Continuing Education Activity
Decorticate and decerebrate posturing are abnormal posturing responses typically to noxious stimuli. They involve stereotypical movements of the trunk and extremities. To avoid the high morbidity and mortality associated with these conditions, it must be promptly diagnosed and treated. This activity reviews the assessment and management of decorticate and decerebrate posturing and highlights the role of the interprofessional team in evaluating and treating patients with this condition.
Objectives:
- Identify the physical exam findings associated with decorticate and decerebrate posturing.
- Describe the pathophysiology of decorticate and decerebrate posturing.
- Outline the prognosis of patients with decorticate and decerebrate posturing.
- Review the etiology of decorticate and decerebrate posturing.
Introduction
Decorticate and decerebrate posturing are both considered pathological posturing responses to usually noxious stimuli from an external or internal source. Both involve stereotypical movements of the trunk and extremities and are typically indicative of significant brain or spinal injury.[1] The Nobel Laurette Charles Sherrington first described decerebrate posturing in 1898 after transecting the brainstems of live monkeys and cats.[2]
Synonymous terms for decorticate posturing include abnormal flexion, decorticate rigidity, flexor posturing, or decorticate response. Synonymous terms for decerebrate posturing include abnormal extension, decerebrate rigidity, extensor posturing, or decerebrate response.[2]
There is a criticism within the literature of the use of the terms decorticate and decerebrate posturing in clinical contexts due to their association with discrete anatomical locations that, in reality, may not be so prescriptive. Brain lesions of several anatomical regions may cause both postures, though they do usually involve some degree of brainstem injury.[1] It is, however, accepted that decorticate typically requires an injury more rostral than decerebrate posturing. In most literature, this level is considered the red nucleus at the intercollicular level of the midbrain.[2][3]
Etiology
There are numerous causes of abnormal posturing including supratentorial and infratentorial lesions, alongside more diffuse pathologies such as metabolic and infective causes:[4][1]
Supratentorial Lesions
- Abscess
- Extra-axial hematoma: subdural and extradural
- Hydrocephalus
- Intracerebral hemorrhage
- Raised intracranial pressure
- Traumatic brain injury including diffuse axonal injury
- Tumor
Infratentorial Lesion
- Abscess
- Hydrocephalus
- Infarct: brainstem or bilateral diencephalic
- Intracerebral hemorrhage: cerebellar or brainstem
- Traumatic brain injury including diffuse axonal injury
- Tumor
Diffuse and Metabolic
- Cerebral malaria[5]
- Electrolyte abnormalities: hyponatremia, hypomagnesemia, hypocalcemia
- Encephalitis
- Hepatic encephalopathy
- Hypoxic brain injury
- Hypoglycemia
- Lead poisoning
- Meningitis
- Reye syndrome
In patients with preexisting structural lesions of the central nervous system, episodes of decerebrate posturing can occur in response to numerous physiological factors including, but not exclusive to, fever, hypoxia; metabolic disturbance; sensory irritation; hypoglycemia; and meningeal irritation.[6]
Epidemiology
Data on the incidence of abnormal posturing is not published. However, the incidence of pathologies that commonly cause decorticate and decerebrate posturing is available. The commonest cause of decorticate and decerebrate posturing is traumatic brain injury (TBI).[4] A 2019 systematic review estimates there to be 69 million TBI worldwide each year, with 7.95% being classed as severe. Severe TBI is defined as patients with a GCS of 8 or less.[7]
Pathophysiology
Typically, the anatomical divide associated with decorticate and decerebrate posturing is the intercollicular line at the level of the red nucleus. However, this concept has been criticized as lesions in the supratentorial region can also cause both decorticate and decerebrate posturing, though the brainstem is typically involved.[1]
Both posturing types are stereotypical; however, they can be present in varying degrees and even asymmetrically with decorticate on one side and decerebrate contralaterally. An inconsistent presentation of a given posture tends to suggest a lesser degree of injury.[1]
Decerebrate Posturing
Decerebrate posturing has been demonstrated in monkeys with a transecting lesion at the level between the superior and inferior colliculi within the midbrain. This is also described as being at or below the level of the red nucleus. Sherrington performed live animal studies where anesthetized monkeys had their cerebral cortex removed from the intercollicular level upwards. The animals continued to breathe independently and developed stereotypical extension rigidity of the extremities. [2] A study with intercollicular transection showed that extensor posturing only occurred in the context of either noxious stimuli, passive hyperextension of the head, or metabolic disturbance such as hypoxia.[6]
Decerebrate posturing can be seen in patients with large bilateral forebrain lesions with progression caudally into the diencephalon and midbrain. It can also be caused by a posterior fossa lesion compressing the midbrain or rostral pons. Though decerebrate posturing implies a destructive structural lesion, it can also be caused by reversible metabolic disturbances such as hypoglycemia and hepatic encephalopathy.[1]
Through animal models and human studies, it has been shown that the vestibulospinal tract plays a major role in decerebrate posturing. The vestibulospinal pathways have an excitatory effect on extensor motor neurons in the spine, while inhibition of flexor motor neurons. The vestibular nuclei receive input from the vestibular apparatus and spinal somatosensory pathways while receiving modulatory signals from the cerebral cortex and the fastigial nucleus of the cerebellum. In isolation, the vestibular nucleus, via the vestibulospinal tract, causes activation of extensor motor neurons in the spinal cord and inhibition of flexor motor neurons. However, under normal physiology, the higher brain centers of the cortex and cerebellum inhibit the vestibular nuclei, thus preventing this reflex. Decerebrate posturing results from a disconnection between the modulatory higher centers and the vestibular nuclei, resulting in unsuppressed extensor posturing. [8]
Decorticate Posturing
The mechanism for decorticate posturing is not as well studied as that of decerebrate. Phylogenetically, the region of the red nucleus within the midbrain plays a significant part in locomotion. In primates, the rubrospinal tract influences primitive grasp reflexes, particularly in infants and is, incidentally, responsible for crawling.[9] The rubrospinal tract carries signals from the red nucleus to the spinal motor neurons. Primates are reliant on fine motor skills, and therefore the motor cortex via the corticospinal tracts is more prominent in movement than phylogenetically lower regions. Extensive lesions involving the forebrain, diencephalon, or rostral midbrain are known to cause decorticate posturing. This includes the motor cortex, premotor cortex, corona radiata, internal capsule, and thalamus.[3][1] In primates, the rubrospinal tract descends as far as the thoracic spine, it, therefore, has effects on the upper limbs but not lower. The red nucleus, via the rubrospinal tract, causes a flexion, grasping type reflex of the upper limbs. The higher brain centers, such as the cerebral cortex, inhibit this reflex during normal physiology. With a lesion of the corticospinal tract, the red nucleus is disinhibited, and the flexion reflex of the upper limbs is unimpeded. The vestibulospinal tracts, as discussed above, are also left disinhibited, and extension of the lower limbs occurs. This flexion of the upper limbs and extension of lower limbs is decorticate posturing. [3]
The red nucleus is anatomically at the intercollicular level, and thus lesions above the red nucleus tend to cause decortication and lesions below, decerebration.[3][8] As compression advances from the regions of the forebrain and diencephalon to the brainstem, abnormal posturing can progress from decorticate to decerebrate.[1]
History and Physical
Teasdale et al., in their original paper on the Glasgow coma scale, suggested that distinguishing between normal and abnormal flexion requires an experienced observer, and thus, simply defining the response as either flexion or extension may be sufficient in the early stages following brain insult.[10]
Decorticate Posturing
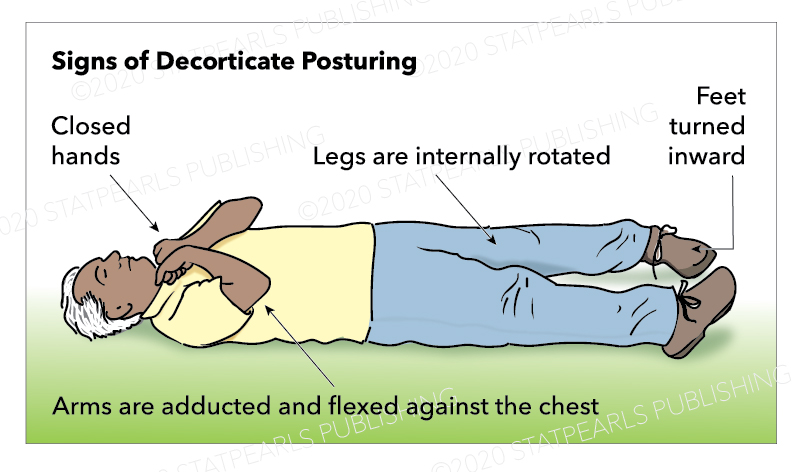
Decorticate posturing is described as abnormal flexion of the arms with the extension of the legs. Specifically, it involves slow flexion of the elbow, wrist, and fingers with adduction and internal rotation at the shoulder. The lower limbs show extension and internal rotation at the hip, with the extension of the knee and plantar flexion of the feet. Toes are typically abducted and hyperextended.[1]
Decerebrate Posturing
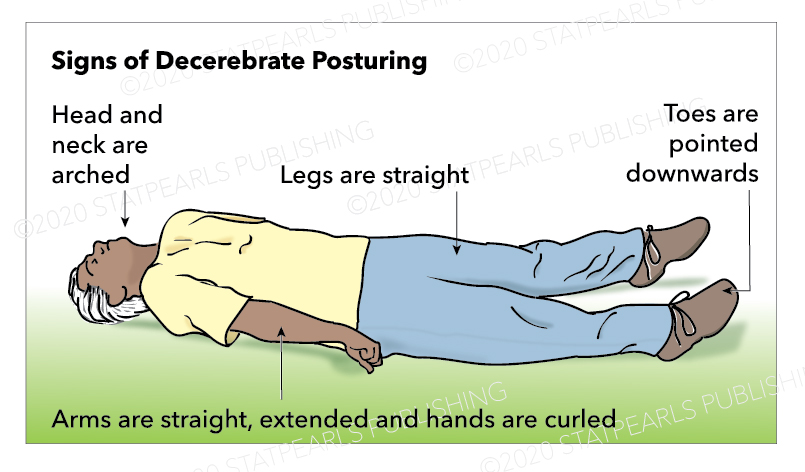
Decerebrate posturing is described as adduction and internal rotation of the shoulder, extension at the elbows with pronation of the forearm, and flexion of the fingers. As with decorticate posturing, the lower limbs show extension and internal rotation at the hip, with the extension of the knee and plantar flexion of the feet. Toes are typically abducted and hyperextended.[1] Teasdale and Jennett advocated not using the term 'decerebrate' in the assessment of coma due to its association with a specific physioanatomical correlation, but to rather use the term 'extension.'[10]
Stages of Brain Herniation
In central herniation, the neurological status of a patient can be seen to progress through a series of stages. Starting with the diencephalic stage, phases proceed through: obeying commands, bilateral Babinski reflexes, paratonia, grasp reflex, decortication. Later as the midbrain and upper pons are involved, the patient becomes decerebrated. As the lower pons and upper medulla are involved, the patient becomes flaccid.[1]
Evaluation
Decorticate and decerebrate posturing are clinical diagnoses and should be considered a pathological sign of a neurological injury rather than a diagnosis unto themselves. Due to the variety of etiologies that can cause these findings, numerous investigations may be appropriate. As discussed above, compressive lesions may benefit from surgical intervention, best done in a timely manner; therefore, early intracranial imaging such as CT or MRI is warranted.[11] Other causes, such as infection and metabolic disturbance, can be investigated with laboratory tests, including serum and CSF sampling.[4][1][5]
Treatment / Management
Treatment is directed at the underlying cause, for instance, correcting, where possible, metabolic derangements, and treating infections.[5] In TBI, evacuation of extra-axial hematoma can improve survival.[11][12] Some pathologies may not be reversible, such as hypoxic brain injury, and thus supportive approaches are taken.[13]
Differential Diagnosis
Normal flexion, also known as withdrawal to pain, can appear similar to decorticate posturing. However, normal flexion is a rapid and variable movement compared with the slow and stereotypical movements of abnormal flexion. In normal flexion, the shoulder abducts away from the body, the wrist is either neutral or extended, and importantly, there is no extension in the lower limbs. In abnormal flexion, the shoulder adducts, and internal rotates, the wrist flexes with forearm pronation. A key feature is the extension of the lower limbs.[1][14]
Spinal reflexes may still be present following brain death. One study found motor spinal responses present in 22% of patients with confirmed brain death. These can be mistaken for abnormal posturing movements such as decerebration.[15]
Opisthotonus is extra-pyramidal abnormal posturing seen in cerebral palsy, tetanus, drowning victims, TBI, and poisoning. It is abnormal posturing with a dramatically arched back and neck.[16] Paratonia, or gegenhalten, can also look like abnormal posturing and is typically caused by encephalopathy or neurodegenerative conditions.[1]
Lastly, patients with corticospinal tract injuries, such as ischaemic strokes, hemorrhages, or tumors, can develop spasticity. Usually in the upper limb flexor muscles dominate and extensor in the lower limb, similar to decorticate posturing. The most striking difference between spasticity and abnormal posturing is the preservation of consciousness in spastic patients.[8]
Prognosis
Abnormal posturing is an ominous sign, with only 37% of decorticate patients surviving following head injury and only 10% in decerebrate.[1][14] Overall, children requiring admission to hospital due to head injury have a mortality of 10% to 13%; however, in severe cases with decerebrate posturing, the mortality is 71%.[17] Other studies have shown similar mortality rates of 68% to 83% in TBI with decerebrate posturing.[11][18][12] Factors that favored survival in TBI with decerebrate posturing included younger patient age, admission within 6 hours of injury, and extradural hematoma. Poorer outcomes were found in acute subdural hematoma and older age.[11]
Outcomes in patients with abnormal posturing following hypoxic brain injury were reviewed in 210 patients. Abnormal posturing, or GCS motor score of less than 4, after 1-day post-insult, suggested virtually no chance of regaining independence.[13] Patients with gunshot wounds to the head who demonstrated either decorticate or decerebrate posturing had a close to zero chance of a meaningful recovery.[19] The Hunt and Hess subarachnoid hemorrhage severity grading scale includes decerebrate posturing in grades 4 and 5. They quoted 42% and 77% mortality, respectively.[20]
Complications
As discussed under prognosis, complications include death and poor functional neurological outcome. In the immediate period, patients with coma can also have problems maintaining their airway and controlling their cardiorespiratory system.[21][22]
Deterrence and Patient Education
Given the most common cause of abnormal posturing is TBI, public health measures to reduce traumatic events such as road traffic collisions and workplace accidents are important.[7]
Enhancing Healthcare Team Outcomes
Decorticate and decerebrate posturing are medical emergencies. Early diagnosis and intervention may improve survival and functional outcome.[11] Herniation syndromes, raised intracranial pressure (ICP), intracranial hemorrhage, TBI, and penetrating trauma may warrant emergent neurosurgical involvement.[1][19] Infective and metabolic causes require supportive therapies and the involvement of medical, neurological, or infectious disease specialists.[5] Patients with abnormal posturing likely require aggressive goal-directed therapies, and thus an intensive care environment may be appropriate.[22] Alongside this, medical therapies to reduce acutely raised intracranial pressure such as mannitol and hypertonic saline have level III evidence.[23] Level II evidence has shown hyperventilation may benefit raised ICP, though only as a temporizing measure and not prophylactically.[24] That said, given the often poor outcome in patients with abnormal posturing, particularly decerebration, aggressive treatment may be ethically questionable, and a case by case assessment is required.[1][14]