[1]
Rawla P. Epidemiology of Prostate Cancer. World journal of oncology. 2019 Apr:10(2):63-89. doi: 10.14740/wjon1191. Epub 2019 Apr 20
[PubMed PMID: 31068988]
[2]
Selman SH. The McNeal prostate: a review. Urology. 2011 Dec:78(6):1224-8. doi: 10.1016/j.urology.2011.07.1395. Epub 2011 Sep 9
[PubMed PMID: 21908026]
[3]
Packer JR, Maitland NJ. The molecular and cellular origin of human prostate cancer. Biochimica et biophysica acta. 2016 Jun:1863(6 Pt A):1238-60. doi: 10.1016/j.bbamcr.2016.02.016. Epub 2016 Feb 26
[PubMed PMID: 26921821]
[5]
Humphrey PA. Histopathology of Prostate Cancer. Cold Spring Harbor perspectives in medicine. 2017 Oct 3:7(10):. doi: 10.1101/cshperspect.a030411. Epub 2017 Oct 3
[PubMed PMID: 28389514]
Level 3 (low-level) evidence
[6]
Mottet N, Bellmunt J, Bolla M, Briers E, Cumberbatch MG, De Santis M, Fossati N, Gross T, Henry AM, Joniau S, Lam TB, Mason MD, Matveev VB, Moldovan PC, van den Bergh RCN, Van den Broeck T, van der Poel HG, van der Kwast TH, Rouvière O, Schoots IG, Wiegel T, Cornford P. EAU-ESTRO-SIOG Guidelines on Prostate Cancer. Part 1: Screening, Diagnosis, and Local Treatment with Curative Intent. European urology. 2017 Apr:71(4):618-629. doi: 10.1016/j.eururo.2016.08.003. Epub 2016 Aug 25
[PubMed PMID: 27568654]
[7]
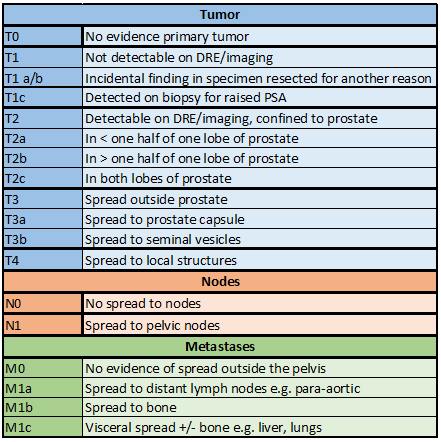
Fine SW. Evolution in Prostate Cancer Staging: Pathology Updates From AJCC 8th Edition and Opportunities That Remain. Advances in anatomic pathology. 2018 Sep:25(5):327-332. doi: 10.1097/PAP.0000000000000200. Epub
[PubMed PMID: 29870405]
Level 3 (low-level) evidence
[8]
Albertsen PC, Hanley JA, Fine J. 20-year outcomes following conservative management of clinically localized prostate cancer. JAMA. 2005 May 4:293(17):2095-101
[PubMed PMID: 15870412]
[9]
Bernstein AN, Shoag JE, Golan R, Halpern JA, Schaeffer EM, Hsu WC, Nguyen PL, Sedrakyan A, Chen RC, Eggener SE, Hu JC. Contemporary Incidence and Outcomes of Prostate Cancer Lymph Node Metastases. The Journal of urology. 2018 Jun:199(6):1510-1517. doi: 10.1016/j.juro.2017.12.048. Epub 2017 Dec 26
[PubMed PMID: 29288121]
[10]
Barry MJ, Simmons LH. Prevention of Prostate Cancer Morbidity and Mortality: Primary Prevention and Early Detection. The Medical clinics of North America. 2017 Jul:101(4):787-806. doi: 10.1016/j.mcna.2017.03.009. Epub
[PubMed PMID: 28577627]
[11]
Vinceti M, Filippini T, Del Giovane C, Dennert G, Zwahlen M, Brinkman M, Zeegers MP, Horneber M, D'Amico R, Crespi CM. Selenium for preventing cancer. The Cochrane database of systematic reviews. 2018 Jan 29:1(1):CD005195. doi: 10.1002/14651858.CD005195.pub4. Epub 2018 Jan 29
[PubMed PMID: 29376219]
Level 1 (high-level) evidence
[12]
Markozannes G, Tzoulaki I, Karli D, Evangelou E, Ntzani E, Gunter MJ, Norat T, Ioannidis JP, Tsilidis KK. Diet, body size, physical activity and risk of prostate cancer: An umbrella review of the evidence. European journal of cancer (Oxford, England : 1990). 2016 Dec:69():61-69. doi: 10.1016/j.ejca.2016.09.026. Epub 2016 Nov 4
[PubMed PMID: 27816833]
Level 1 (high-level) evidence
[13]
Pernar CH, Ebot EM, Wilson KM, Mucci LA. The Epidemiology of Prostate Cancer. Cold Spring Harbor perspectives in medicine. 2018 Dec 3:8(12):. doi: 10.1101/cshperspect.a030361. Epub 2018 Dec 3
[PubMed PMID: 29311132]
Level 3 (low-level) evidence
[14]
Welch HG, Albertsen PC. Reconsidering Prostate Cancer Mortality - The Future of PSA Screening. The New England journal of medicine. 2020 Apr 16:382(16):1557-1563. doi: 10.1056/NEJMms1914228. Epub
[PubMed PMID: 32294352]
[15]
US Preventive Services Task Force, Grossman DC, Curry SJ, Owens DK, Bibbins-Domingo K, Caughey AB, Davidson KW, Doubeni CA, Ebell M, Epling JW Jr, Kemper AR, Krist AH, Kubik M, Landefeld CS, Mangione CM, Silverstein M, Simon MA, Siu AL, Tseng CW. Screening for Prostate Cancer: US Preventive Services Task Force Recommendation Statement. JAMA. 2018 May 8:319(18):1901-1913. doi: 10.1001/jama.2018.3710. Epub
[PubMed PMID: 29801017]
[16]
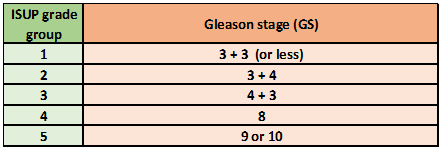
Epstein JI. An update of the Gleason grading system. The Journal of urology. 2010 Feb:183(2):433-40. doi: 10.1016/j.juro.2009.10.046. Epub 2009 Dec 14
[PubMed PMID: 20006878]
[17]
Egevad L, Delahunt B, Srigley JR, Samaratunga H. International Society of Urological Pathology (ISUP) grading of prostate cancer - An ISUP consensus on contemporary grading. APMIS : acta pathologica, microbiologica, et immunologica Scandinavica. 2016 Jun:124(6):433-5. doi: 10.1111/apm.12533. Epub
[PubMed PMID: 27150257]
Level 3 (low-level) evidence
[18]
Merriel SWD, Funston G, Hamilton W. Prostate Cancer in Primary Care. Advances in therapy. 2018 Sep:35(9):1285-1294. doi: 10.1007/s12325-018-0766-1. Epub 2018 Aug 10
[PubMed PMID: 30097885]
Level 3 (low-level) evidence
[19]
Ilic D, Djulbegovic M, Jung JH, Hwang EC, Zhou Q, Cleves A, Agoritsas T, Dahm P. Prostate cancer screening with prostate-specific antigen (PSA) test: a systematic review and meta-analysis. BMJ (Clinical research ed.). 2018 Sep 5:362():k3519. doi: 10.1136/bmj.k3519. Epub 2018 Sep 5
[PubMed PMID: 30185521]
Level 1 (high-level) evidence
[20]
Carroll PH,Mohler JL, NCCN Guidelines Updates: Prostate Cancer and Prostate Cancer Early Detection. Journal of the National Comprehensive Cancer Network : JNCCN. 2018 May;
[PubMed PMID: 29784740]
[21]
Schröder FH, Hugosson J, Roobol MJ, Tammela TL, Zappa M, Nelen V, Kwiatkowski M, Lujan M, Määttänen L, Lilja H, Denis LJ, Recker F, Paez A, Bangma CH, Carlsson S, Puliti D, Villers A, Rebillard X, Hakama M, Stenman UH, Kujala P, Taari K, Aus G, Huber A, van der Kwast TH, van Schaik RH, de Koning HJ, Moss SM, Auvinen A, ERSPC Investigators. Screening and prostate cancer mortality: results of the European Randomised Study of Screening for Prostate Cancer (ERSPC) at 13 years of follow-up. Lancet (London, England). 2014 Dec 6:384(9959):2027-35. doi: 10.1016/S0140-6736(14)60525-0. Epub 2014 Aug 6
[PubMed PMID: 25108889]
Level 1 (high-level) evidence
[22]
Hamdy FC, Donovan JL, Lane JA, Mason M, Metcalfe C, Holding P, Wade J, Noble S, Garfield K, Young G, Davis M, Peters TJ, Turner EL, Martin RM, Oxley J, Robinson M, Staffurth J, Walsh E, Blazeby J, Bryant R, Bollina P, Catto J, Doble A, Doherty A, Gillatt D, Gnanapragasam V, Hughes O, Kockelbergh R, Kynaston H, Paul A, Paez E, Powell P, Prescott S, Rosario D, Rowe E, Neal D. Active monitoring, radical prostatectomy and radical radiotherapy in PSA-detected clinically localised prostate cancer: the ProtecT three-arm RCT. Health technology assessment (Winchester, England). 2020 Aug:24(37):1-176. doi: 10.3310/hta24370. Epub
[PubMed PMID: 32773013]
[23]
Loeb S, Vellekoop A, Ahmed HU, Catto J, Emberton M, Nam R, Rosario DJ, Scattoni V, Lotan Y. Systematic review of complications of prostate biopsy. European urology. 2013 Dec:64(6):876-92. doi: 10.1016/j.eururo.2013.05.049. Epub 2013 Jun 4
[PubMed PMID: 23787356]
Level 1 (high-level) evidence
[24]
Smith RA,Andrews KS,Brooks D,Fedewa SA,Manassaram-Baptiste D,Saslow D,Wender RC, Cancer screening in the United States, 2019: A review of current American Cancer Society guidelines and current issues in cancer screening. CA: a cancer journal for clinicians. 2019 May;
[PubMed PMID: 30875085]
[25]
Carroll PR, Parsons JK, Andriole G, Bahnson RR, Castle EP, Catalona WJ, Dahl DM, Davis JW, Epstein JI, Etzioni RB, Farrington T, Hemstreet GP 3rd, Kawachi MH, Kim S, Lange PH, Loughlin KR, Lowrance W, Maroni P, Mohler J, Morgan TM, Moses KA, Nadler RB, Poch M, Scales C, Shaneyfelt TM, Smaldone MC, Sonn G, Sprenkle P, Vickers AJ, Wake R, Shead DA, Freedman-Cass DA. NCCN Guidelines Insights: Prostate Cancer Early Detection, Version 2.2016. Journal of the National Comprehensive Cancer Network : JNCCN. 2016 May:14(5):509-19
[PubMed PMID: 27160230]
[26]
Heijnsdijk EAM, Bangma CH, Borràs JM, de Carvalho TM, Castells X, Eklund M, Espinàs JA, Graefen M, Grönberg H, Lansdorp-Vogelaar I, Leeuwen PJV, Nelen V, Recker F, Roobol MJ, Vandenbulcke P, de Koning HJ. Summary statement on screening for prostate cancer in Europe. International journal of cancer. 2018 Feb 15:142(4):741-746. doi: 10.1002/ijc.31102. Epub 2017 Oct 25
[PubMed PMID: 29023685]
[27]
Puech P, Sufana Iancu A, Renard B, Villers A, Lemaitre L. Detecting prostate cancer with MRI - why and how. Diagnostic and interventional imaging. 2012 Apr:93(4):268-78. doi: 10.1016/j.diii.2012.01.019. Epub 2012 Mar 31
[PubMed PMID: 22465788]
[28]
Khoo CC, Eldred-Evans D, Peters M, Bertoncelli Tanaka M, Noureldin M, Miah S, Shah T, Connor MJ, Reddy D, Clark M, Lakhani A, Rockall A, Hosking-Jervis F, Cullen E, Arya M, Hrouda D, Qazi H, Winkler M, Tam H, Ahmed HU. Likert vs PI-RADS v2: a comparison of two radiological scoring systems for detection of clinically significant prostate cancer. BJU international. 2020 Jan:125(1):49-55. doi: 10.1111/bju.14916. Epub 2019 Nov 1
[PubMed PMID: 31599113]
[29]
Ahmed HU, El-Shater Bosaily A, Brown LC, Gabe R, Kaplan R, Parmar MK, Collaco-Moraes Y, Ward K, Hindley RG, Freeman A, Kirkham AP, Oldroyd R, Parker C, Emberton M, PROMIS study group. Diagnostic accuracy of multi-parametric MRI and TRUS biopsy in prostate cancer (PROMIS): a paired validating confirmatory study. Lancet (London, England). 2017 Feb 25:389(10071):815-822. doi: 10.1016/S0140-6736(16)32401-1. Epub 2017 Jan 20
[PubMed PMID: 28110982]
[30]
Faria R, Soares MO, Spackman E, Ahmed HU, Brown LC, Kaplan R, Emberton M, Sculpher MJ. Optimising the Diagnosis of Prostate Cancer in the Era of Multiparametric Magnetic Resonance Imaging: A Cost-effectiveness Analysis Based on the Prostate MR Imaging Study (PROMIS). European urology. 2018 Jan:73(1):23-30. doi: 10.1016/j.eururo.2017.08.018. Epub 2017 Sep 19
[PubMed PMID: 28935163]
[31]
Rosario DJ, Lane JA, Metcalfe C, Donovan JL, Doble A, Goodwin L, Davis M, Catto JW, Avery K, Neal DE, Hamdy FC. Short term outcomes of prostate biopsy in men tested for cancer by prostate specific antigen: prospective evaluation within ProtecT study. BMJ (Clinical research ed.). 2012 Jan 9:344():d7894. doi: 10.1136/bmj.d7894. Epub 2012 Jan 9
[PubMed PMID: 22232535]
[32]
Berry B, Parry MG, Sujenthiran A, Nossiter J, Cowling TE, Aggarwal A, Cathcart P, Payne H, van der Meulen J, Clarke N. Comparison of complications after transrectal and transperineal prostate biopsy: a national population-based study. BJU international. 2020 Jul:126(1):97-103. doi: 10.1111/bju.15039. Epub 2020 Apr 6
[PubMed PMID: 32124525]
[33]
Sanda MG, Cadeddu JA, Kirkby E, Chen RC, Crispino T, Fontanarosa J, Freedland SJ, Greene K, Klotz LH, Makarov DV, Nelson JB, Rodrigues G, Sandler HM, Taplin ME, Treadwell JR. Clinically Localized Prostate Cancer: AUA/ASTRO/SUO Guideline. Part I: Risk Stratification, Shared Decision Making, and Care Options. The Journal of urology. 2018 Mar:199(3):683-690. doi: 10.1016/j.juro.2017.11.095. Epub 2017 Dec 15
[PubMed PMID: 29203269]
[34]
D'Amico AV, Whittington R, Malkowicz SB, Schultz D, Blank K, Broderick GA, Tomaszewski JE, Renshaw AA, Kaplan I, Beard CJ, Wein A. Biochemical outcome after radical prostatectomy, external beam radiation therapy, or interstitial radiation therapy for clinically localized prostate cancer. JAMA. 1998 Sep 16:280(11):969-74
[PubMed PMID: 9749478]
[35]
Martínez-González NA, Plate A, Markun S, Senn O, Rosemann T, Neuner-Jehle S. Shared decision making for men facing prostate cancer treatment: a systematic review of randomized controlled trials. Patient preference and adherence. 2019:13():1153-1174. doi: 10.2147/PPA.S202034. Epub 2019 Jul 17
[PubMed PMID: 31413545]
Level 1 (high-level) evidence
[36]
Brundage M, Sydes MR, Parulekar WR, Warde P, Cowan R, Bezjak A, Kirkbride P, Parliament M, Moynihan C, Bahary JP, Parmar MK, Sanders K, Chen BE, Mason MD. Impact of Radiotherapy When Added to Androgen-Deprivation Therapy for Locally Advanced Prostate Cancer: Long-Term Quality-of-Life Outcomes From the NCIC CTG PR3/MRC PR07 Randomized Trial. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 2015 Jul 1:33(19):2151-7. doi: 10.1200/JCO.2014.57.8724. Epub 2015 May 26
[PubMed PMID: 26014295]
Level 2 (mid-level) evidence
[37]
Hamdy FC, Donovan JL, Lane JA, Mason M, Metcalfe C, Holding P, Davis M, Peters TJ, Turner EL, Martin RM, Oxley J, Robinson M, Staffurth J, Walsh E, Bollina P, Catto J, Doble A, Doherty A, Gillatt D, Kockelbergh R, Kynaston H, Paul A, Powell P, Prescott S, Rosario DJ, Rowe E, Neal DE, ProtecT Study Group. 10-Year Outcomes after Monitoring, Surgery, or Radiotherapy for Localized Prostate Cancer. The New England journal of medicine. 2016 Oct 13:375(15):1415-1424. doi: 10.1056/NEJMoa1606220. Epub 2016 Sep 14
[PubMed PMID: 27626136]
[38]
Vernooij RW, Lancee M, Cleves A, Dahm P, Bangma CH, Aben KK. Radical prostatectomy versus deferred treatment for localised prostate cancer. The Cochrane database of systematic reviews. 2020 Jun 4:6(6):CD006590. doi: 10.1002/14651858.CD006590.pub3. Epub 2020 Jun 4
[PubMed PMID: 32495338]
Level 1 (high-level) evidence
[39]
Warde P, Mason M, Ding K, Kirkbride P, Brundage M, Cowan R, Gospodarowicz M, Sanders K, Kostashuk E, Swanson G, Barber J, Hiltz A, Parmar MK, Sathya J, Anderson J, Hayter C, Hetherington J, Sydes MR, Parulekar W, NCIC CTG PR.3/MRC UK PR07 investigators. Combined androgen deprivation therapy and radiation therapy for locally advanced prostate cancer: a randomised, phase 3 trial. Lancet (London, England). 2011 Dec 17:378(9809):2104-11. doi: 10.1016/S0140-6736(11)61095-7. Epub 2011 Nov 2
[PubMed PMID: 22056152]
Level 1 (high-level) evidence
[40]
Ilic D, Evans SM, Allan CA, Jung JH, Murphy D, Frydenberg M. Laparoscopic and robotic-assisted versus open radical prostatectomy for the treatment of localised prostate cancer. The Cochrane database of systematic reviews. 2017 Sep 12:9(9):CD009625. doi: 10.1002/14651858.CD009625.pub2. Epub 2017 Sep 12
[PubMed PMID: 28895658]
Level 1 (high-level) evidence
[41]
Ziehr DR, Chen MH, Zhang D, Braccioforte MH, Moran BJ, Mahal BA, Hyatt AS, Basaria SS, Beard CJ, Beckman JA, Choueiri TK, D'Amico AV, Hoffman KE, Hu JC, Martin NE, Sweeney CJ, Trinh QD, Nguyen PL. Association of androgen-deprivation therapy with excess cardiac-specific mortality in men with prostate cancer. BJU international. 2015 Sep:116(3):358-65. doi: 10.1111/bju.12905. Epub 2014 Oct 29
[PubMed PMID: 25124891]
[42]
Hickey BE, James ML, Daly T, Soh FY, Jeffery M. Hypofractionation for clinically localized prostate cancer. The Cochrane database of systematic reviews. 2019 Sep 3:9(9):CD011462. doi: 10.1002/14651858.CD011462.pub2. Epub 2019 Sep 3
[PubMed PMID: 31476800]
Level 1 (high-level) evidence
[43]
Weg ES, Pei X, Kollmeier MA, McBride SM, Zelefsky MJ. Dose-Escalated Intensity Modulated Radiation Therapy for Prostate Cancer: 15-Year Outcomes Data. Advances in radiation oncology. 2019 Jul-Sep:4(3):492-499. doi: 10.1016/j.adro.2019.03.012. Epub 2019 Apr 4
[PubMed PMID: 31360805]
Level 3 (low-level) evidence
[44]
Dutta SW, Alonso CE, Libby B, Showalter TN. Prostate cancer high dose-rate brachytherapy: review of evidence and current perspectives. Expert review of medical devices. 2018 Jan:15(1):71-79. doi: 10.1080/17434440.2018.1419058. Epub 2017 Dec 22
[PubMed PMID: 29251165]
Level 3 (low-level) evidence
[45]
Zaorsky NG, Davis BJ, Nguyen PL, Showalter TN, Hoskin PJ, Yoshioka Y, Morton GC, Horwitz EM. The evolution of brachytherapy for prostate cancer. Nature reviews. Urology. 2017 Jun 30:14(7):415-439. doi: 10.1038/nrurol.2017.76. Epub
[PubMed PMID: 28664931]
[46]
Ahdoot M, Lebastchi AH, Turkbey B, Wood B, Pinto PA. Contemporary treatments in prostate cancer focal therapy. Current opinion in oncology. 2019 May:31(3):200-206. doi: 10.1097/CCO.0000000000000515. Epub
[PubMed PMID: 30865133]
Level 3 (low-level) evidence
[47]
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2019. CA: a cancer journal for clinicians. 2019 Jan:69(1):7-34. doi: 10.3322/caac.21551. Epub 2019 Jan 8
[PubMed PMID: 30620402]