[1]
Taylor PN, Albrecht D, Scholz A, Gutierrez-Buey G, Lazarus JH, Dayan CM, Okosieme OE. Global epidemiology of hyperthyroidism and hypothyroidism. Nature reviews. Endocrinology. 2018 May:14(5):301-316. doi: 10.1038/nrendo.2018.18. Epub 2018 Mar 23
[PubMed PMID: 29569622]
[2]
Garber JR, Cobin RH, Gharib H, Hennessey JV, Klein I, Mechanick JI, Pessah-Pollack R, Singer PA, Woeber KA, American Association Of Clinical Endocrinologists And American Thyroid Association Taskforce On Hypothyroidism In Adults. Clinical practice guidelines for hypothyroidism in adults: cosponsored by the American Association of Clinical Endocrinologists and the American Thyroid Association. Thyroid : official journal of the American Thyroid Association. 2012 Dec:22(12):1200-35. doi: 10.1089/thy.2012.0205. Epub 2012 Nov 6
[PubMed PMID: 22954017]
Level 1 (high-level) evidence
[3]
Jonklaas J, Bianco AC, Bauer AJ, Burman KD, Cappola AR, Celi FS, Cooper DS, Kim BW, Peeters RP, Rosenthal MS, Sawka AM, American Thyroid Association Task Force on Thyroid Hormone Replacement. Guidelines for the treatment of hypothyroidism: prepared by the american thyroid association task force on thyroid hormone replacement. Thyroid : official journal of the American Thyroid Association. 2014 Dec:24(12):1670-751. doi: 10.1089/thy.2014.0028. Epub
[PubMed PMID: 25266247]
[4]
Peng CC, Pearce EN. An update on thyroid disorders in the postpartum period. Journal of endocrinological investigation. 2022 Aug:45(8):1497-1506. doi: 10.1007/s40618-022-01762-1. Epub 2022 Feb 18
[PubMed PMID: 35181848]
[5]
Phowira J, Coffey KL, Bartholomew PH, Vennart N, Moreira M, Emerson H, Kennedy D, Weaver JU. Radioactive Iodine for the Treatment of Subclinical Thyrotoxicosis Grade 1 and 2: Outcome of up to 18-Year Follow Up. Frontiers in endocrinology. 2022:13():843857. doi: 10.3389/fendo.2022.843857. Epub 2022 Mar 9
[PubMed PMID: 35370990]
[6]
Xing YZ, Zhang K, Jin G. Predictive factors for the outcomes of Graves' disease patients with radioactive iodine (131I) treatment. Bioscience reports. 2020 Jan 31:40(1):. doi: 10.1042/BSR20191609. Epub
[PubMed PMID: 31840740]
[7]
Paparella R, Menghi M, Micangeli G, Leonardi L, Profeta G, Tarani F, Petrella C, Ferraguti G, Fiore M, Tarani L. Autoimmune Polyendocrine Syndromes in the Pediatric Age. Children (Basel, Switzerland). 2023 Mar 19:10(3):. doi: 10.3390/children10030588. Epub 2023 Mar 19
[PubMed PMID: 36980146]
[8]
Bjørklund G, Pivin M, Hangan T, Yurkovskaya O, Pivina L. Autoimmune polyendocrine syndrome type 1: Clinical manifestations, pathogenetic features, and management approach. Autoimmunity reviews. 2022 Aug:21(8):103135. doi: 10.1016/j.autrev.2022.103135. Epub 2022 Jun 9
[PubMed PMID: 35690244]
[10]
El Sabbagh R, Azar NS, Eid AA, Azar ST. Thyroid Dysfunctions Due to Immune Checkpoint Inhibitors: A Review. International journal of general medicine. 2020:13():1003-1009. doi: 10.2147/IJGM.S261433. Epub 2020 Nov 4
[PubMed PMID: 33177863]
[11]
Muir CA, Clifton-Bligh RJ, Long GV, Scolyer RA, Lo SN, Carlino MS, Tsang VHM, Menzies AM. Thyroid Immune-related Adverse Events Following Immune Checkpoint Inhibitor Treatment. The Journal of clinical endocrinology and metabolism. 2021 Aug 18:106(9):e3704-e3713. doi: 10.1210/clinem/dgab263. Epub
[PubMed PMID: 33878162]
[12]
Pappa T, Refetoff S. Resistance to Thyroid Hormone Beta: A Focused Review. Frontiers in endocrinology. 2021:12():656551. doi: 10.3389/fendo.2021.656551. Epub 2021 Mar 31
[PubMed PMID: 33868182]
[13]
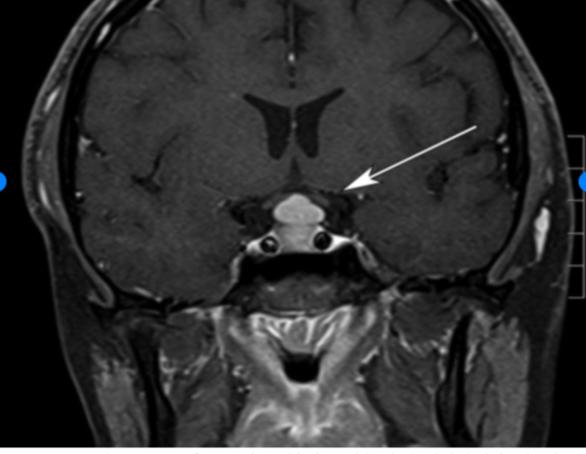
Ju JS, Cui T, Zhao J, Chen JL, Ju HB. Clinical presentation and magnetic resonance imaging characteristics of lymphocytic hypophysitis: a systematic review with meta-analysis. Archives of medical science : AMS. 2023:19(4):976-986. doi: 10.5114/aoms/144628. Epub 2021 Dec 14
[PubMed PMID: 37560735]
Level 1 (high-level) evidence
[14]
Khoury T, Kadah A, Mari A, Sbeit W, Drori A, Mahamid M. Thyroid Dysfunction is Prevalent in Autoimmune Hepatitis: A Case Control Study. The Israel Medical Association journal : IMAJ. 2020 Feb:22(2):100-103
[PubMed PMID: 32043327]
Level 2 (mid-level) evidence
[15]
Hollowell JG, Staehling NW, Flanders WD, Hannon WH, Gunter EW, Spencer CA, Braverman LE. Serum TSH, T(4), and thyroid antibodies in the United States population (1988 to 1994): National Health and Nutrition Examination Survey (NHANES III). The Journal of clinical endocrinology and metabolism. 2002 Feb:87(2):489-99
[PubMed PMID: 11836274]
Level 3 (low-level) evidence
[16]
Feldt-Rasmussen U, Effraimidis G, Klose M. The hypothalamus-pituitary-thyroid (HPT)-axis and its role in physiology and pathophysiology of other hypothalamus-pituitary functions. Molecular and cellular endocrinology. 2021 Apr 5:525():111173. doi: 10.1016/j.mce.2021.111173. Epub 2021 Feb 4
[PubMed PMID: 33549603]
[17]
Anand A, Singh KR, Kushwaha JK, Hussain N, Sonkar AA. Papillary Thyroid Cancer and Hashimoto's Thyroiditis: An Association Less Understood. Indian journal of surgical oncology. 2014 Sep:5(3):199-204. doi: 10.1007/s13193-014-0325-4. Epub 2014 Jul 4
[PubMed PMID: 25419066]
[18]
Hou J, Yu P, Zhu H, Pan H, Li N, Yang H, Jiang Y, Wang L, Wang B, Wang Y, You L, Chen S. The impact of maternal hypothyroidism during pregnancy on neonatal outcomes: a systematic review and meta-analysis. Gynecological endocrinology : the official journal of the International Society of Gynecological Endocrinology. 2016:32(1):9-13. doi: 10.3109/09513590.2015.1104296. Epub 2015 Nov 3
[PubMed PMID: 26527131]
Level 2 (mid-level) evidence
[19]
Samuels MH. Psychiatric and cognitive manifestations of hypothyroidism. Current opinion in endocrinology, diabetes, and obesity. 2014 Oct:21(5):377-83. doi: 10.1097/MED.0000000000000089. Epub
[PubMed PMID: 25122491]
Level 3 (low-level) evidence
[20]
Zare-Khormizi MR, Rahmanian M, Pourrajab F, Akbarnia S. Massive pericardial effusion and rhabdomyolysis secondary to untreated severe hypothyroidism: the first report. Acta clinica Belgica. 2014 Oct:69(5):375-8. doi: 10.1179/2295333714Y.0000000049. Epub 2014 Jul 24
[PubMed PMID: 25056490]
[21]
Khalid S, Asad-Ur-Rahman F, Abbass A, Gordon D, Abusaada K. Myxedema Ascites: A Rare Presentation of Uncontrolled Hypothyroidism. Cureus. 2016 Dec 5:8(12):e912. doi: 10.7759/cureus.912. Epub 2016 Dec 5
[PubMed PMID: 28083456]
[22]
Saini V, Yadav A, Arora MK, Arora S, Singh R, Bhattacharjee J. Correlation of creatinine with TSH levels in overt hypothyroidism - a requirement for monitoring of renal function in hypothyroid patients? Clinical biochemistry. 2012 Feb:45(3):212-4. doi: 10.1016/j.clinbiochem.2011.10.012. Epub 2011 Oct 28
[PubMed PMID: 22061337]
[24]
Barlow BT, Roberts RJ, Newman K, Harrison SK, Sin JH. Economic Evaluation of a Pharmacist-Led 5-Day Therapeutic Hold of IV Levothyroxine at an Academic Medical Center. Hospital pharmacy. 2022 Feb:57(1):20-25. doi: 10.1177/0018578720970457. Epub 2020 Nov 9
[PubMed PMID: 35521003]
[25]
Antonelli A, Elia G, Ragusa F, Paparo SR, Cavallini G, Benvenga S, Ferrari SM, Fallahi P. The Stability of TSH, and Thyroid Hormones, in Patients Treated With Tablet, or Liquid Levo-Thyroxine. Frontiers in endocrinology. 2021:12():633587. doi: 10.3389/fendo.2021.633587. Epub 2021 Mar 10
[PubMed PMID: 33790863]
[26]
Trimboli P, Scappaticcio L, De Bellis A, Maiorino MI, Knappe L, Esposito K, Bellastella G, Giovanella L. Different Formulations of Levothyroxine for Treating Hypothyroidism: A Real-Life Study. International journal of endocrinology. 2020:2020():4524759. doi: 10.1155/2020/4524759. Epub 2020 Jan 20
[PubMed PMID: 32184819]
[27]
Laurent I, Tang S, Astère M, Wang KR, Deng S, Xiao L, Li QF. Liquid L-thyroxine versus tablet L-thyroxine in patients on L- thyroxine replacement or suppressive therapy: a meta-analysis. Endocrine. 2018 Jul:61(1):28-35. doi: 10.1007/s12020-018-1574-8. Epub 2018 Mar 23
[PubMed PMID: 29572710]
Level 1 (high-level) evidence
[28]
Trimboli P, Virili C, Centanni M, Giovanella L. Thyroxine Treatment With Softgel Capsule Formulation: Usefulness in Hypothyroid Patients Without Malabsorption. Frontiers in endocrinology. 2018:9():118. doi: 10.3389/fendo.2018.00118. Epub 2018 Mar 21
[PubMed PMID: 29619010]
[29]
Cappelli C, Negro R, Pirola I, Gandossi E, Agosti B, Castellano M. Levothyroxine liquid solution versus tablet form for replacement treatment in pregnant women. Gynecological endocrinology : the official journal of the International Society of Gynecological Endocrinology. 2016:32(4):290-2. doi: 10.3109/09513590.2015.1113518. Epub 2015 Nov 20
[PubMed PMID: 26585420]
Level 2 (mid-level) evidence
[30]
Benvenga S. Liquid and softgel capsules of l-thyroxine results lower serum thyrotropin levels more than tablet formulations in hypothyroid patients. Journal of clinical & translational endocrinology. 2019 Dec:18():100204. doi: 10.1016/j.jcte.2019.100204. Epub 2019 Aug 6
[PubMed PMID: 31844631]
[31]
Gonzales KM, Stan MN, Morris JC 3rd, Bernet V, Castro MR. The Levothyroxine Absorption Test: A Four-Year Experience (2015-2018) at The Mayo Clinic. Thyroid : official journal of the American Thyroid Association. 2019 Dec:29(12):1734-1742. doi: 10.1089/thy.2019.0256. Epub 2019 Dec 4
[PubMed PMID: 31680654]
[32]
Ghosh S, Pramanik S, Biswas K, Bhattacharjee K, Sarkar R, Chowdhury S, Mukhopadhyay P. Levothyroxine Absorption Test to Differentiate Pseudomalabsorption from True Malabsorption. European thyroid journal. 2020 Jan:9(1):19-24. doi: 10.1159/000504218. Epub 2019 Nov 20
[PubMed PMID: 32071898]
[33]
Caron P, Declèves X. The Use of Levothyroxine Absorption Tests in Clinical Practice. The Journal of clinical endocrinology and metabolism. 2023 Jul 14:108(8):1875-1888. doi: 10.1210/clinem/dgad132. Epub
[PubMed PMID: 36916146]
[34]
Ettleson MD, Bianco AC. Individualized Therapy for Hypothyroidism: Is T4 Enough for Everyone? The Journal of clinical endocrinology and metabolism. 2020 Sep 1:105(9):e3090-104. doi: 10.1210/clinem/dgaa430. Epub
[PubMed PMID: 32614450]
[35]
Hennessey JV, Espaillat R. Current evidence for the treatment of hypothyroidism with levothyroxine/levotriiodothyronine combination therapy versus levothyroxine monotherapy. International journal of clinical practice. 2018 Feb:72(2):. doi: 10.1111/ijcp.13062. Epub 2018 Jan 30
[PubMed PMID: 29381251]
[36]
Guglielmi R, Frasoldati A, Zini M, Grimaldi F, Gharib H, Garber JR, Papini E. ITALIAN ASSOCIATION OF CLINICAL ENDOCRINOLOGISTS STATEMENT-REPLACEMENT THERAPY FOR PRIMARY HYPOTHYROIDISM: A BRIEF GUIDE FOR CLINICAL PRACTICE. Endocrine practice : official journal of the American College of Endocrinology and the American Association of Clinical Endocrinologists. 2016 Nov:22(11):1319-1326
[PubMed PMID: 27482609]
[37]
Abdullatif HD, Ashraf AP. Reversible subclinical hypothyroidism in the presence of adrenal insufficiency. Endocrine practice : official journal of the American College of Endocrinology and the American Association of Clinical Endocrinologists. 2006 Sep-Oct:12(5):572
[PubMed PMID: 17002934]
[38]
Mathew V, Misgar RA, Ghosh S, Mukhopadhyay P, Roychowdhury P, Pandit K, Mukhopadhyay S, Chowdhury S. Myxedema coma: a new look into an old crisis. Journal of thyroid research. 2011:2011():493462. doi: 10.4061/2011/493462. Epub 2011 Sep 15
[PubMed PMID: 21941682]