Continuing Education Activity
Hypospadias is an anatomical congenital malformation of the male external genitalia. It is characterized by abnormal development of the urethral fold and the ventral foreskin of the penis that causes abnormal positioning of the urethral opening. In hypospadias, the external urethral meatus may present various degrees of malpositioning and may be found with associated penile curvature. Depending on the location of the defect, patients may have an additional genitourinary malformation. This activity reviews the evaluation and treatment of hypospadias and highlights the role of the interprofessional team in evaluating and treating this condition.
Objectives:
- Describe the embryological abnormalities that lead to hypospadias.
- Outline the classification of hypospadias.
- Identify the interventions required once a patient has been identified with hypospadias.
- Describe the importance of prompt identification of hyspospadias in order to coordinate the appropriate timing for referral, imaging, and surgical interventions.
Introduction
Hypospadias is an anatomical congenital malformation of the male external genitalia. It is characterized by abnormal development of the urethral fold and the ventral foreskin of the penis that causes abnormal positioning of the urethral opening. [1]
In hypospadias, the external urethral meatus may present various degrees of malpositioning and may be found with associated penile curvature. Depending on the location of the defect, patients may have an additional genitourinary malformation. [2][3]
Recent advances have made it possible for children less than 12 months to undergo a single-stage repair with relatively good outcomes.
Etiology
The exact etiology of hypospadias is unknown but is believed to include genetic, endocrine, and environmental factors.[4]
Inheritance is believed to be polygenic, and it has been noticed more often in males with a family history of hypospadias.
Hypospadias is commonly seen in males with decreased androgens or with receptors that have decreased sensitivity to androgens. Recent studies also suggest that in-utero exposure to estrogens found in pesticides used in fruits and vegetables as well as in plastic linings can have an antiandrogenic activity.[5]
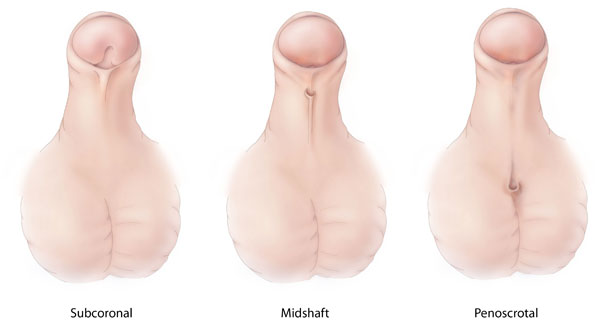
Hypospadias are classified by the location of the abnormal urethral meatus. One of the most commonly used classifications is as follows:
- Anterior (subcoronal and glandular)
- Middle (distal penile, proximal penile and midshaft)
- Posterior (scrotal, penoscrotal and perineal)
In nearly 50% of cases, the location is anterior, 20% of cases have a middle location and the rest are posterior. Overall, the subcoronal position is the most common abnormal location.
Epidemiology
Hypospadias is the second most common congenital disorder in males after cryptorchidism, but it is the most common penile congenital malformation.
The incidence of hypospadias in the US has been reported to be one out of every 250 males (0.4%) while in Denmark the estimated prevalence is 0.5% to 0.8%. A South American study estimated the global prevalence at 11.3 of 10,000 newborns (less than 0.1%). [6][7]
Pathophysiology
The main pathophysiological event for the development of hypospadias is the anomalous or partial urethral closure in the first weeks of embryonal development. External genitalia development occurs in two phases. The first phase, which happens between the fifth and eighth weeks of gestation, is characterized by the formation of the primordial genitalia in the absence of hormonal stimulation. In this phase, the cloacal folds are formed from mesodermal cells that are aligned laterally to the cloacal membrane. These folds fuse anteriorly and form the structure called genital tubercle (GT), and posteriorly they split into urogenital folds that surround the urogenital sinus and anal folds. The GT has three layers of cells: the lateral plate mesoderm, surface face ectoderm, and endodermal urethral epithelium. The latter is the main signaling center for the development, differentiation, and outgrowth of the GT.
The second phase, which is a hormone-dependent stage is initiated with the differentiation of gonads into testes in males with chromosomes XY. The testosterone synthesized in the testes has two very important functions: GT elongation and the appearance of the urethral groove. The distal portion of the urethral groove, which is called the urethral plate, is defined laterally by the urethral folds and outspreads into the glans penis. The urethra is eventually formed once the urethral folds are fused, and the skin of the penis is formed from the outermost layer of ectodermal cells, which fuses into the ventral aspect of the phallus and forms the median raphe.
Any genetic disruption or alteration in the signaling pathways in the male external genital development and urethral growth may develop different malformation which includes hypospadias, chordee (abnormal curvature of the penis), or abnormal penile foreskin formation. [2]
History and Physical
A thorough history and physical exam are necessary including a family history of hypospadias. Today, the diagnosis of hypospadias is made soon after birth. Key features include a glandular groove and a dorsal hood of the foreskin but in almost all cases the prepuce is incomplete ventrally. In addition, the urethral meatus is usually in an abnormal location. If the infant has a complete foreskin, the hypospadias may become obvious after circumcision. Any hypospadias discovered on circumcision should immediately be referred to a urologist; the circumcision should be halted.
Some patients may have an associated penile curvature which may be obvious during an erection.
Evaluation
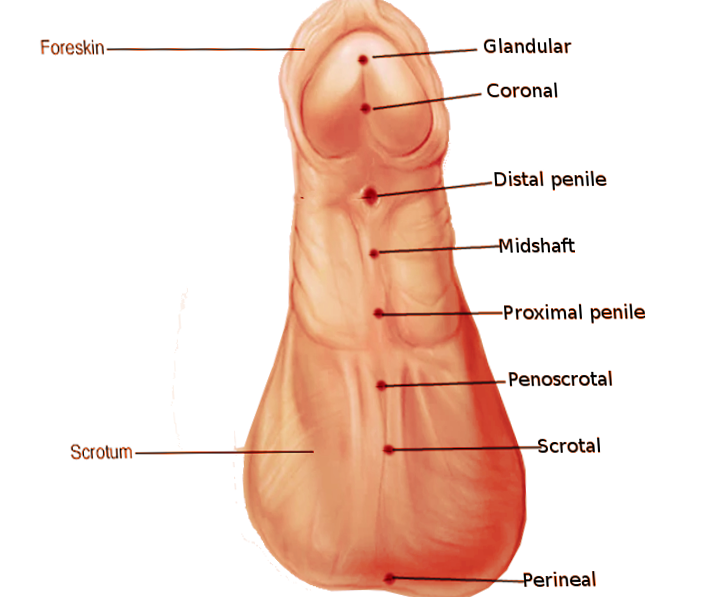
The classification of the hypospadias has changed throughout time. The classification most commonly used, divides hypospadias as follows: distal, midshaft, proximal.
- Distal hypospadias is the most common (60% to 70%). It is also called anterior or minor and includes glandular and sub-coronal hypospadias.
- Midshaft hypospadias, also called penile, includes distal, midshaft and proximal hypospadias.
- Posterior hypospadias includes peno-scrotal, scrotal, and perineal hypospadias.
Despite the location of the meatus, patients may have a hooded foreskin, which is described as abundant foreskin in the dorsal aspect of the penis and absent or scant in the ventral surface of the penis.[3]
In mild hypospadias, the urinary stream is usually not affected, but in more severe forms, the meatus may be narrowed, and the angle of the stream may be angled downward. Patients with abnormal angulation of the penis may have painful erections, compromised fertility with abnormal ejaculation, and in some cases inability of penetration during intercourse.
Hypospadias in most patients present as an isolated malformation, but in some cases, it can present with additional genito-urinary malformations. The most common malformation associated with proximal hypospadias is cryptorchidism (8% to 10% of cases), and inguinal hernias (9% to 15%). Other genito-urinary abnormalities such as vesicoureteral reflux, ureteropelvic junction obstruction, pelvic or horseshoe kidney, crossed renal ectopia, and renal agenesis is most commonly seen in proximal hypospadias but may be seen in a small percentage of the patients affected with distal hypospadias. [4]
Hypospadias is an associated finding in up to 200 different syndromes. These syndromes include WAGR syndrome or complex (Wilms tumor, aniridia, genitourinary anomalies, a range of intellectual abilities), Denys-Drash, Smith-Lemli-Opitz, Wolff-Hirschhorn, CHARGE (coloboma, heart defect, atresia choanae, retarded growth, genital anomalies, ear abnormalities).[2][4]
Treatment / Management
Patients diagnosed with hypospadias should be referred for surgical evaluation within the first weeks of life. If parents want circumcisions for their newborns, the presence of any penile abnormality should contraindicate the procedure, given that the foreskin is used in the arthroplasties.
Proximal hypospadias is usually associated with additional genito-urinary malformations. One of the first interventions is to order a karyotype, if there is associated cryptorchidism of one or both testes, to rule out an intersex condition. In this situation, patients require renal, bladder, and an abdominal ultrasound to assess internal organs and or associated malformations.
Midshaft and distal hypospadias once diagnosed should prompt surgical evaluation but do not require imaging or additional workup, given that this patient has the same risk of having renal anomalies as the general population.
The main objectives of surgical correction of hypospadias are to straighten the penis with an adequate caliber of the meatus to give an appearance resembling a typical circumcised penis or a penis with a foreskin and an adequate cosmetic result from the repair.
Surgical correction is the mainstay treatment of proximal hypospadias. In proximal penile, penoscrotal, and scrotal hypospadias with chordee, the patient commonly undergoes a two-stage repair with initial surgery consisting of urethroplasty and penile straightening with subsequent urethral plate tabularization. [3][6]
In some cases, patients before surgical repair undergo systemic treatment with testosterone derivatives to increase the penile size and allow better tubularization and avoid or decrease dehiscence, although there are no specific guidelines for the use of testosterone.
Surgery is recommended when the patient is between 6 and 18 months of life to limit psychological stress and behavioral problems that have been seen in patients who undergo the treatment later in life. [3]
Late repairs are usually associated with more complications including urethrocutaneous fistulas. The repair of hypospadias is complex and involves many steps.
Differential Diagnosis
- Genital anomalies
- Disorders of sex development
Prognosis
The prognosis of hypospadias has improved significantly over the past 3 decades. Earlier treatments were fraught with poor technical skills, limited appreciation of the anatomy and poor surgical timing. Studies from the past 20 years indicate that the short term results are good but the long term results are guarded. Many adults continue to have some type of psychological issue with the cosmesis and others continue to have unsatisfactory sex lives.
Newer techniques to repair hypospadias continue to evolve; the availability of tissue glues and laser devices has led to better and faster wound healing. But is should not be forgotten that this condition does have a significant psychological impact on the person and long term followup is essential.
Complications
The older the age, the higher the rate of complications after surgery. Hypospadias is best repaired by a team that has experience in dealing with this deformity. Immediately after surgery, edema and minor blood spotting are common. Some cases of bleeding may require a return to the operating room. Infections are rare. In the long term, urethrocutaneous fistulas are a major concern; they are far less common when the surgery is done in a single stage. These fistulas rarely close spontaneously and usually require a skin flap. However, it is vital to avoid hair-bearing skin as a flap because it may result in recurrent urinary tract infections and a nidus for stone formation. Even after repair, recurrence of the fistula is common. Other complications including meatal stenosis, urethral strictures, urethral diverticulum, and erectile dysfunction.
Enhancing Healthcare Team Outcomes
Once the primary care provider, obstetrician, pediatrician or nurse practitioner identifies hypospadias, the patient must be referred to a urologist. The condition should generally be treated within the first few weeks of life. In some cases, the infant may have other congenital anomalies that may need workup. The outlook for hypospadias after surgical correction is good. Most infants have function use of the penis and the deformity is reversed. However, repeat surgery is also needed in a number of infants. [3]
It is also recommended that the patient who underwent hypospadias repair in prepubertal age be seen after puberty to assess overall genital development. [8]
A significant number of adults continue to have emotional conflicts regarding the cosmesis and functionality of the penis, especially during sex. Thus, a mental health nurse should counsel these patients. Finally, hypospadias is best repaired by a dedicated team of urologist with experience in managing this anomaly. A team approach is vital to help improve outcomes.