S, Arakawa T, Otake H, Baba T, Matsuzaki S, Shishido Y, Akiyama H, Tachibana T, Tsutsui K, Morohashi K. Contribution of Leydig and Sertoli cells to testosterone production in mouse fetal testes. Molecular endocrinology (Baltimore, Md.). 2013 Jan:27(1):63-73. doi: 10.1210/me.2012-1256. Epub 2012 Nov 2
[PubMed PMID: 23125070]
[17]
Donjacour AA, Thomson AA, Cunha GR. FGF-10 plays an essential role in the growth of the fetal prostate. Developmental biology. 2003 Sep 1:261(1):39-54
[PubMed PMID: 12941620]
[18]
Tomaszewski J, Joseph A, Archambeault D, Yao HH. Essential roles of inhibin beta A in mouse epididymal coiling. Proceedings of the National Academy of Sciences of the United States of America. 2007 Jul 3:104(27):11322-7
[PubMed PMID: 17592132]
[19]
Nie X, Arend LJ. Pkd1 is required for male reproductive tract development. Mechanisms of development. 2013 Nov-Dec:130(11-12):567-76. doi: 10.1016/j.mod.2013.07.006. Epub 2013 Aug 9
[PubMed PMID: 23933588]
[20]
Jequier AM, Ansell ID, Bullimore NJ. Congenital absence of the vasa deferentia presenting with infertility. Journal of andrology. 1985 Jan-Feb:6(1):15-9
[PubMed PMID: 3918979]
[21]
McCallum TJ, Milunsky JM, Cunningham DL, Harris DH, Maher TA, Oates RD. Fertility in men with cystic fibrosis: an update on current surgical practices and outcomes. Chest. 2000 Oct:118(4):1059-62
[PubMed PMID: 11035677]
[22]
Tizzano EF, Silver MM, Chitayat D, Benichou JC, Buchwald M. Differential cellular expression of cystic fibrosis transmembrane regulator in human reproductive tissues. Clues for the infertility in patients with cystic fibrosis. The American journal of pathology. 1994 May:144(5):906-14
[PubMed PMID: 7513949]
[23]
MORRIS JM. The syndrome of testicular feminization in male pseudohermaphrodites. American journal of obstetrics and gynecology. 1953 Jun:65(6):1192-1211
[PubMed PMID: 13057950]
[24]
Hannema SE, Scott IS, Hodapp J, Martin H, Coleman N, Schwabe JW, Hughes IA. Residual activity of mutant androgen receptors explains wolffian duct development in the complete androgen insensitivity syndrome. The Journal of clinical endocrinology and metabolism. 2004 Nov:89(11):5815-22
[PubMed PMID: 15531547]
[25]
Chen F, Knecht K, Leu C, Rutledge SJ, Scafonas A, Gambone C, Vogel R, Zhang H, Kasparcova V, Bai C, Harada S, Schmidt A, Reszka A, Freedman L. Partial agonist/antagonist properties of androstenedione and 4-androsten-3beta,17beta-diol. The Journal of steroid biochemistry and molecular biology. 2004 Aug:91(4-5):247-57
[PubMed PMID: 15336702]
[26]
Berthezène F, Forest MG, Grimaud JA, Claustrat B, Mornex R. Leydig-cell agenesis: a cause of male pseudohermaphroditism. The New England journal of medicine. 1976 Oct 28:295(18):969-72
[PubMed PMID: 184390]
[27]
Chu L, Li J, Liu Y, Hu W, Cheng CH. Targeted gene disruption in zebrafish reveals noncanonical functions of LH signaling in reproduction. Molecular endocrinology (Baltimore, Md.). 2014 Nov:28(11):1785-95. doi: 10.1210/me.2014-1061. Epub 2014 Sep 19
[PubMed PMID: 25238195]