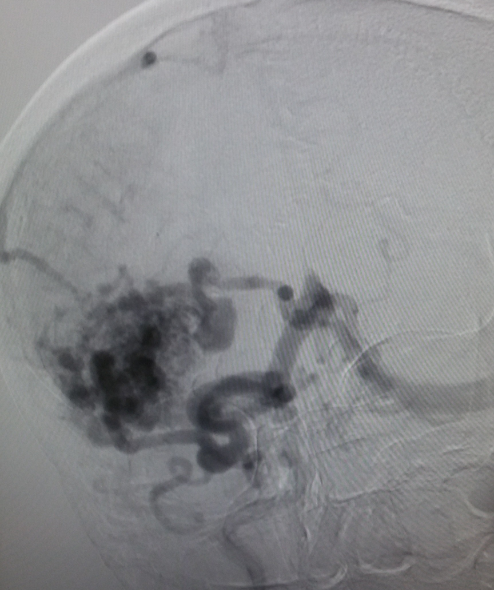
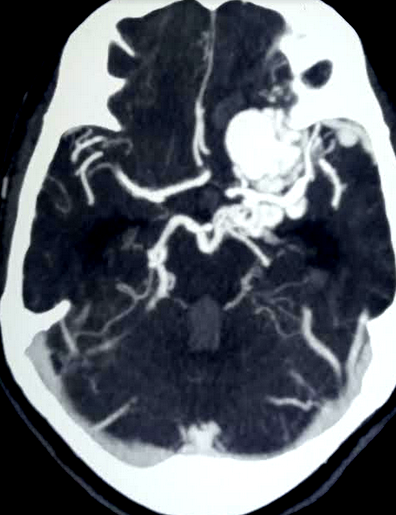
[1]
Claro E, Dias A, Girithari G, Massano A, Duarte MA. Non-traumatic Hematomyelia: A Rare Finding in Clinical Practice. European journal of case reports in internal medicine. 2018:5(11):000961. doi: 10.12890/2018_000961. Epub 2018 Nov 28
[PubMed PMID: 30755987]
Level 3 (low-level) evidence
[2]
Wu EM, El Ahmadieh TY, McDougall CM, Aoun SG, Mehta N, Neeley OJ, Plitt A, Shen Ban V, Sillero R, White JA, Batjer HH, Welch BG. Embolization of brain arteriovenous malformations with intent to cure: a systematic review. Journal of neurosurgery. 2019 Feb 1:132(2):388-399. doi: 10.3171/2018.10.JNS181791. Epub
[PubMed PMID: 30717053]
Level 1 (high-level) evidence
[3]
Xu H, Wang L, Guan S, Li D, Quan T. Embolization of brain arteriovenous malformations with the diluted Onyx technique: initial experience. Neuroradiology. 2019 Apr:61(4):471-478. doi: 10.1007/s00234-019-02176-2. Epub 2019 Feb 2
[PubMed PMID: 30712140]
[4]
Heit JJ, Thakur NH, Iv M, Fischbein NJ, Wintermark M, Dodd RL, Steinberg GK, Chang SD, Kapadia KB, Zaharchuk G. Arterial-spin labeling MRI identifies residual cerebral arteriovenous malformation following stereotactic radiosurgery treatment. Journal of neuroradiology = Journal de neuroradiologie. 2020 Feb:47(1):13-19. doi: 10.1016/j.neurad.2018.12.004. Epub 2019 Jan 15
[PubMed PMID: 30658138]
[5]
Li W, Sun Q, Duan X, Yi F, Zhou Y, Hu Y, Yao L, Xu H, Zhou L. [Etiologies and risk factors for young people with intracerebral hemorrhage]. Zhong nan da xue xue bao. Yi xue ban = Journal of Central South University. Medical sciences. 2018 Nov 28:43(11):1246-1250. doi: 10.11817/j.issn.1672-7347.2018.11.013. Epub
[PubMed PMID: 30643071]
[6]
Caranfa JT, Baldwin MT, Rutter CE, Bulsara KR. Synchronous cerebral arteriovenous malformation and lung adenocarcinoma carcinoma brain metastases: A case study and literature review. Neuro-Chirurgie. 2019 Feb:65(1):36-39. doi: 10.1016/j.neuchi.2018.07.003. Epub 2019 Jan 9
[PubMed PMID: 30638546]
Level 3 (low-level) evidence
[7]
Khandelwal A, Chaturvedi A, Singh GP, Mishra RK. Intractable brain swelling during cerebral arteriovenous malformation surgery due to contralateral acute subdural haematoma. Indian journal of anaesthesia. 2018 Dec:62(12):984-987. doi: 10.4103/ija.IJA_491_18. Epub
[PubMed PMID: 30636801]
[8]
Hofman M, Jamróz T, Kołodziej I, Jaskólski J, Ignatowicz A, Jakutowicz I, Przybyłko N, Kocur D, Baron J. Cerebral arteriovenous malformations - usability of Spetzler-Martin and Spetzler-Ponce scales in qualification to endovascular embolisation and neurosurgical procedure. Polish journal of radiology. 2018:83():e243-e247. doi: 10.5114/pjr.2018.76750. Epub 2018 May 29
[PubMed PMID: 30627242]
[9]
Kocer N, Kandemirli SG, Dashti R, Kizilkilic O, Hanimoglu H, Sanus GZ, Tunali Y, Tureci E, Islak C, Kaynar MY. Single-stage planning for total cure of grade III-V brain arteriovenous malformations by embolization alone or in combination with microsurgical resection. Neuroradiology. 2019 Feb:61(2):195-205. doi: 10.1007/s00234-018-2140-z. Epub 2018 Nov 28
[PubMed PMID: 30488257]
[10]
Ironside N, Chen CJ, Ding D, Ilyas A, Kumar JS, Buell TJ, Taylor D, Lee CC, Sheehan JP. Seizure Outcomes After Radiosurgery for Cerebral Arteriovenous Malformations: An Updated Systematic Review and Meta-Analysis. World neurosurgery. 2018 Dec:120():550-562.e3. doi: 10.1016/j.wneu.2018.08.121. Epub 2018 Aug 24
[PubMed PMID: 30149174]
Level 1 (high-level) evidence
[11]
Mosimann PJ, Chapot R. Contemporary endovascular techniques for the curative treatment of cerebral arteriovenous malformations and review of neurointerventional outcomes. Journal of neurosurgical sciences. 2018 Aug:62(4):505-513. doi: 10.23736/S0390-5616.18.04421-1. Epub 2018 Mar 26
[PubMed PMID: 29582979]
[12]
Shovlin CL, Condliffe R, Donaldson JW, Kiely DG, Wort SJ, British Thoracic Society. British Thoracic Society Clinical Statement on Pulmonary Arteriovenous Malformations. Thorax. 2017 Dec:72(12):1154-1163. doi: 10.1136/thoraxjnl-2017-210764. Epub
[PubMed PMID: 29141890]
[13]
Cenzato M, Boccardi E, Beghi E, Vajkoczy P, Szikora I, Motti E, Regli L, Raabe A, Eliava S, Gruber A, Meling TR, Niemela M, Pasqualin A, Golanov A, Karlsson B, Kemeny A, Liscak R, Lippitz B, Radatz M, La Camera A, Chapot R, Islak C, Spelle L, Debernardi A, Agostoni E, Revay M, Morgan MK. European consensus conference on unruptured brain AVMs treatment (Supported by EANS, ESMINT, EGKS, and SINCH). Acta neurochirurgica. 2017 Jun:159(6):1059-1064. doi: 10.1007/s00701-017-3154-8. Epub 2017 Apr 7
[PubMed PMID: 28389875]
Level 3 (low-level) evidence
[14]
van Essen MJ, Han KS, Lo RTH, Woerdeman P, van der Zwan A, van Doormaal TPC. Functional and educational outcomes after treatment for intracranial arteriovenous malformations in children. Acta neurochirurgica. 2018 Nov:160(11):2199-2205. doi: 10.1007/s00701-018-3665-y. Epub 2018 Sep 7
[PubMed PMID: 30191363]