Continuing Education Activity
Similar to their adult counterparts, pediatric cerebral aneurysms are associated with a variety of systemic and intracranial disorders. The most common conditions associated with increased risk of pediatric cerebral aneurysm include coarctation of the aorta, polycystic kidney disease, fibromuscular dysplasia, tuberous sclerosis, Ehlers-Danlos syndrome, Marfan syndrome, and certain familial syndromes. This activity describes the evaluation, diagnosis, and management of pediatric cerebral aneurysms and highlights the role of team-based interprofessional care for affected patients.
Objectives:
- Describe a patient presentation for which pediatric cerebral aneurysm should be on the differential diagnosis.
- List the evaluation components for pediatric cerebral aneurysms.
- Depict considerations that influence the management of pediatric cerebral aneurysms.
- Examine the role of interprofessional team members in optimizing collaboration and communication to ensure children with cerebral aneurysms are recognized early and every effort is made to deliver high-quality care, which will lead to enhanced outcomes.
Introduction
One of the earliest recorded cases of a childhood aneurysm was a 15-year-old boy reported by the German pathologist Eppinger in 1871. Postmortem examination revealed stenosis of the aortic isthmus along with the unusual finding of free blood at the base of the brain arising from a ruptured saccular aneurysm of the right anterior cerebral artery. Edvard Bull subsequently described the first antemortem case of a 17-year-old girl who presented with a severe headache and third nerve palsy. The clinical diagnosis of a ruptured posterior communicating artery aneurysm that autopsy subsequently confirmed. Whereas “acquired” factors such as diabetes, alcohol, hypertension, high fat and cholesterol diets, obesity, cigarette consumption, and oral contraceptive use have correlations with subarachnoid hemorrhage in adults, such influences are minimal or altogether absent in children. Several authors have postulated the greater role of congenital factors in the development of childhood aneurysms.
Etiology
Idiopathic Aneurysms
Significant controversy exists as to the exact nature of idiopathic aneurysm formation. Whether aneurysm development is a question of nature versus nurture remains unanswered. Postmortem studies of the cerebral vasculature of aneurysm patients have revealed multiple defects in the media at multiple arterial bifurcations. Histological exam routinely demonstrates a fragmentation of both the internal elastic membrane and the muscularis layer of the media. Within the aneurysm pouch, the smooth muscle is generally absent, but the internal elastic membrane may continue for a variable length into the neck of the lesion. The basement membrane underneath the endothelium is also frequently thickened and reticulated. What remains within the wall is typically an endothelial lining, fibrous tissue, adventitia, inflammatory infiltrates, and hemorrhage of varying ages. Calcifications are also commonly encountered. The site of rupture is usually at the apex of the lesion with a small diverticulum often seen at the site of perforation. Most reported cases of pediatric aneurysm seem to indicate that the histology of childhood lesions resembles that of adult saccular aneurysms. [1][2][3]
Saccular aneurysms most often occur at the bifurcation of the internal carotid artery or one of its intermediate branches. Not only are these branching points physically more susceptible to injury as a result of high shear forces, but pathologic analysis has also demonstrated that the number of internal elastic membrane fenestrations is frequently also highest at these areas with an injury to the internal elastic lamina Usually the initial pathophysiological alteration subsequently resulting in aneurysm formation.
What is less clear is the exact mechanism leading to this fragmentation of the elastica. Whereas developmental defects of the muscularis layer of the media are a common finding in the cerebral arteries of normal patients, their frequency is still far greater than is the incidence of aneurysm formation. Crompton, in a summary of 149 autopsies, noted that the size and the number of these medial gaps appears to increase with age.
Aneurysms of childhood have correlated with various collagen vascular diseases and connective tissue disorders. The cerebral vasculature is distinguished from systemic vessels by the thinness of its walls and lack of perivascular support. Type I and type III collagen predominate in the intracranial circulation and provide the major load-bearing capabilities of the blood vessels. Approximately half of the adult aneurysm patients are lacking or deficient in type III collagen. Similar findings have also been observed in the aneurysm walls of young children. Østergaard has postulated that because type III collagen deficiency produces an increased distensibility of the vessel wall, the larger size of childhood aneurysms could be explained by the occurrence of a more compliant aneurysm wall lacking in such fibers.[4][5][6]
Another early theory proposed that childhood aneurysms arose from remnants of small vascular trunks stemming from arterial bifurcations. They believed that saccular aneurysms arose from the stumps of vessels that disappear when the fetal cerebrovascular network matures to form the adult system of major arterial trunks. Such vestigial or incompletely aborted nubbins could become progressively dilated to form aneurysms over time. Isolated reports have described children with aneurysms in which only a medial defect with an intact elastica was present. Nonetheless, substantial corroborative proof of this theory is to date lacking. Given both the histologic and clinical features of childhood aneurysms, it would seem an attractive hypothesis to assume that these lesions are mainly congenital. On the contrary, Stehbens in his careful analysis of the literature reviewed the age incidence, associated conditions, prevalence of hypertension and atherosclerosis, histology, and electron microscopic structure of childhood cases and concluded that a purely degenerative pathogenetic model could explain aneurysm formation in these children. Early damage from hypertension, birth injury, trauma, and in utero insults may play a significant role in such cases.[7]
Atheromatous degeneration and hemodynamic factors are thought to further weaken the vessels of congenitally predisposed patients. However, in our own experience and that of the literature, most children with intracranial aneurysms have little or no atheromatous changes near the aneurysm. The majority of children diagnosed with aneurysms are normotensive. However, childhood arterial hypertension does exist as secondary phenomena in children with aneurysms who also have an aortic coarctation or polycystic kidney disease. Though the coexistence of these processes with aneurysms may be simply fortuitous, the possibility that arterial hypertension may contribute to the formation of aneurysms in these children cannot be excluded.[8]
There are postulations that “intrinsic” defects might cause variable vessel wall dysfunction producing transient or permanent failure to repair subsequent “insults.” The growing body of literature supports the role of both “intrinsic defects” and acquired “insults” in the development of aneurysmal disease in children. The balance between these “defects” and “insults” and the ability of the vessel wall to repair and compensate for these changes may predict whether an aneurysm forms, grows, ruptures, thromboses or regresses.
Traumatic Aneurysms
In cases where a history of closed head injury is present, a traumatic injury to the vessel wall may lead to subsequent aneurysm formation. Such injury of the arterial wall may occur with increased frequency at certain locations within the cranium thus leading to the clinical observation that traumatic childhood aneurysms often occur at stereotypical sites. These include the anterior cerebral artery distribution and its distal branches resulting from injury along the falx, the posterior cerebral artery near the free edge of the tentorium cerebelli, and the middle cerebral artery along its course on the sphenoid ridge. Some authors have postulated a traumatic origin for many lesions in unusual locations characteristic of childhood cases. For example, an increased prevalence of P2 segment aneurysms has frequently been reported in children. Meyer et al. postulated that these lesions might develop during times of increased intracranial pressure early in development (i.e., during delivery) when herniation of the posterior cerebral artery over the tentorium could cause vessel injury. True traumatic aneurysms have only an intact layer of adventitia in their walls with the remaining normal structures undergoing disruption. False traumatic aneurysms result from a fibrous organization around a prior hematoma encasing the parent vessel. False lesions often develop after direct penetrating injury as in the case of depressed skull fractures or of penetrating injuries such as gunshot and stab wounds. The histology of the lesions ultimately has little bearing on the clinical management of such lesions.[9][10][11][12]
Infectious Aneurysms
Originally termed “mycotic” aneurysms by Sir William Osler in 1885, the majority of infectious childhood aneurysms result from bacterial rather than fungal infection, which is quite rare. Mielke in his comprehensive review of the literature up to 1981 found only 13 confirmed cases of fungal intracranial aneurysms. Aspergillus, Candida, and Phycomycetes species were, in descending order, most commonly implicated. With few exceptions, the patients were immunocompromised with a near one-hundred percent mortality rate after diagnosis of their aneurysm. Karsner in his analysis of infectious aneurysms classified primarily three forms. The first, being the most frequent, occurs in the setting of bacterial endocarditis with subsequent embolization; this is commonly described as the “intravascular” variety of an infectious aneurysm. The second type involves a direct extension from an adjacent nidus of infection. This extravascular source may be secondary to meningitis, cavernous sinus thrombophlebitis, adjacent osteomyelitis, sinus or postoperative infection. For such lesions, the anatomical location of the proximal carotid and other large proximal vessels at the base of the brain make them particularly vulnerable to injury. The third group requires the diagnosis of an infectious aneurysm with stereotypical radiologic and histologic findings in the face of no other obvious inflammatory lesion elsewhere.
Alpha Streptococcus, Staphylococcus, Pseudomonas, and Haemophilus species are encountered most frequently in children with bacterial intracranial aneurysms. The interval from the embolic event to the time of aneurysm formation can be surprisingly short with the entire course of embolism, local arteritis, aneurysm formation, and hemorrhage usually occurring within 2 days in Molinari’s experimental animal models. Necrosis and destruction proceeded from the adventitial surface to the media with the internal elastic membrane and intima being involved last.[13][14][15]
Epidemiology
Pediatric autopsy series reveals no incidental cases of cerebral aneurysms in 3000 pediatric autopsies.[16][17] The retrospective literature estimates an incidence of 3-4%. We suggest a rough estimate of 1 to 3 cases of a childhood aneurysm per 1 million population.[18][7][19]
Demographics and Location
Childhood aneurysms most often become symptomatic from birth to age 6 and then from 8 years to adolescence A peak in the first six months characterizes the early childhood group with the majority of cases occurring within the first two years of life. Early childhood cases have also had associations with a higher incidence of lesions localized along the middle cerebral artery and its distal branches and involving the vertebrobasilar system.
In adults, there is a 1.6 to 1 female predilection. The incidence is equal before age 40. In ages 70 to 87 years, there is a 4.16 to 1 female to male ratio. The ratio in childhood cases is believed to be 1.3 to 2.8:1 male to female ratio. The male to female ratio was 1.3 to 1 in our own series and 4 to 1 for our 13 cases under the age of 5. We found that 62% of giant aneurysms occurred in boys.
Carotid bifurcation aneurysms are found in 29 to 54% of all childhood cases, primarily in juvenile and adolescent cases, as compared to 4.4 to 4.5% of all aneurysms in adults. Carotid bifurcation aneurysms can also present in very young patients. The childhood data do not reflect the typical adult predilection for the anterior circulation (93%), anterior communicating artery (28%), and the posterior-communicating artery (25%).
Multiple aneurysms are not uncommon in the adult population and are observed in 10-20% of cases and are far less common in children occurring in only 4 to 5 percent of cases; this is likely the result of the relationship between the presence of hypertension and the formation of multiple intracranial aneurysms in adults. When multiple aneurysms are present in children, they usually associate with factors such as prior cranial irradiation, moyamoya disease, fibromuscular dysplasia, sickle cell disease, cardiac myxoma or arteriovenous malformations.
Pathophysiology
Similar to their adult counterparts, aneurysms of childhood have associations and links with a variety of systemic and intracranial disorders. Whereas some may represent purely coincidental occurrences, other conditions such as aortic coarctation are generally acknowledged as bearing an increased risk for a cerebral aneurysm. The most common of such conditions include coarctation of the aorta, polycystic renal disease, fibromuscular dysplasia, tuberous sclerosis, Ehlers-Danlos syndrome, Marfan syndrome, and familial syndromes. Aside from the more common arteriopathies already mentioned, both syphilis and pseudoxanthoma elasticum have also been involved in cases of childhood SAH. Azarelli reported the case of a child with a history of prior curative irradiation for intracranial germinoma who subsequently developed multiple aneurysms and expired from a ruptured fusiform lesion of the ICA. Moyamoya disease also has correlations with an increased risk of multiple childhood aneurysms.[20][21] As prior irradiation has been implicated in the development of Moyamoya syndrome, one could postulate that Azarelli’s case might have fit into this category. Other vascular structural anomalies include abnormal or persistent fetal patterns or vestigial remnants that frequently present in children with aneurysms.[22][23][24]
Intracranial aneurysms also correlate with various hematologic diseases in children. Several cases of infants and young children with maternally acquired human immunodeficiency virus (HIV) have developed aneurysms of the proximal circulation. Whether these lesions represent direct vessel wall injury from the retrovirus itself or associated infectious conditions remains unclear. There is an unusually high incidence of reports of multiple aneurysms in HIV cases. There are also observations of aneurysms associated with non-infectious hematological disorders such as thalassemia minor, sickle-cell disease, and glucose-6-phosphatase deficiency.
Isolated case reports of cerebral aneurysms in children with concurrent cerebral tumors are also present in the literature. From our current series, Zee reported a fascinating case of a young boy with a temporal MCA aneurysm encased in an anaplastic sarcoma of the temporal lobe. Along with several other authors, we have also observed a child whose aneurysm was diagnosed upon evaluation for hypopituitarism secondary to a pituitary adenoma. Reports also exist of patients with cardiac myxoma and multiple intracranial aneurysms.
Traumatic Aneurysms
Traumatic aneurysms typically develop from a direct penetrating injury or in association with a closed head injury. Classically, the location of these lesions in children is related to the direct path of the penetrating missile, to vascular suspensory regions of the brain, or in areas of vascular contiguity with dural margins. Peripheral cortical vessels are often directly involved below the edges of a fracture of the cranium. Ventureya subdivided traumatic aneurysms into four major groups. The first group represents lesions of the internal carotid artery which were sub-classified into cavernous, supraclinoid, and petrous types. These were the most commonly seen. Supraclinoid aneurysms were rare. The second group was comprised of lesions of the anterior cerebral artery circulation, typically distal to the anterior communicating artery. The third group included middle cerebral artery distribution aneurysms which were usually near or distal to its bifurcation. Finally, there were those aneurysms located near the vertebrobasilar confluence. Traumatic multiple aneurysms occurred with the least frequency.[25][26][10]
Giant Aneurysms
Saccular aneurysms exceeding 25 mm in their maximal diameter have traditionally been classified as “giant” in size. Whether these lesions result as a dynamic evolution from smaller lesions with an intermittent breakdown of the aneurysmal wall remains unclear. From aneurysmal series covering all age groups, giant lesions comprise approximately 2 to 5 percent of all cases. Giant aneurysms of children occur with far greater frequency. Several authors have reported remarkably high proportions of large or giant lesions in their series: Amacher & Drake 45%, Meyer & Sundt 54%, Gerosa et al. 20%, and Humphreys 28.5%. In a large review of 500 cases of childhood aneurysms, 20 percent were giant in nature. For aneurysms of children younger than five years of age, the proportion of large (50%) and giant (30%) aneurysms is even higher. Anecdotal cases of massive aneurysms in very young infants also are present in the literature. Although several of these series reflect the referral bias of their practice, the frequency of giant aneurysms does seem to increase with younger age in the literature.
It is also a known fact that adult giant intracranial aneurysms are different in their localizations as compared to that of smaller aneurysms. Approximately 40 percent present in the carotid distribution, 25 percent in the anterior and middle cerebral artery distribution, and 30 percent in the vertebrobasilar territory; this represents a six-fold increase in the proportion of posterior fossa lesions usually seen in the adult population. Giant intracranial aneurysms of children share this predilection for the posterior circulation but to a much greater degree. From Hacker’s review of 500 literature cases, he noted that “there is approximately an equal distribution between the anterior and posterior system” of giant aneurysms. Our series of 52 children included 12 giant aneurysms of which 6 (50%) were in the vertebrobasilar territory, and 8 (67%) were male. Whereas 60% of adult giant aneurysms are in women, 62% of giant childhood aneurysms occur in males.[27][28]
Coarctation of the aorta
As discussed earlier, one of the first descriptions of a ruptured pediatric aneurysm was that of a 15-year-old boy with a stenotic lesion of his aortic isthmus. As Matson noted, the relationship of aortic coarctation and ruptured cerebral aneurysms in children has been repeatedly echoed throughout the past century’s literature. In 1927. Postmortem examinations performed on 32 children with aortic coarctation implicated cerebral hemorrhage as the cause of death in 37.5% of cases. The following year, Abbott found that 7 of 20 cases with cerebral hemorrhage and aortic coarctation had a confirmed ruptured cerebral aneurysm on autopsy.
The actual incidence of cerebral aneurysms in children with aortic coarctation is difficult to estimate as the majority of afflicted children do not become symptomatic until later in life. Tyler and Clark in their review of 200 patients with aortic coarctation reported only five patients diagnosed in life with cerebral aneurysms. They reported an average age of rupture at 25 years from their literature review. Often, neurologic manifestations may provide the first clues towards the diagnosis of aortic coarctation. LeBlanc wrote, “in young, hypertensive, non-cyanosed patients, intermittent neurological symptoms or sudden cerebral insult may suggest the existence of aortic coarctation ” (80). He went on to confirm the association of aortic coarctation with ruptured cerebral aneurysm and also pointed out the predilection for multiple aneurysms in such patients. In patients with this clinical picture, a continuous murmur, arterial hypertension in the vasculature proximal to the stenosis, and diminished pulses with hypotension in the distal extremities should be sought.
Along with ruptured aneurysms, patients with coarctation may also suffer complications reduced to direct arterial hypertension and resultant intracerebral hemorrhage, emboli arising from associated aortic aneurysms or surgical sites with a resultant stroke, and cerebral abscess arising from infected emboli. Alleviating the aortic stenosis usually results in a reduction of the blood pressure proximal to the coarctation, and may reduce the risk of intracerebral hemorrhage and aneurysmal rupture. Some authors have even recommended surgery for a cerebral aneurysm prior to correction of the aortic coarctation in stable patients. Patel and Richardson reported 7 of their 58 aneurysm patients as having coarctation of the aorta. In our own series, we have encountered two children with aortic stenosis and symptomatic cerebral aneurysm. Whenever possible, we recommend a magnetic resonance angiogram (MRA) or spinal CT angiogram with three-dimensional reconstructions before adolescence for patients with aortic coarctation. Although less sensitive than standard arteriography, MRA and CT have been shown to be safe, noninvasive, and effective screening modalities for cerebral aneurysms. If normal, repeat surveillance scans are obtainable at the clinician’s discretion. For patients with clinical evidence of aneurysmal rupture or in cases where the MRA is questionable, four-vessel cerebral angiography is advisable. It is essential that an experienced neuroradiologist be available to perform specialized techniques such as brachial catheterization as severe stenoses may preclude standard femoral catheterization and patient age increases the potential complexity of catheter placement and contrast load.
Polycystic Kidney Disease
There are two distinct forms of polycystic renal disease. The first is an autosomal dominant form of the disorder that afflicts multiple organs including the heart, gastrointestinal tract, and kidneys. Affected patients also demonstrate an increased risk of harboring an intracranial aneurysm although reported prevalence rates vary from 0 to 41 percent. This broad range is due in part to the fact that the diagnosis of autosomal dominant polycystic kidney disease (ADPKD) is often delayed until adulthood. Indeed, the majority of ADPKD patients with aneurysms experience rupture after the second decade of life. The inconsistent use of angiography, fine-cut computed tomography (CT), and MRI/MRA has made calculating the incidence of aneurysms an even more difficult task. Isolated case reports have confirmed the presence of cerebral aneurysms in infants with this form of the disease. Through linkage studies, adult polycystic kidney disease has been mapped to a 100kb locus on chromosome 16 entitled PKD1. Genetic studies have shown up to an 18% incidence of aneurysms in patients of PKD1 families with at least one affected family member. The PKD1 gene also correlates in other conditions associated with renal cysts such as tuberous sclerosis (TS). Several authors have reported similar arteriopathic findings in ADPKD, fibromuscular dysplasia, and TS patients and thus postulate a common role for PKD1 in vessel wall development.
The second type, infantile polycystic kidney disease is inherited in an autosomal recessive fashion and typically presents early in life as a flank mass or enlarged abdomen. The life expectancy of these patients is typically quite short, and as such, aneurysms in this population are extremely rare (44). In a series of 14 patients with the infantile form of the disease, 12 expired before adolescence with no evidence of intracranial aneurysm. Only one long-term survivor has been reported to develop a basilar aneurysm at age 18.
Chapman in his study of 92 patients with ADPKD quoted a 4 percent frequency of unruptured aneurysms in affected patients as opposed to the 1 percent frequency typically seen in the general population (P<0.04, 95 percent confidence interval). Of note is that fine cut CT with multi-planar reconstruction was their primary screening modality with angiography used to confirm only suspicious findings. Although cerebral angiography is the most sensitive and specific test for intracranial aneurysms, most clinicians do not routinely obtain angiograms in all patients with polycystic kidney disease due to cost, risk, and outcome issues. Indeed, several authors have reported an increased risk of complications when performing angiography in polycystic kidney patients, and have recommended its cautious use in such cases. From the few series of angiographic studies available, a higher frequency of unruptured aneurysms reports exists as compared to when CT is the screening modality: 41% of 17 subjects without neurologic symptoms and 37% of 19 patients with symptoms.
Based on these observations, one cannot make a definitive statement as to the exact risk of ADPKD patients for harboring an intracranial aneurysm. Cerebral angiography, on the other hand, does carry a clear amount of risk in ADPKD patients. As such, our current recommendations are that either a fine-cut CT or MRI / MRA is necessary for patients diagnosed with ADPKD. As in patients with aortic coarctation, those presenting with neurological symptoms or with abnormal noninvasive imaging should undergo four-vessel cerebral angiography. As other authors have recommended, we do not routinely screen for aneurysms in patients with the infantile form of the disease as their risk is low and their life-expectancy short.
Tuberous Sclerosis (TS)
Bourneville’s disease (TS) also correlates with multiple intracranial aneurysms and renal cysts. Tuberous sclerosis is a disorder with protean manifestations involving multiple organ systems, and the best explanation is that it is a developmental tissue dysplasia involving both ectodermal and mesodermal elements. Its characteristic clinical presentation is by a classic triad of mental retardation, epilepsy, and adenoma sebaceum. The putative gene for TS also shares a common location with the polycystic kidney disease gene PKD1 (100kb) on chromosome 16. Blumenkopf in his review of this subject discovered only five cases of TS with associated intracranial aneurysms. The tissue dysplasia of TS is felt to be partly responsible for a congenital defect of the arterial wall thus explaining the clinical observation of an increased number of multiple and fusiform aneurysms.
Interestingly, the histological findings in renal cysts of TS are identical to those found in the cysts of ADPKD patients. Both diseases have been known to co-exist in the same patient and can often be difficult to differentiate in cases exhibiting non-classic phenotypes. A central role for the PKD1 gene pertaining to these histological changes has been postulated but remains to be proven. Other congenital syndromes associated with renal cysts such as Zellweger disease or Meckel-Gruber syndrome do not demonstrate an increased risk for cerebral aneurysms. Screening angiograms in patients with tuberous sclerosis (TS) have been ineffective as children typically go on to bleed from lesions not initially documented on screening angiography.
Fibromuscular Dysplasia
Described primarily as an angiopathy often associated with hypertension, fibromuscular dysplasia (FMD) is characterized by dysplastic vessel wall changes that spread multifocally within branches of the aorta. Found primarily in hypertensive middle-aged women, FMD has been associated with an increased number of intracranial aneurysms as well as with multiple other congenital anomalies. Although the establishment of this linkage between FMD and cerebral aneurysms is clear in adult patients, cases of subarachnoid hemorrhage and aneurysms in children with FMD are exceedingly rare.
Ehlers-Danlos and Marfan syndrome
Intracranial aneurysms are most frequently associated with patients having type IV Ehlers-Danlos syndrome. As an autosomal dominant defect in collagen synthesis, Ehlers-Danlos patients classically have hyperplastic skin and hyperextensible joints. As a clinical entity, it further separates into several clinical types (I-IV) with differences in the specific collagen synthesis defect, clinical features, and inheritance pattern evident. The classic phenotypical features are not typically seen in patients with type IV disease and thus can cause delayed diagnosis until rupture of the cerebral aneurysm occurs, usually in adulthood. Obtaining an MRI / MRA or CT of a child with known type IV disease would not be unwarranted. Intracranial aneurysms have also been occasionally reported in patients with Ehlers-Danlos type I and type VI as well.
Whereas the existence of large dissecting aneurysms of the aorta is a well-recognized feature of Marfan Syndrome, less clear is the association between Marfans and intracerebral aneurysms. A review of the literature reveals that no definitive conclusion has been reached concerning the relationship of berry aneurysms to Marfan’s syndrome.
Familial aneurysm occurrence
Given the rare incidence of intracranial aneurysms in children as a whole, it is not surprising to discover that familial cases are even more uncommon. Although most of these patients remain asymptomatic until adulthood, familial aneurysms do tend to rupture at an earlier age than would be expected. Additionally, familial lesions show a propensity for the peripheral vasculature especially that of the middle cerebral artery territory. Reports of an increased incidence of multiple aneurysms also exist with familial cases, especially in cases associated with Marfan syndrome, Ehlers-Danlos syndrome, polycystic kidney disease, hereditary hemorrhagic telangiectasia, cavernous angiomas, Von-Hippel-Lindau disease, arteriovenous malformations, and Sturge-Weber syndrome.
It has been challenging to develop a rational, safe, and cost-effective method of screening children with a familial history. Angiography in children carries with it a quantifiable amount of risk. Given recent advances in imaging, we recommend screening MRI/MRA or CT for such patients. We do not routinely screen asymptomatic children with a single parent who has an aneurysm as the risk is relatively low. In cases where a large number of younger family members are affected, a more aggressive evaluation may be necessary. Standard angiography should be obtained in all clinically or radiographically suspicious cases.
History and Physical
Numerous clinical conditions associated with cerebral aneurysms have been discussed earlier. When encountering these conditions, an increased index of clinical suspicion and heightened surveillance is necessary. However, as a group, the presentation of intracranial aneurysms in children is quite similar to that of adults, though there are clinical features that are unique to pediatric aneurysms. Subarachnoid Hemorrhage (SAH) remains the most common initial manifestation of cerebral aneurysms in both adults and children.[29][30][31][18]
Subarachnoid Hemorrhage
SAH remains the most common initial manifestation of cerebral aneurysms in both adults and children. Rates of SAH in children can range from 1.9% to 4.6%. Increased recognition of SAH in children is likely the result of improved diagnostic capabilities and increased clinical awareness.
For children with aneurysms, the incidence of rupture is surprisingly high. Amacher and Drake reported that 65% of their 52 childhood cases had already ruptured at diagnosis. Our own experience mirrors Drake’s experience, with a 74% incidence of SAH. When compared to the adult population, a greater proportion of childhood aneurysms are symptomatic at the time of diagnosis.
The onset of SAH is typically sudden, with headache, vomiting, deterioration in consciousness, seizures, coma, paresis, cranial nerve palsies, fever, and meningismus commonly reported. Warning signs heralding a major bleed are sometimes seen in the form of a "sentinel bleed" with an associated headache in 10 to 15% of patients. Patients can also present with focal neurological deficits correlating with the expansion of the aneurysm or, symptoms of mass effect and cerebrospinal fluid (CSF) obstruction in the scenario of giant aneurysms. Unlike SAH, these warning signs are often transient and can be challenging to find in children. Aneurysms remain the leading cause of atraumatic SAH in patients under the age of 20.
Of children with aneurysms that rupture, the estimated mortality rate after the first bleed is 11 to 20%, as compared to 20 to 30% in adults. Of the ruptured aneurysms in our own experience, 7% rebled, with a subsequent 25% mortality rate. Proust reported a 52.4%incidence of rebleed in 22 pediatric aneurysm patients. The overall mortality rate of children with ruptured aneurysms ranges from 13 to 34% in the larger series.[29][30][31][32]
Mass Symptoms
A large proportion of children present with signs and symptoms of a mass lesion. In our experience of 52 children with aneurysms, one third presented with signs of increased intracranial pressure. This finding shows special emphasis in certain clinical subgroups including children under the age of 5 years (56%, mass symptoms) and cases of giant aneurysms (90%). Concurrently, hydrocephalus was observed in 30% of all cases. Giant aneurysms (56%), posterior circulation lesions (60%), and early childhood cases (younger than five years; [36%]) were particularly prone to the development of obstructed CSF flow.
Findings such as lethargy; nausea; vomiting; a tense, anterior fontanelle; splayed sutures; papilledema; opisthotonic posture; and paresis or plegia can be present in children with mass effect related to an aneurysm.
Mass symptoms often correlate with ICH in children with ruptured aneurysms. Intraparenchymal and interventricular hemorrhage can often occur in conjunction with the initial SAH with an incidence of 30 to 40%.
The predilection for childhood aneurysms at the internal carotid bifurcation and distal middle cerebral vessels is thought to be partially responsible for ICH at presentation. The presence of ICH is associated with a higher risk of mortality and overall poorer prognosis. In one study of 43 children, the overall mortality of cases with an ICH (50%) was twice that of children without ICH (24%).
Enlargement of an aneurysm, hematoma formation, or hydrocephalus can often lead to focal neurological findings from cranial nerve parenchymal compression, which can include sixth-nerve palsies, focal third-nerve palsies, ophthalmoplegia, dysphasia, and other lower cranial palsies. Hemiparesis, hemiplegia, and quadriparesis secondary to pyramidal compression may also be present. Amacher and Drake reported a 34.4% incidence of neural compression in 32 children.[27][33][34]
Seizures
Although seizures and epilepsy are often common for children with AVMs, they are comparatively uncommon with an unruptured aneurysm. Focal or generalized seizures can occur in 15 to 25% of children with SAH and may be more common in infants. In our experience, we found a 7% seizure rate at the time of presentation. Seizures occurred most frequently in children with giant (25%) and posterior (27%) circulation aneurysms. Other series have reported an incidence as high as 25% of seizures arising from possible microemboli from the aneurysmal sac itself. The majority of seizures occur within 24 hours of the initial hemorrhage but sometimes progress to epilepsy or status epilepticus. Elevations of arterial blood pressure and the concurrent risk of re-bleed should be avoided by effective management with anticonvulsant medications. From our experience, however, long-term seizure prophylaxis has generally not been necessary.[35][36]
Evaluation
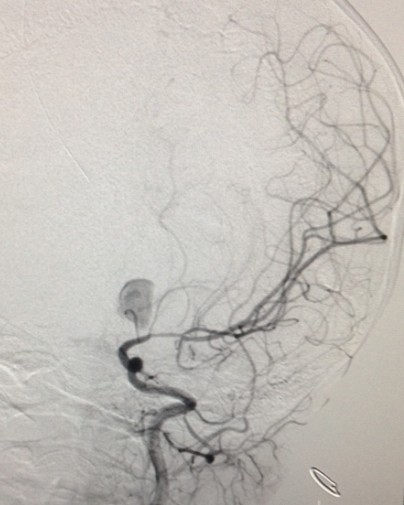
For asymptomatic children, who are at increased risk for an intracranial aneurysm, a CT-angiogram or an MR-angiogram is a useful and reasonable first screening tool. As discussed earlier, children with a strong family history of aneurysms, Ehlers-Danlos syndrome, FMD, polycystic kidney disease, and other high-risk conditions are good candidates for MR angiography. MR imaging and angiography can visualize larger lesions of the circle of Willis, but cerebral angiography remains the gold standard for localization of cerebral aneurysms.[37][13][38]
For asymptomatic children, who are at increased risk for an intracranial aneurysm, a CT-angiogram or an MR-angiogram is a useful and reasonable first screening tool. As discussed earlier (see Associated Conditions), children with a strong family history of aneurysms, Ehlers-Danlos syndrome, FMD, polycystic kidney disease, and other high-risk conditions are good candidates for MR angiography. MR imaging and angiography are able to visualize larger lesions of the circle of Willis, but cerebral angiography remains the gold standard for localization of cerebral aneurysms and should be utilized in any case where MRI and/or CT is indeterminate in the potential clinical setting of an aneurysm.
For ruptured aneurysms, lumbar puncture (LP) and CT/MR imaging remain the mainstay in the initial evaluation of SAH. Eighty-five percent of children with subarachnoid blood can be accurately identified with a non-contrast CT scan when performed within the first 2 days after hemorrhage becoming less reliable 5 days.
CT or MR angiography is extremely useful in localizing larger cerebral aneurysms. For such cases, detection of the mass lesion may be quite easy, even on non-contrast screening studies. Contrast studies can provide additional clues to the rupture site. In very young infants, intracranial ultrasound has also been successfully used to diagnose large cerebral aneurysms. Ultrasonography can also show hydrocephalus, hematoma, shift, and cerebral edema.
Due to the high incidence of increased intracranial pressures in these children, we recommend deferring LP until the exclusion of the presence of a mass lesion (e.g., the aneurysm), hematoma, cerebral edema, and hydrocephalus from imaging studies. An apparent SAH on a head CT scan obviates the need for LP. If the imaging study results are negative for SAH and there is no radiographic or clinical evidence of increased intracranial pressure, CSF can be safely obtained to confirm suspicious cases.
Once the presence of SAH has been established in a child, a standard, four-vessel cerebral angiogram should be obtained. For children under the 6 years of age, general anesthesia is often required, with careful attention paid to fluid balance, contrast load, blood pressure control, and neural protection. Although controversy exists about the best timing of the angiogram, studies have demonstrated no increased morbidity of early studies (performed within a few hours after SAH) over delayed studies. Indeed, some large series have actually shown lower morbidity with early studies.
Overall, cerebral angiography can demonstrate a structural cause for SAH in 50 to 70% of cases. Of the negative studies, another 10 to 20% will subsequently be positive for an aneurysm on a repeat study at 2weeks’ time. Focal arterial spasm and thrombosis within the aneurysmal neck and some may prevent filling of the lesion during angiography resulting in negative studies.
When no aneurysm can is identifiable in a confirmed case of a traumatic SAH, the rest of the differential diagnosis should then be considered. Systemic childhood causes of SAH include leukemia, idiopathic thrombocytopenic purpura, hemorrhagic disease of the newborn, various hemophilia disorders, and other hemorrhagic diatheses.[39][17][40]
Treatment / Management
Medical Management
In patients with SAH and suspected aneurysm, appropriate medical management should be initiated immediately to stabilize the child and to prevent potential secondary complications of hemorrhage, as rebleeding represents the most serious complication within the first 48 hours after rupture.[41][42][43] Definitive diversion of the CSF is often indicated if hydrocephalus is present. The decision to place a catheter before definitive management of the aneurysm commences requires caution; placement of a ventricular catheter has correlations with rupture thought to be due to the associated change in transluminal pressure from the altered spinal fluid dynamics.[44][45][17]
Paying strict attention to the patient’s volume status usually requires placement of an indwelling urinary catheter and arterial line. Adequate analgesia and sedation to treat the associated anxiety and headache of SAH must be weighed against the need for frequent evaluation of the neurological examination. Allowing the parents to remain with young patients often helps alleviate the child’s fear. Antihypertensive medications, including calcium channel blockers, beta-blockers, and vasodilators may be a requirement for pressure control.
We routinely initially administer antiepileptic agents to prevent post-SAH seizures and the undesirable effects of secondary hypertension and hypoxia. Stool softeners, breathing treatments, and antiemetic medication may also be helpful in reducing fluctuations of intracranial pressure caused by straining, coughing, and vomiting.
Calcium channel blockers, such as nimodipine, may reduce the incidence of severe neurological deficits associated with vasospasm. Angiographic vasospasm is less common in children, with rare clinical progression and experience with nimodipine in children with SAH is limited. In cases of clinical vasospasm, the use of volume expansion, hyperdynamic therapy with pressors and inotropes, and hemodilution should be instituted.
Prevention and recognition of delayed medical issues such as gastrointestinal hemorrhage (complicates approximately 4% of patients), pulmonary edema, and hyperglycemia are important. Aggressive pulmonary toilet should be implemented with appropriate use of antibiotic agents in cases of suspected infection. Unrecognized hyponatremia (cerebral salt wasting) may exacerbate alterations in the level of consciousness, increase seizure activity, and worse cerebral edema. Decreased plasma sodium may be present in up to 35% of SAH patients, with a peak incidence between the second and tenth days after rupture. Extracellular volume assessment in these patients typically shows a hypovolemic state. In most cases of hyponatremia, sodium replacement (oral sodium supplements and hypertonic intravenous saline), combined with volume expansion (normal saline, colloids, and red cells), is often adequate and appropriate therapy.
Surgical Management
Surgical and endovascular intervention remains the definitive treatment of cerebral aneurysms in children. Several clinical series have correlated nonsurgical management with an overall poorer outcome. This improved outcome from surgery must be weighed against the increased rate of spontaneous aneurysm thrombosis that experience shows may occur in childhood lesions. In cases which partial thrombosis of the lesion has already occurred, a conservative course of observation and supportive care may be necessary.
In cases of mycotic aneurysms, conservative treatment with antibiotic agents is a reasonable, although controversial, an alternative to direct surgical repair in some cases. For children with multiple aneurysms and those with preexisting cardiac disease and endocarditis, the risk of surgery may be unexceptionally high, even when weighed against the ongoing risk of a potentially devastating hemorrhage. After institution of antimicrobial therapy, lesions should be followed closely with serial angiography every 2 to 3 weeks. Failure to involute or progressive enlargement of lesions despite antibiotics is a strong indication for surgical intervention. CT-angiogram and MR-angiogram can be utilized to assess the response of mycotic aneurysms to treatment.
Postponement of intervention for 7 to 14 days after the SAH may allow for stabilization of high-grade, critically ill children. Resolution of the cerebral edema and vasospasm also helps reduce surgical morbidity and mortality. These advantages balance against the risk of rebleed. We think that obliteration of the aneurysm should be performed for low-grade patients at the earliest possible date. The indications and timing for surgery in high-grade cases varies for each child.
From a technical perspective, surgical management of aneurysms in children is quite similar to that of adults. Special attention must be paid to the smaller size of anatomic structures and the decreased tolerance for temperature changes, blood loss, and large fluid shifts due to smaller physiologic reserve in children.
Due to the often giant size and unusual locations of pediatric aneurysms, surgical obliterations frequently not straightforward. A review of the literature shows that direct clip ligation of the aneurysmal neck is possible in only 29.5% of cases. A high percentage of pediatric patients require specialized and often innovative procedures to obliterate their lesions.[46][47][16][18][48]
Differential Diagnosis
- Acute management of stroke
- Anterior circulation stroke
- Apraxia and related syndromes
- Basilar artery thrombosis
Enhancing Healthcare Team Outcomes
In the modern era, endovascular management of pediatric cerebral aneurysms represents a viable alternative to open surgical obliteration.[37][49][50][13] The minimally invasive nature of the procedure, smaller associated fluid shifts, and blood losses, and decreased surgical manipulation of brain tissue are all attractive features of interventional therapy. Like open surgery, endovascular management carries with it several serious risks and potential complications that must be recognized. Vessel perforation, stroke, hemorrhage, and incomplete obliteration may all result after an intervention.[51][38][52][53]