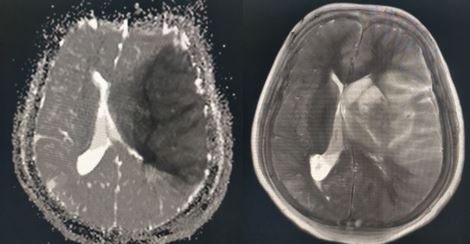
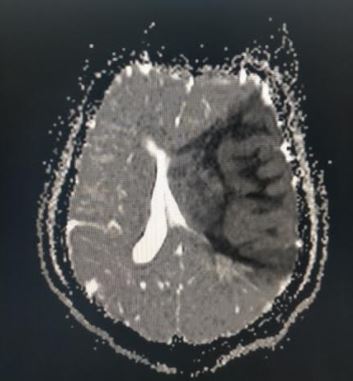
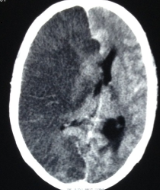
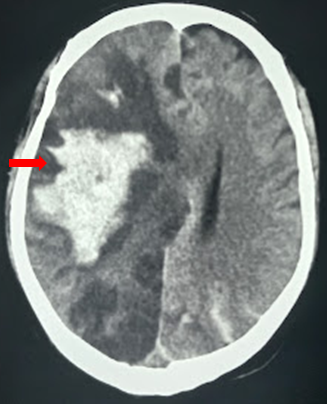
[1]
Li Y, Zhang E, Yuan H. Cerebral carbon dioxide embolism after kidney cancer laparoscopic surgery with full neurological recovery: A case report. Medicine. 2020 Jul 2:99(27):e20986. doi: 10.1097/MD.0000000000020986. Epub
[PubMed PMID: 32629713]
Level 3 (low-level) evidence
[2]
Zhou Y, Li W, Wang D, Mao L, Jin H, Li Y, Hong C, Chen S, Chang J, He Q, Wang M, Hu B. Clinical time course of COVID-19, its neurological manifestation and some thoughts on its management. Stroke and vascular neurology. 2020 Jun:5(2):177-179. doi: 10.1136/svn-2020-000398. Epub 2020 May 4
[PubMed PMID: 32366614]
[3]
Adams HP Jr, Bendixen BH, Kappelle LJ, Biller J, Love BB, Gordon DL, Marsh EE 3rd. Classification of subtype of acute ischemic stroke. Definitions for use in a multicenter clinical trial. TOAST. Trial of Org 10172 in Acute Stroke Treatment. Stroke. 1993 Jan:24(1):35-41
[PubMed PMID: 7678184]
Level 1 (high-level) evidence
[4]
Benjamin EJ, Muntner P, Alonso A, Bittencourt MS, Callaway CW, Carson AP, Chamberlain AM, Chang AR, Cheng S, Das SR, Delling FN, Djousse L, Elkind MSV, Ferguson JF, Fornage M, Jordan LC, Khan SS, Kissela BM, Knutson KL, Kwan TW, Lackland DT, Lewis TT, Lichtman JH, Longenecker CT, Loop MS, Lutsey PL, Martin SS, Matsushita K, Moran AE, Mussolino ME, O'Flaherty M, Pandey A, Perak AM, Rosamond WD, Roth GA, Sampson UKA, Satou GM, Schroeder EB, Shah SH, Spartano NL, Stokes A, Tirschwell DL, Tsao CW, Turakhia MP, VanWagner LB, Wilkins JT, Wong SS, Virani SS, American Heart Association Council on Epidemiology and Prevention Statistics Committee and Stroke Statistics Subcommittee. Heart Disease and Stroke Statistics-2019 Update: A Report From the American Heart Association. Circulation. 2019 Mar 5:139(10):e56-e528. doi: 10.1161/CIR.0000000000000659. Epub
[PubMed PMID: 30700139]
[5]
Formisano L, Guida N, Mascolo L, Serani A, Laudati G, Pizzorusso V, Annunziato L. Transcriptional and epigenetic regulation of ncx1 and ncx3 in the brain. Cell calcium. 2020 May:87():102194. doi: 10.1016/j.ceca.2020.102194. Epub 2020 Mar 7
[PubMed PMID: 32172011]
[6]
Lee JM, Grabb MC, Zipfel GJ, Choi DW. Brain tissue responses to ischemia. The Journal of clinical investigation. 2000 Sep:106(6):723-31
[PubMed PMID: 10995780]
[7]
Takase KI. Cardiogenic cerebral infarction in the parietal lobe predicts the development of post-stroke epilepsy. Seizure. 2020 Aug:80():196-200. doi: 10.1016/j.seizure.2020.06.018. Epub 2020 Jun 20
[PubMed PMID: 32623354]
Level 3 (low-level) evidence
[8]
Kwah LK, Diong J. National Institutes of Health Stroke Scale (NIHSS). Journal of physiotherapy. 2014 Mar:60(1):61. doi: 10.1016/j.jphys.2013.12.012. Epub 2014 May 3
[PubMed PMID: 24856948]
[9]
Byrne D, Walsh JP, Sugrue G, Nicolaou S, Rohr A. CT Imaging of Acute Ischemic Stroke [Formula: see text]. Canadian Association of Radiologists journal = Journal l'Association canadienne des radiologistes. 2020 Aug:71(3):266-280. doi: 10.1177/0846537120902068. Epub 2020 Mar 11
[PubMed PMID: 32157894]
[10]
Albers GW, Lansberg MG, Kemp S, Tsai JP, Lavori P, Christensen S, Mlynash M, Kim S, Hamilton S, Yeatts SD, Palesch Y, Bammer R, Broderick J, Marks MP. A multicenter randomized controlled trial of endovascular therapy following imaging evaluation for ischemic stroke (DEFUSE 3). International journal of stroke : official journal of the International Stroke Society. 2017 Oct:12(8):896-905. doi: 10.1177/1747493017701147. Epub 2017 Mar 24
[PubMed PMID: 28946832]
Level 1 (high-level) evidence
[11]
Atchaneeyasakul K, Shang T, Haussen D, Ortiz G, Yavagal D. Impact of MRI Selection on Triage of Endovascular Therapy in Acute Ischemic Stroke: The MRI in Acute Management of Ischemic Stroke (MIAMIS) Registry. Interventional neurology. 2020 Jan:8(2-6):135-143. doi: 10.1159/000490580. Epub 2019 Mar 15
[PubMed PMID: 32508895]
[12]
Silva GS, Nogueira RG. Endovascular Treatment of Acute Ischemic Stroke. Continuum (Minneapolis, Minn.). 2020 Apr:26(2):310-331. doi: 10.1212/CON.0000000000000852. Epub
[PubMed PMID: 32224754]
[13]
Ramakrishnan TCR, Kumaravelu S, Narayan SK, Buddha SS, Murali C, Majeed PHA, Meenakshi-Sundaram S, Wadia RS, Sharma V, Basu I, Vijaya P, Salam KA, Barmare S, Vaid Z, Nirmal Raj KK, Wattamwar PR, Asokan K, Dhonge V, Nellikunja S, Namjoshi D, Srinivasa R, Laddhad DS, Deshpande SD, Raghunath B, Kalita J, Kumar M, Misra UK, Pradeep M, Tenecteplase in Stroke Investigators. Efficacy and Safety of Intravenous Tenecteplase Bolus in Acute Ischemic Stroke: Results of Two Open-Label, Multicenter Trials. American journal of cardiovascular drugs : drugs, devices, and other interventions. 2018 Oct:18(5):387-395. doi: 10.1007/s40256-018-0284-1. Epub
[PubMed PMID: 29948822]
[14]
Cronin CA. Intravenous tissue plasminogen activator for stroke: a review of the ECASS III results in relation to prior clinical trials. The Journal of emergency medicine. 2010 Jan:38(1):99-105. doi: 10.1016/j.jemermed.2009.08.004. Epub 2009 Sep 17
[PubMed PMID: 19765940]
[15]
Pan Y, Elm JJ, Li H, Easton JD, Wang Y, Farrant M, Meng X, Kim AS, Zhao X, Meurer WJ, Liu L, Dietrich D, Wang Y, Johnston SC. Outcomes Associated With Clopidogrel-Aspirin Use in Minor Stroke or Transient Ischemic Attack: A Pooled Analysis of Clopidogrel in High-Risk Patients With Acute Non-Disabling Cerebrovascular Events (CHANCE) and Platelet-Oriented Inhibition in New TIA and Minor Ischemic Stroke (POINT) Trials. JAMA neurology. 2019 Dec 1:76(12):1466-1473. doi: 10.1001/jamaneurol.2019.2531. Epub
[PubMed PMID: 31424481]
[16]
Powers WJ, Rabinstein AA, Ackerson T, Adeoye OM, Bambakidis NC, Becker K, Biller J, Brown M, Demaerschalk BM, Hoh B, Jauch EC, Kidwell CS, Leslie-Mazwi TM, Ovbiagele B, Scott PA, Sheth KN, Southerland AM, Summers DV, Tirschwell DL. Guidelines for the Early Management of Patients With Acute Ischemic Stroke: 2019 Update to the 2018 Guidelines for the Early Management of Acute Ischemic Stroke: A Guideline for Healthcare Professionals From the American Heart Association/American Stroke Association. Stroke. 2019 Dec:50(12):e344-e418. doi: 10.1161/STR.0000000000000211. Epub 2019 Oct 30
[PubMed PMID: 31662037]
[17]
Casula M, Fortuni F, Fabris F, Leonardi S, Gnecchi M, Sanzo A, Greco A, Rordorf R. Direct oral Xa inhibitors versus warfarin in patients with cancer and atrial fibrillation: a meta-analysis. Journal of cardiovascular medicine (Hagerstown, Md.). 2020 Aug:21(8):570-576. doi: 10.2459/JCM.0000000000001041. Epub
[PubMed PMID: 32628422]
[18]
Liljehult J, Christensen T, Molsted S, Overgaard D, Mesot Liljehult M, Møller T. Effect and efficacy of lifestyle interventions as secondary prevention. Acta neurologica Scandinavica. 2020 Oct:142(4):299-313. doi: 10.1111/ane.13308. Epub 2020 Jul 15
[PubMed PMID: 32620044]
[19]
Béjot Y, Duloquin G, Crespy V, Durier J, Garnier L, Graber M, Giroud M. Influence of Preexisting Cognitive Impairment on Clinical Severity of Ischemic Stroke: The Dijon Stroke Registry. Stroke. 2020 Jun:51(6):1667-1673. doi: 10.1161/STROKEAHA.119.028845. Epub 2020 May 13
[PubMed PMID: 32397931]
[20]
Kwah LK, Harvey LA, Diong J, Herbert RD. Models containing age and NIHSS predict recovery of ambulation and upper limb function six months after stroke: an observational study. Journal of physiotherapy. 2013 Sep:59(3):189-97. doi: 10.1016/S1836-9553(13)70183-8. Epub
[PubMed PMID: 23896334]
Level 2 (mid-level) evidence
[21]
Pan J, Konstas AA, Bateman B, Ortolano GA, Pile-Spellman J. Reperfusion injury following cerebral ischemia: pathophysiology, MR imaging, and potential therapies. Neuroradiology. 2007 Feb:49(2):93-102
[PubMed PMID: 17177065]
[22]
Katsanos AH, Lioutas VA, Charidimou A, Catanese L, Ng KKH, Perera K, de Sa Boasquevisque D, Tsivgoulis G, Smith EE, Sharma M, Selim MH, Shoamanesh A. Statin treatment and accrual of covert cerebral ischaemia on neuroimaging: a systematic review and meta-analysis of randomized trials. European journal of neurology. 2020 Jun:27(6):1023-1027. doi: 10.1111/ene.14196. Epub 2020 Mar 23
[PubMed PMID: 32133735]
Level 1 (high-level) evidence
[23]
Sussman ES, Jin M, Pendharkar AV, Pulli B, Feng A, Heit JJ, Telischak NA. Dual antiplatelet therapy after carotid artery stenting: trends and outcomes in a large national database. Journal of neurointerventional surgery. 2021 Jan:13(1):8-13. doi: 10.1136/neurintsurg-2020-016008. Epub 2020 May 15
[PubMed PMID: 32414894]
[24]
Fassbender K, Walter S, Grunwald IQ, Merzou F, Mathur S, Lesmeister M, Liu Y, Bertsch T, Grotta JC. Prehospital stroke management in the thrombectomy era. The Lancet. Neurology. 2020 Jul:19(7):601-610. doi: 10.1016/S1474-4422(20)30102-2. Epub
[PubMed PMID: 32562685]
[25]
Fatima N, Saqqur M, Hussain MS, Shuaib A. Mobile stroke unit versus standard medical care in the management of patients with acute stroke: A systematic review and meta-analysis. International journal of stroke : official journal of the International Stroke Society. 2020 Aug:15(6):595-608. doi: 10.1177/1747493020929964. Epub 2020 Jun 9
[PubMed PMID: 32515695]
Level 1 (high-level) evidence
[26]
Darehed D, Blom M, Glader EL, Niklasson J, Norrving B, Eriksson M. In-Hospital Delays in Stroke Thrombolysis: Every Minute Counts. Stroke. 2020 Aug:51(8):2536-2539. doi: 10.1161/STROKEAHA.120.029468. Epub 2020 Jun 26
[PubMed PMID: 32586222]
[27]
Glader EL, Stegmayr B, Johansson L, Hulter-Asberg K, Wester PO. Differences in long-term outcome between patients treated in stroke units and in general wards: a 2-year follow-up of stroke patients in sweden. Stroke. 2001 Sep:32(9):2124-30
[PubMed PMID: 11546906]
[28]
Krespi Y,Gurol ME,Coban O,Tuncay R,Bahar S, Stroke unit versus neurology ward--a before and after study. Journal of neurology. 2003 Nov
[PubMed PMID: 14648154]
[29]
Jauch EC, Saver JL, Adams HP Jr, Bruno A, Connors JJ, Demaerschalk BM, Khatri P, McMullan PW Jr, Qureshi AI, Rosenfield K, Scott PA, Summers DR, Wang DZ, Wintermark M, Yonas H, American Heart Association Stroke Council, Council on Cardiovascular Nursing, Council on Peripheral Vascular Disease, Council on Clinical Cardiology. Guidelines for the early management of patients with acute ischemic stroke: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2013 Mar:44(3):870-947. doi: 10.1161/STR.0b013e318284056a. Epub 2013 Jan 31
[PubMed PMID: 23370205]