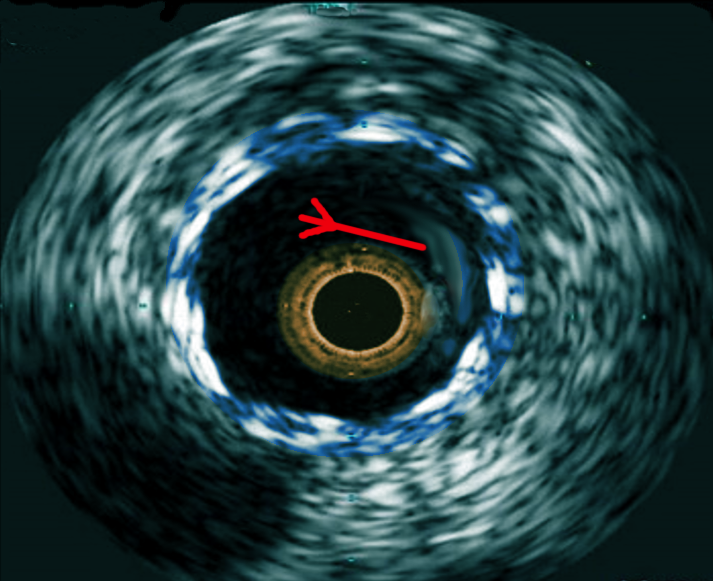
[1]
Longobardo L, Mattesini A, Valente S, Di Mario C. OCT-guided Percutaneous Coronary Intervention in Bifurcation Lesions. Interventional cardiology (London, England). 2019 Feb:14(1):5-9. doi: 10.15420/icr.2018.17.2. Epub
[PubMed PMID: 30858885]
[2]
Piranavan P, Kaur NJ, Marmoush F, Burton A, Hannan J. Ticagrelor-induced Angioedema After Percutaneous Coronary Intervention in a Patient with a History of Ischemic Stroke and Low Response to Clopidogrel: A Rare Dilemma. Cureus. 2018 Dec 11:10(12):e3720. doi: 10.7759/cureus.3720. Epub 2018 Dec 11
[PubMed PMID: 30891370]
[3]
Lee SN, Moon D, Moon KW, Yoo KD. The Glasgow prognostic score as a significant predictor of clinical outcomes in patients with acute coronary syndrome. Journal of cardiology. 2019 Aug:74(2):130-135. doi: 10.1016/j.jjcc.2019.02.007. Epub 2019 Mar 13
[PubMed PMID: 30878352]
Level 2 (mid-level) evidence
[4]
Cutlip DE, Nakazawa G, Krucoff MW, Vorpahl M, Mehran R, Finn AV, Vranckx P, Kimmelstiel C, Berger C, Petersen JL, Palabrica T, Virmani R. Autopsy validation study of the academic research consortium stent thrombosis definition. JACC. Cardiovascular interventions. 2011 May:4(5):554-9. doi: 10.1016/j.jcin.2011.01.011. Epub
[PubMed PMID: 21596329]
Level 1 (high-level) evidence
[5]
Che QQ, Wu Q, Liang YB, Sun RM, Lyu QW, Ma JL, Hu H, Lin X, Xu GL, Sun SG, Zhang C, Wang QY, Yu J, Bai F. [Meta-analysis on safety and efficacy of dual antiplatelet therapy combining with proton pump inhibitors for patients after percutaneous coronary intervention]. Zhonghua xin xue guan bing za zhi. 2019 Feb 24:47(2):129-140. doi: 10.3760/cma.j.issn.0253-3758.2019.02.010. Epub
[PubMed PMID: 30818941]
Level 1 (high-level) evidence
[6]
Guo C, Li M, Lv YH, Zhang MB, Wang ZL. De-escalation versus standard dual antiplatelet therapy in patients undergoing percutaneous coronary intervention: a systematic review and meta-analysis. Platelets. 2020:31(1):15-25. doi: 10.1080/09537104.2019.1574969. Epub 2019 Feb 13
[PubMed PMID: 30759033]
Level 1 (high-level) evidence
[7]
Wang Z, Xie Q, Xiang Q, Gong Y, Jiang J, Cui Y. Predictive Value of Methods Measuring Platelet Activation for Ischemic Events in Patients Receiving Clopidogrel: A Systematic Review and Meta-analysis. Current pharmaceutical design. 2018:24(44):5313-5333. doi: 10.2174/1381612825666190206114724. Epub
[PubMed PMID: 30727868]
Level 1 (high-level) evidence
[8]
Généreux P, Stone GW, Harrington RA, Gibson CM, Steg PG, Brener SJ, Angiolillo DJ, Price MJ, Prats J, LaSalle L, Liu T, Todd M, Skerjanec S, Hamm CW, Mahaffey KW, White HD, Bhatt DL, CHAMPION PHOENIX Investigators. Impact of intraprocedural stent thrombosis during percutaneous coronary intervention: insights from the CHAMPION PHOENIX Trial (Clinical Trial Comparing Cangrelor to Clopidogrel Standard of Care Therapy in Subjects Who Require Percutaneous Coronary Intervention). Journal of the American College of Cardiology. 2014 Feb 25:63(7):619-629. doi: 10.1016/j.jacc.2013.10.022. Epub 2013 Oct 30
[PubMed PMID: 24184169]
[9]
Dangas GD, Claessen BE, Mehran R, Xu K, Stone GW. Stent thrombosis after primary angioplasty for STEMI in relation to non-adherence to dual antiplatelet therapy over time: results of the HORIZONS-AMI trial. EuroIntervention : journal of EuroPCR in collaboration with the Working Group on Interventional Cardiology of the European Society of Cardiology. 2013 Jan 22:8(9):1033-9. doi: 10.4244/EIJV8I9A159. Epub
[PubMed PMID: 23339809]
[10]
Husted S, Boersma E. Case Study: Ticagrelor in PLATO and Prasugrel in TRITON-TIMI 38 and TRILOGY-ACS Trials in Patients With Acute Coronary Syndromes. American journal of therapeutics. 2016 Nov/Dec:23(6):e1876-e1889
[PubMed PMID: 25830867]
Level 3 (low-level) evidence
[11]
Noaman S, O'Brien J, Andrianopoulos N, Brennan AL, Dinh D, Reid C, Sharma A, Chan W, Clark D, Stub D, Biswas S, Freeman M, Ajani A, Yip T, Duffy SJ, Oqueli E, Melbourne Interventional Group Investigators. Clinical outcomes following ST-elevation myocardial infarction secondary to stent thrombosis treated by percutaneous coronary intervention. Catheterization and cardiovascular interventions : official journal of the Society for Cardiac Angiography & Interventions. 2020 Oct 1:96(4):E406-E415. doi: 10.1002/ccd.28802. Epub 2020 Feb 22
[PubMed PMID: 32087042]
Level 2 (mid-level) evidence
[12]
Kim YH, Her AY, Jeong MH, Kim BK, Hong SJ, Ahn CM, Kim JS, Ko YG, Choi D, Hong MK, Jang Y. A comparison of the impact of current smoking on 2-year major clinical outcomes of first- and second-generation drug-eluting stents in acute myocardial infarction: Data from the Korea Acute Myocardial Infarction Registry. Medicine. 2019 Mar:98(10):e14797. doi: 10.1097/MD.0000000000014797. Epub
[PubMed PMID: 30855497]
Level 2 (mid-level) evidence
[13]
Zhao XY, Li JX, Tang XF, Xu JJ, Song Y, Jiang L, Chen J, Song L, Gao LJ, Gao Z, Qiao SB, Yang YJ, Gao RL, Xu B, Yuan JQ. Validation of Predictive Value of Patterns of Nonadherence to Antiplatelet Regimen in Stented Patients Thrombotic Risk Score in Chinese Population Undergoing Percutaneous Coronary Intervention: A Prospective Observational Study. Chinese medical journal. 2018 Nov 20:131(22):2699-2704. doi: 10.4103/0366-6999.245263. Epub
[PubMed PMID: 30425196]
Level 2 (mid-level) evidence
[14]
Yahagi K, Kolodgie FD, Otsuka F, Finn AV, Davis HR, Joner M, Virmani R. Pathophysiology of native coronary, vein graft, and in-stent atherosclerosis. Nature reviews. Cardiology. 2016 Feb:13(2):79-98. doi: 10.1038/nrcardio.2015.164. Epub 2015 Oct 27
[PubMed PMID: 26503410]
[15]
Ge J, Yu H, Li J. Acute Coronary Stent Thrombosis in Modern Era: Etiology, Treatment, and Prognosis. Cardiology. 2017:137(4):246-255. doi: 10.1159/000464404. Epub 2017 May 5
[PubMed PMID: 28472800]
[16]
Godschalk TC, Willemsen LM, Zwart B, Bergmeijer TO, Janssen PWA, Kelder JC, Hackeng CM, Ten Berg JM. Effect of Tailored Antiplatelet Therapy to Reduce Recurrent Stent Thrombosis and Cardiac Death After a First Episode of Stent Thrombosis. The American journal of cardiology. 2017 May 15:119(10):1500-1506. doi: 10.1016/j.amjcard.2017.02.033. Epub 2017 Feb 27
[PubMed PMID: 28318511]
[17]
Redfors B, Dworeck C, Haraldsson I, Angerås O, Odenstedt J, Ioanes D, Petursson P, Völz S, Albertsson P, Råmunddal T, Persson J, Koul S, Erlinge D, Omerovic E. Pretreatment with P2Y12 receptor antagonists in ST-elevation myocardial infarction: a report from the Swedish Coronary Angiography and Angioplasty Registry. European heart journal. 2019 Apr 14:40(15):1202-1210. doi: 10.1093/eurheartj/ehz069. Epub
[PubMed PMID: 30851037]
[18]
Guy JM, Wilson M, Schnell F, Chevalier L, Verdier JC, Corone S, Doutreleau S, Kervio G, Carré F. Incidence of major adverse cardiac events in men wishing to continue competitive sport following percutaneous coronary intervention. Archives of cardiovascular diseases. 2019 Apr:112(4):226-233. doi: 10.1016/j.acvd.2018.11.008. Epub 2019 Jan 3
[PubMed PMID: 30612894]
[19]
Capodanno D, Alfonso F, Levine GN, Valgimigli M, Angiolillo DJ. ACC/AHA Versus ESC Guidelines on Dual Antiplatelet Therapy: JACC Guideline Comparison. Journal of the American College of Cardiology. 2018 Dec 11:72(23 Pt A):2915-2931. doi: 10.1016/j.jacc.2018.09.057. Epub
[PubMed PMID: 30522654]
[20]
Torrado J, Buckley L, Durán A, Trujillo P, Toldo S, Valle Raleigh J, Abbate A, Biondi-Zoccai G, Guzmán LA. Restenosis, Stent Thrombosis, and Bleeding Complications: Navigating Between Scylla and Charybdis. Journal of the American College of Cardiology. 2018 Apr 17:71(15):1676-1695. doi: 10.1016/j.jacc.2018.02.023. Epub
[PubMed PMID: 29650125]
[21]
Rokoszak V, Syed MH, Salata K, Greco E, de Mestral C, Hussain MA, Aljabri B, Verma S, Al-Omran M. A systematic review and meta-analysis of plain versus drug-eluting balloon angioplasty in the treatment of juxta-anastomotic hemodialysis arteriovenous fistula stenosis. Journal of vascular surgery. 2020 Mar:71(3):1046-1054.e1. doi: 10.1016/j.jvs.2019.07.075. Epub
[PubMed PMID: 32089200]
Level 1 (high-level) evidence
[22]
Abdelaziz HK, Abuomara HZ, Ali MH, Eichhofer J, Patel B, Saad M. Routine use of optical coherence tomography in bioresorbable vascular scaffold implantation: insights on technique optimization and long-term outcomes. Coronary artery disease. 2019 Jun:30(4):263-269. doi: 10.1097/MCA.0000000000000736. Epub
[PubMed PMID: 30883433]
[23]
Agarwal N, Mahmoud AN, Mojadidi MK, Golwala H, Elgendy IY. Dual versus triple antithrombotic therapy in patients undergoing percutaneous coronary intervention-meta-analysis and meta-regression. Cardiovascular revascularization medicine : including molecular interventions. 2019 Dec:20(12):1134-1139. doi: 10.1016/j.carrev.2019.02.022. Epub 2019 Mar 5
[PubMed PMID: 30862490]
Level 1 (high-level) evidence
[24]
Hommels TM, Hermanides RS, Rasoul S, Berta B, IJsselmuiden AJJ, Jessurun GAJ, Benit E, Pereira B, De Luca G, Kedhi E. Everolimus-eluting bioresorbable scaffolds for treatment of coronary artery disease in patients with diabetes mellitus: the midterm follow-up of the prospective ABSORB DM Benelux study. Cardiovascular diabetology. 2019 Mar 9:18(1):25. doi: 10.1186/s12933-019-0827-z. Epub 2019 Mar 9
[PubMed PMID: 30851731]