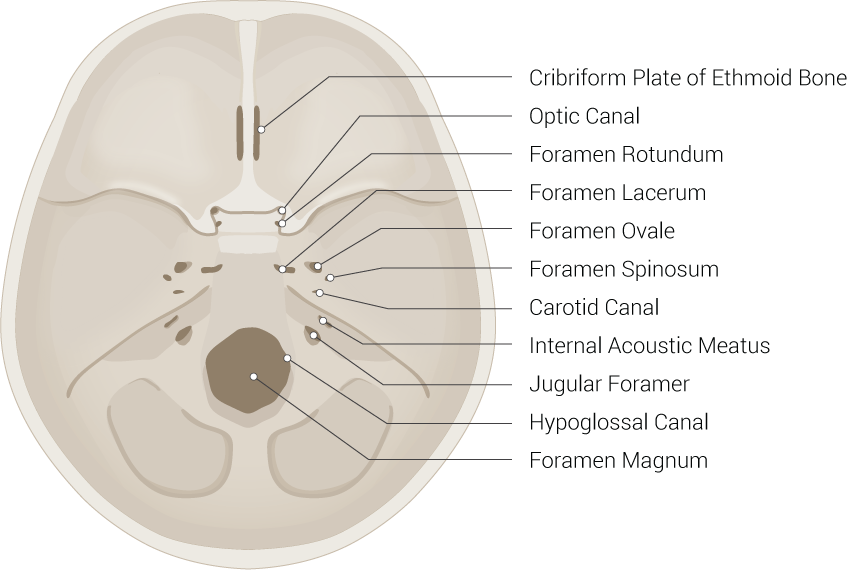
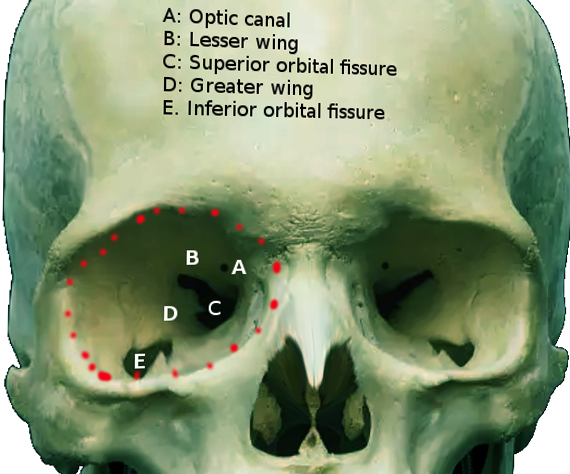
[1]
Regoli M, Bertelli E. The revised anatomy of the canals connecting the orbit with the cranial cavity. Orbit (Amsterdam, Netherlands). 2017 Apr:36(2):110-117. doi: 10.1080/01676830.2017.1279662. Epub 2017 Mar 3
[PubMed PMID: 28388344]
[2]
Kier EL. Embryology of the normal optic canal and its anomalies. An anatomic and roentgenographic study. Investigative radiology. 1966 Sep-Oct:1(5):346-62
[PubMed PMID: 5970634]
[3]
Dumic-Cule I, Eljuga D, Izadpanah A, Erjavec I, Prgomet S, Hladnik A, Bicanic I, Rora M, Vinter I, Grgurevic L. Dynamics of optic canal and orbital cavity development revealed by microCT. Surgical and radiologic anatomy : SRA. 2014 Dec:36(10):989-92. doi: 10.1007/s00276-014-1296-4. Epub 2014 Apr 19
[PubMed PMID: 24748403]
[4]
Hart CK,Theodosopoulos PV,Zimmer LA, Anatomy of the optic canal: a computed tomography study of endoscopic nerve decompression. The Annals of otology, rhinology, and laryngology. 2009 Dec;
[PubMed PMID: 20112517]
[5]
Prado PA, Ribeiro EC, De Angelis MA, Smith RL. Biometric study of the optic canal during cranial development. Orbit (Amsterdam, Netherlands). 2007 Jun:26(2):107-11
[PubMed PMID: 17613857]
[7]
van Overbeeke J, Sekhar L. Microanatomy of the blood supply to the optic nerve. Orbit (Amsterdam, Netherlands). 2003 Jun:22(2):81-8
[PubMed PMID: 12789588]
[8]
Toma N. Anatomy of the Ophthalmic Artery: Embryological Consideration. Neurologia medico-chirurgica. 2016 Oct 15:56(10):585-591
[PubMed PMID: 27298261]
[9]
Michalinos A, Zogana S, Kotsiomitis E, Mazarakis A, Troupis T. Anatomy of the Ophthalmic Artery: A Review concerning Its Modern Surgical and Clinical Applications. Anatomy research international. 2015:2015():591961. doi: 10.1155/2015/591961. Epub 2015 Nov 9
[PubMed PMID: 26635976]
[11]
De Moraes CG. Anatomy of the visual pathways. Journal of glaucoma. 2013 Jun-Jul:22 Suppl 5():S2-7. doi: 10.1097/IJG.0b013e3182934978. Epub
[PubMed PMID: 23733119]
[13]
Renn WH, Rhoton AL Jr. Microsurgical anatomy of the sellar region. Journal of neurosurgery. 1975 Sep:43(3):288-98
[PubMed PMID: 1151464]
[15]
Liu Y, Yu H, Zhen H. Navigation-assisted, endonasal, endoscopic optic nerve decompression for the treatment of nontraumatic optic neuropathy. Journal of cranio-maxillo-facial surgery : official publication of the European Association for Cranio-Maxillo-Facial Surgery. 2019 Feb:47(2):328-333. doi: 10.1016/j.jcms.2018.12.009. Epub 2018 Dec 13
[PubMed PMID: 30600198]
[16]
Chen C, Selva D, Floreani S, Wormald PJ. Endoscopic optic nerve decompression for traumatic optic neuropathy: an alternative. Otolaryngology--head and neck surgery : official journal of American Academy of Otolaryngology-Head and Neck Surgery. 2006 Jul:135(1):155-7
[PubMed PMID: 16815203]
[17]
Dinca EB, Brehar F, Giovani A, Ciurea AV. Challenges in a case of ophthalmic artery aneurysm associated with abnormal internal carotid arteries. Asian journal of neurosurgery. 2017 Jan-Mar:12(1):106-108. doi: 10.4103/1793-5482.144160. Epub
[PubMed PMID: 28413549]
Level 3 (low-level) evidence
[18]
Lenaers G, Hamel C, Delettre C, Amati-Bonneau P, Procaccio V, Bonneau D, Reynier P, Milea D. Dominant optic atrophy. Orphanet journal of rare diseases. 2012 Jul 9:7():46. doi: 10.1186/1750-1172-7-46. Epub 2012 Jul 9
[PubMed PMID: 22776096]
[19]
Adam MP, Mirzaa GM, Pagon RA, Wallace SE, Bean LJH, Gripp KW, Amemiya A, Yu-Wai-Man P, Chinnery PF. Leber Hereditary Optic Neuropathy. GeneReviews(®). 1993:():
[PubMed PMID: 20301353]
[20]
Weinreb RN, Aung T, Medeiros FA. The pathophysiology and treatment of glaucoma: a review. JAMA. 2014 May 14:311(18):1901-11. doi: 10.1001/jama.2014.3192. Epub
[PubMed PMID: 24825645]
[21]
Hollands H, Johnson D, Hollands S, Simel DL, Jinapriya D, Sharma S. Do findings on routine examination identify patients at risk for primary open-angle glaucoma? The rational clinical examination systematic review. JAMA. 2013 May 15:309(19):2035-42. doi: 10.1001/jama.2013.5099. Epub
[PubMed PMID: 23677315]
Level 1 (high-level) evidence
[22]
Hoorbakht H, Bagherkashi F. Optic neuritis, its differential diagnosis and management. The open ophthalmology journal. 2012:6():65-72. doi: 10.2174/1874364101206010065. Epub 2012 Jul 24
[PubMed PMID: 22888383]
[23]
Hayreh SS. Management of ischemic optic neuropathies. Indian journal of ophthalmology. 2011 Mar-Apr:59(2):123-36. doi: 10.4103/0301-4738.77024. Epub
[PubMed PMID: 21350282]
[24]
Rigi M, Almarzouqi SJ, Morgan ML, Lee AG. Papilledema: epidemiology, etiology, and clinical management. Eye and brain. 2015:7():47-57. doi: 10.2147/EB.S69174. Epub 2015 Aug 17
[PubMed PMID: 28539794]
Level 2 (mid-level) evidence
[25]
Schick U, Jung C, Hassler WE. Primary optic nerve sheath meningiomas: a follow-up study. Central European neurosurgery. 2010 Aug:71(3):126-33. doi: 10.1055/s-0029-1246136. Epub 2010 Feb 1
[PubMed PMID: 20127592]
[26]
Parker RT,Ovens CA,Fraser CL,Samarawickrama C, Optic nerve sheath meningiomas: prevalence, impact, and management strategies. Eye and brain. 2018;
[PubMed PMID: 30498385]
[27]
Xie X, Zhang X, Fu J, Wang H, Jonas JB, Peng X, Tian G, Xian J, Ritch R, Li L, Kang Z, Zhang S, Yang D, Wang N, Beijing iCOP Study Group. Noninvasive intracranial pressure estimation by orbital subarachnoid space measurement: the Beijing Intracranial and Intraocular Pressure (iCOP) study. Critical care (London, England). 2013 Jul 24:17(4):R162. doi: 10.1186/cc12841. Epub 2013 Jul 24
[PubMed PMID: 23883736]
[28]
Zhang X, Lee Y, Olson D, Fleischman D. Evaluation of optic canal anatomy and symmetry using CT. BMJ open ophthalmology. 2019:4(1):e000302. doi: 10.1136/bmjophth-2019-000302. Epub 2019 May 28
[PubMed PMID: 31245611]
[29]
Fard MA, Sahraiyan A, Jalili J, Hejazi M, Suwan Y, Ritch R, Subramanian PS. Optical Coherence Tomography Angiography in Papilledema Compared With Pseudopapilledema. Investigative ophthalmology & visual science. 2019 Jan 2:60(1):168-175. doi: 10.1167/iovs.18-25453. Epub
[PubMed PMID: 30640969]