Continuing Education Activity
Epistaxis (nosebleed) is one of the most common ear, nose, and throat emergencies in the emergency room or primary care. There are 2 types of nosebleeds: anterior (more common) and posterior (less common but more likely to require medical attention). The source of 90% of anterior nosebleeds lies within Kiesselbach's plexus (also known as Little's area) on the anterior nasal septum. This activity reviews the cause, pathophysiology, and presentation of epistaxis and highlights the role of the interprofessional team in its management.
Objectives:
Identify the causes of epistaxis.
Identify the common anatomic locations of epistaxis.
Differentiate the treatment options for epistaxis.
Communicate the importance of improving care coordination among interprofessional team members to improve outcomes for patients affected by epistaxis.
Introduction
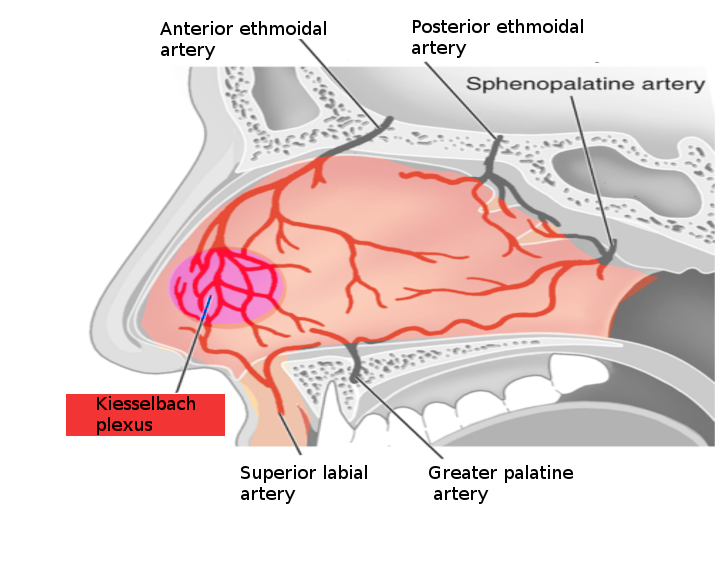
Epistaxis (nosebleed) is one of the most common ear, nose, and throat (ENT) emergencies in the emergency department or the primary care clinic. There are 2 types of nosebleeds: anterior (more common) and posterior (less common but more likely to require medical attention). The source of 90% of anterior nosebleeds is within Kiesselbach's plexus (also known as Little's area) on the anterior nasal septum. There are 5 named vessels whose terminal branches supply the nasal cavity.
- Anterior ethmoidal artery
- Posterior ethmoidal artery
- Sphenopalatine artery
- Greater palatine artery
- Superior labial artery
These 5 vessels' watershed areas are in the anterior nasal septum, comprising Kiesselbach's plexus. This lies at the entrance to the nasal cavity and is subject to extremes of heat, cold, and high and low moisture; it is easily traumatized. The mucosa over the septum in this area is fragile, making this the site of most epistaxis. More rarely, posterior or superior nasal cavity vessels bleed, leading to the so-called "posterior" epistaxis. This is more common in patients on anticoagulants, patients who are hypertensive, and patients with underlying blood dyscrasia or vascular abnormalities. Management depends on the severity of the bleeding and the patient's concurrent medical problems.[1][2][3] See Image. Nosebleed Vessels.
Etiology
There are multiple causes of epistaxis, which can be divided into local, systemic, environmental, and medication-induced.
Local Causes
Local causes of epistaxis include:
- Digital manipulation
- Deviated septum
- Trauma
- Chronic nasal cannula use
Systemic Causes
Local causes of epistaxis include:
- Alcoholism
- Hypertension
- Vascular malformations
- Coagulopathies (von Willebrand disease, hemophilia)
Environmental Factors
Environmental factors in epistaxis include:
- Allergies
- Environmental dryness ( more common in winter months)
Medications
Medications that can cause epistaxis include:
- Nonsteroidal anti-inflammatories (NSAIDs) (ibuprofen, naproxen, aspirin)
- Anticoagulants (warfarin)
- Platelet aggregation inhibitors (clopidogrel)
- Topical nasal steroid sprays
- Supplements or alternative medications (vitamin E, ginkgo, ginseng)
- Illicit drugs (cocaine)
While epistaxis is a widespread spontaneous problem, rarer etiologies such as neoplasms or vascular malformations must always be in the differential diagnosis, particularly if additional symptoms such as unilateral nasal obstruction, pain, or other cranial nerve deficits are noted.[4][5][6]
Epidemiology
Nosebleeds are rarely fatal, accounting for only 4 of the 2.4 million deaths in the United States. About 60% of people have experienced a nosebleed, and only 10% are severe enough to warrant treatment or medical intervention. They occur most commonly in children aged 2 to 10 years old and older people ranging from 50 to 80 years old.
Pathophysiology
The rupture of a blood vessel within the nasal mucosa causes nosebleeds. Rupture can be spontaneous, initiated by trauma, use of certain medications, and secondary to other comorbidities or malignancies. An increase in the patient's blood pressure can increase the episode's length. Anticoagulant medications, as well as clotting disorders, can also increase the bleeding time.
Most nosebleeds occur in the anterior part of the nose (Kiesselbach's plexus), and an etiologic vessel can usually be found on careful nasal examination. Bleeding from the posterior or superior nasal cavity is often termed a posterior nosebleed. This is usually presumed due to bleeding from Woodruff's plexus, the rear, and fine terminal branches of the sphenopalatine and posterior ethmoidal arteries. These are often difficult to control and are associated with bleeding from both nostrils or into the nasopharynx, where it is swallowed or coughed up, presenting as hemoptysis. It can generate a greater blood flow into the posterior pharynx and have a higher risk for airway compromise or aspiration due to increased difficulty controlling the bleed.
History and Physical
The history should include duration, severity, frequency, laterality of the bleeding, inciting event, and interventions provided before seeking care. Inquire about anticoagulants, aspirin, NSAIDs, and topical nasal steroid use. Obtain a relevant family history, particularly relating to coagulopathy and vascular/collagen disease, as well as any history of drug and alcohol use.
Prepare proper equipment and proper personal protective equipment (PPE) before beginning the physical examination. Equipment may include a nasal speculum, bayonet forceps, headlamp, suction catheter, packing, silver nitrate swabs, cotton pledgets, topical vasoconstrictor, and anesthetic. Have the patient seated in an exam chair in a room with suction available. Carefully insert the speculum and slowly open the blades to visualize the bleeding site. A headlight is essential for hands-free illumination, and the clot may need to be suctioned from the nasal cavity to identify the bleeding source.
A posterior nosebleed is not easy to visualize and may be suggested by active bleeding into the posterior pharynx without a visualized vessel on nasal examination. Nasal endoscopy dramatically increases the success in identifying the bleeding source.
Evaluation
Differentiating an anterior or posterior is critical in management. Diagnosis of anterior bleeding can be made by direct visualization using a nasal speculum and light source. A topical spray with anesthetic and epinephrine may be helpful for vasoconstriction to help control bleeding and to aid in the visualization of the source. Usually, posterior bleeding is diagnosed after measures to control anterior bleeding have failed. Clinical features of posterior bleeding can include active bleeding into the posterior pharynx without an identified anterior source; high-flow posterior bleeds may cause blood to emanate from both nares. Labs may be obtained if necessary, including a complete blood cell count, type and cross match, and coagulation studies, though they should not delay treatment of an active bleed. Imaging such as X-rays or computed tomography have no role in the urgent or emergent management of active epistaxis.
Treatment / Management
Start with a primary survey and address the airway, ensuring the airway is patent. Next, assess for hemodynamic compromise. Obtain large-bore intravenous access in patients with severe bleeding and obtain labs. Reverse blood clotting as necessary if there is a concern with medication use.[7][8][9] All patients with moderate to severe nose bleeding should have 2 large-bore intravenous lines and an infusion of crystalloid. The monitoring of oxygen and hemodynamic stability is vital.
Treatment for anterior bleeding can be started with direct pressure for at least 10 minutes. Have the patient apply constant direct pressure by pinching the nose over the cartilaginous tip (instead of over the bony areas) for a few minutes to control the bleeding. Vasoconstrictors such as oxymetazoline or thrombogenic foams or gels can be employed if that is ineffective. It is essential to remove all clots with suction before any attempt at treatment is made. The reasons are twofold:
- Clot prevents any medication from reaching the vessel
- If packing becomes necessary, the clot can be pushed into the nasopharynx and aspirated.
If topical treatments are unsuccessful, proceed with nasal examination to identify and cauterize the vessel with silver nitrate. If this, too, is unsuccessful, anterior nasal packing is necessary. This can be performed with absorbable packing material such as surgical or fibrillar or with devices such as anterior epistaxis balloons or nasal tampons (Rapid Rhino). If silver nitrate is used to cauterize a septal blood vessel, only use it on 1 side of the septum to prevent septal perforation. Thermal coagulation is painful and should rarely be attempted in an emergent setting.
Traditional petrolatum gauze can be used if one does not have access to balloons or tampons. If none of this is successful, the bleeding may be from the posterior or superior nasal cavity. Symptoms can include active bleeding from both nostrils or active bleeding present in the posterior pharynx. Longer (7.5cm) nasal tampons provide more posterior pressure and can be employed in this situation. Formal posterior nasal packing should only be performed by experienced personnel as it requires admission, telemetry monitoring, and sometimes intubation. It is associated with higher rates of complications like pressure necrosis, infection, or hypoxia. It may trigger a nasal-cardiac reflex (ie, sudden bradycardia after nasal packing); if this occurs, remove the pack immediately). Foley catheters can be used by experienced personnel to tamponade a posterior bleed. If a rear pack is placed, a formal petrolatum gauze anterior pack must also be set to create a closed, tamponaded space in the nasopharynx.
If these measures are unsuccessful, the patient should be intubated for airway protection, and interventional radiology should be consulted emergently for embolization. If this service is unavailable, an otolaryngologist can perform operative ligation of the sphenopalatine and ethmoid arteries in the operating room.
Differential Diagnosis
The differential diagnoses for epistaxis include the following:
- Nasal tumor
- DIC
- Hemophilia
- Von Willebrand disease
- Rhinitis
- Foreign body in the nose
- Drug toxicity (warfarin, NSAIDs)
Postoperative and Rehabilitation Care
Once the bleeding is controlled, arranging a timely follow-up (within 1 week) with their primary care physician or an otolaryngologist is essential. If any packing has been placed, this must remain undisturbed for 3 to 5 days before removal. Patients should begin an antistaphylococcal antibiotic to prevent toxic shock syndrome. Underlying causes must be addressed before discharge (tight blood pressure control with goal systolic blood pressure <120 mm Hg, several of any coagulopathy, etc), and patients should use topical nasal saline in both nares to keep the packs moist and facilitate removal.
Pearls and Other Issues
Patients with anterior nosebleeds can be discharged if the bleeding is controlled, hemodynamicc stability is observed for at least 1 hour in the emergency department and all predisposing factors are medically optimized. Follow-up with an otolaryngologist or their primary physician should occur in 1 week, and they should begin nasal saline 3 times daily. If nonbiodegradable packing is used, patients should return to the emergency department or ENT for packing removal in 3 to 5 days. If a patient, including pediatric patients, requires posterior packing, admission is required to monitor for complications, particularly cardiac arrhythmias. All anticoagulants should ideally be discontinued but must be reversed or withheld to achieve the lowest dose acceptable if discontinuation is impossible.
Applying topical saline sprays or ointments to the nasal mucosa to ensure moisturization of the nasal mucosa can help prevent recurrent epistaxis. Patients should also be advised to avoid hot foods, strenuous activity, blowing the nose, or digital manipulation of the nose on discharge.
Enhancing Healthcare Team Outcomes
An interprofessional team best performs the care of nose bleeding. Most patients initially present to the emergency room, and the triage nurse should be fully aware of the importance of admitting patients with significant bleeding. While most anterior nosebleeds can be arrested with digital pressure, a follow-up appointment is recommended in patients with repeat episodes. Even though nurses may not perform invasive procedures to stop the bleeding, they can effectively instruct patients to properly compress the nose with their fingers, which in most cases can arrest the bleeding.
Nasal packing is another option, but the packing must be in place for 3 to 5 days, and repeated insertions and removals of various packs only exacerbate the bleeding (see Image. Epistaxis Management Supplies). Drug-induced nosebleeds may require a reversal of the INR and admission. The pharmacist should ensure that the patient does not restart the NSAID or other anticoagulant while the bleeding is active. A hematologist consult is recommended to deal with patients with coagulopathy. In rare cases, embolization or cauterization may be required to stop a nosebleed. An ENT consultation is necessary if the bleeding is posterior and severe.
In some cases, the invasive radiologist may be required to perform embolization to stop the bleeding. Nurses should monitor the oxygen and hemodynamic status of all patients with moderate to severe nose bleeds. These patients should have intravenous access to the transfusion of crystalloids. The team members should communicate to ensure that the patient receives an acceptable standard of care treatment.