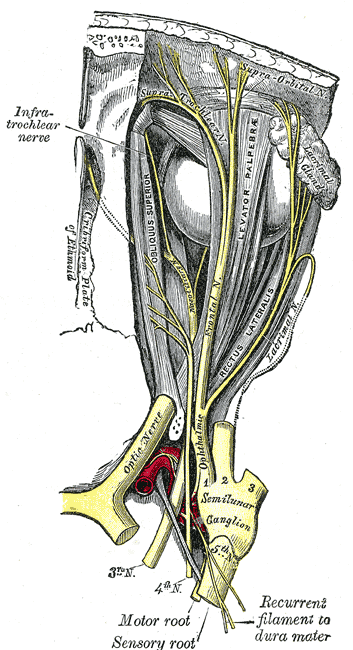
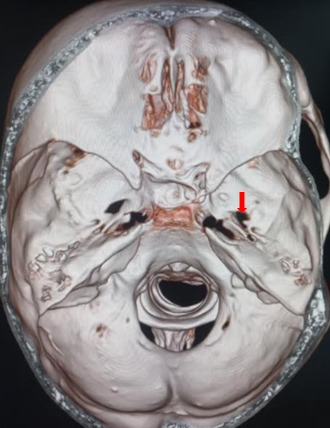
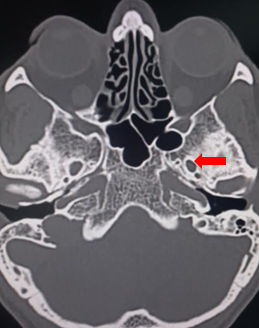
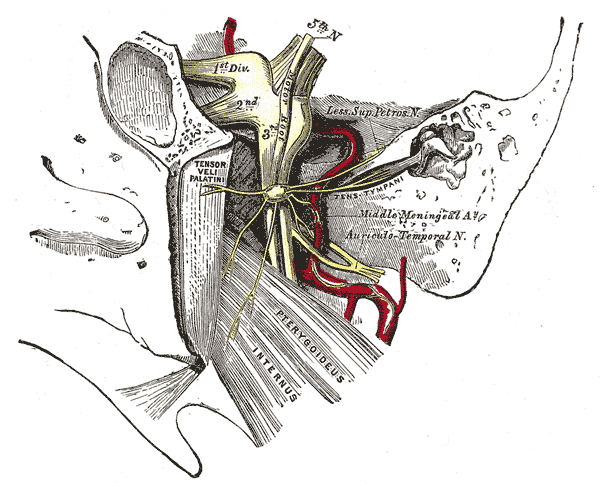
[1]
Yousry I, Moriggl B, Schmid UD, Naidich TP, Yousry TA. Trigeminal ganglion and its divisions: detailed anatomic MR imaging with contrast-enhanced 3D constructive interference in the steady state sequences. AJNR. American journal of neuroradiology. 2005 May:26(5):1128-35
[PubMed PMID: 15891171]
[2]
Messlinger K, Russo AF. Current understanding of trigeminal ganglion structure and function in headache. Cephalalgia : an international journal of headache. 2019 Nov:39(13):1661-1674. doi: 10.1177/0333102418786261. Epub 2018 Jul 10
[PubMed PMID: 29989427]
Level 3 (low-level) evidence
[3]
Youssef S, Kim EY, Aziz KM, Hemida S, Keller JT, van Loveren HR. The subtemporal interdural approach to dumbbell-shaped trigeminal schwannomas: cadaveric prosection. Neurosurgery. 2006 Oct:59(4 Suppl 2):ONS270-7; discussion ONS277-8
[PubMed PMID: 17041497]
[4]
Malhotra A, Tu L, Kalra VB, Wu X, Mian A, Mangla R, Michaelides E, Sanelli P, Gandhi D. Neuroimaging of Meckel's cave in normal and disease conditions. Insights into imaging. 2018 Aug:9(4):499-510. doi: 10.1007/s13244-018-0604-7. Epub 2018 Apr 18
[PubMed PMID: 29671218]
[5]
Sabancı PA, Batay F, Civelek E, Al Mefty O, Husain M, Abdulrauf SI, Karasu A. Meckel's cave. World neurosurgery. 2011 Sep-Oct:76(3-4):335-41; discussion 266-7. doi: 10.1016/j.wneu.2011.03.037. Epub
[PubMed PMID: 21986433]
[6]
Mortazavi MM, Cox MA, Saker E, Krishnamurthy S, Verma K, Griessenauer CJ, Loukas M, Oskouian RJ, Tubbs RS. The superior petrosal sinus: a review of anatomy, embryology, pathology, and neurosurgical relevance. Neurosurgical review. 2018 Jul:41(3):713-718. doi: 10.1007/s10143-016-0785-9. Epub 2016 Sep 19
[PubMed PMID: 27647276]
[7]
Shankland WE 2nd. The trigeminal nerve. Part III: The maxillary division. Cranio : the journal of craniomandibular practice. 2001 Apr:19(2):78-83
[PubMed PMID: 11842868]
[8]
Shankland WE 2nd. The trigeminal nerve. Part I: An over-view. Cranio : the journal of craniomandibular practice. 2000 Oct:18(4):238-48
[PubMed PMID: 11202843]
[9]
LaGuardia JJ, Cohrs RJ, Gilden DH. Numbers of neurons and non-neuronal cells in human trigeminal ganglia. Neurological research. 2000 Sep:22(6):565-6
[PubMed PMID: 11045016]
[10]
Carmine Belin A, Ran C, Edvinsson L. Calcitonin Gene-Related Peptide (CGRP) and Cluster Headache. Brain sciences. 2020 Jan 6:10(1):. doi: 10.3390/brainsci10010030. Epub 2020 Jan 6
[PubMed PMID: 31935868]
[11]
Freter S, Fleenor SJ, Freter R, Liu KJ, Begbie J. Cranial neural crest cells form corridors prefiguring sensory neuroblast migration. Development (Cambridge, England). 2013 Sep:140(17):3595-600. doi: 10.1242/dev.091033. Epub
[PubMed PMID: 23942515]
[12]
Xu H, Dude CM, Baker CV. Fine-grained fate maps for the ophthalmic and maxillomandibular trigeminal placodes in the chick embryo. Developmental biology. 2008 May 1:317(1):174-86. doi: 10.1016/j.ydbio.2008.02.012. Epub 2008 Feb 21
[PubMed PMID: 18367162]
[13]
Ćetković M, Štimec BV, Mucić D, Dožić A, Ćetković D, Reçi V, Çerkezi S, Ćalasan D, Milisavljević M, Bexheti S. Arterial supply of the trigeminal ganglion, a micromorphological study. Folia morphologica. 2020:79(1):58-64. doi: 10.5603/FM.a2019.0062. Epub 2019 Jul 8
[PubMed PMID: 31282551]
[14]
Dankbaar JW, Pameijer FA, Hendrikse J, Schmalfuss IM. Easily detected signs of perineural tumour spread in head and neck cancer. Insights into imaging. 2018 Dec:9(6):1089-1095. doi: 10.1007/s13244-018-0672-8. Epub 2018 Nov 16
[PubMed PMID: 30446949]
[15]
Gambeta E, Chichorro JG, Zamponi GW. Trigeminal neuralgia: An overview from pathophysiology to pharmacological treatments. Molecular pain. 2020 Jan-Dec:16():1744806920901890. doi: 10.1177/1744806920901890. Epub
[PubMed PMID: 31908187]
Level 3 (low-level) evidence
[16]
Montano N, Ioannoni E, Rapisarda A. The risk of mastication weakness after percutaneous balloon compression for the treatment of trigeminal neuralgia. Clinical neurology and neurosurgery. 2020 Aug:195():105880. doi: 10.1016/j.clineuro.2020.105880. Epub 2020 May 4
[PubMed PMID: 32413677]
[17]
Belber CJ, Rak RA. Balloon compression rhizolysis in the surgical management of trigeminal neuralgia. Neurosurgery. 1987 Jun:20(6):908-13
[PubMed PMID: 3614571]
[18]
Olivero WC, Wang H, Rak R, Sharrock MF. Percutaneous balloon rhizotomy for trigeminal neuralgia using three-dimensional fluoroscopy. World neurosurgery. 2012 Jan:77(1):202.e1-3. doi: 10.1016/j.wneu.2011.03.041. Epub 2011 Nov 18
[PubMed PMID: 22405398]
[19]
Skirving DJ, Dan NG. A 20-year review of percutaneous balloon compression of the trigeminal ganglion. Journal of neurosurgery. 2001 Jun:94(6):913-7
[PubMed PMID: 11409519]
[20]
Iwanaga J, Badaloni F, Laws T, Oskouian RJ, Tubbs RS. Anatomic Study of Extracranial Needle Trajectory Using Hartel Technique for Percutaneous Treatment of Trigeminal Neuralgia. World neurosurgery. 2018 Feb:110():e245-e248. doi: 10.1016/j.wneu.2017.10.140. Epub 2017 Nov 22
[PubMed PMID: 29104153]
[21]
Baabor MG, Perez-Limonte L. Percutaneous balloon compression of the gasserian ganglion for the treatment of trigeminal neuralgia: personal experience of 206 patients. Acta neurochirurgica. Supplement. 2011:108():251-4. doi: 10.1007/978-3-211-99370-5_39. Epub
[PubMed PMID: 21107968]
[22]
Harries AM, Mitchell RD. Percutaneous glycerol rhizotomy for trigeminal neuralgia: safety and efficacy of repeat procedures. British journal of neurosurgery. 2011 Apr:25(2):268-72. doi: 10.3109/02688697.2011.558946. Epub
[PubMed PMID: 21545328]
[23]
Wang Z, Wang Z, Li K, Su X, Du C, Tian Y. Radiofrequency thermocoagulation for the treatment of trigeminal neuralgia. Experimental and therapeutic medicine. 2022 Jan:23(1):17. doi: 10.3892/etm.2021.10939. Epub 2021 Oct 30
[PubMed PMID: 34815769]
[24]
Cheng JS, Lim DA, Chang EF, Barbaro NM. A review of percutaneous treatments for trigeminal neuralgia. Neurosurgery. 2014 Mar:10 Suppl 1():25-33; discussion 33. doi: 10.1227/NEU.00000000000001687. Epub
[PubMed PMID: 24509496]
[25]
Udupi BP, Chouhan RS, Dash HH, Bithal PK, Prabhakar H. Comparative evaluation of percutaneous retrogasserian glycerol rhizolysis and radiofrequency thermocoagulation techniques in the management of trigeminal neuralgia. Neurosurgery. 2012 Feb:70(2):407-12; discussion 412-3. doi: 10.1227/NEU.0b013e318233a85f. Epub
[PubMed PMID: 21866065]
Level 2 (mid-level) evidence
[26]
Texakalidis P, Xenos D, Tora MS, Wetzel JS, Boulis NM. Comparative safety and efficacy of percutaneous approaches for the treatment of trigeminal neuralgia: A systematic review and meta-analysis. Clinical neurology and neurosurgery. 2019 Jul:182():112-122. doi: 10.1016/j.clineuro.2019.05.011. Epub 2019 May 14
[PubMed PMID: 31121470]
Level 2 (mid-level) evidence
[27]
Noorani I, Lodge A, Durnford A, Vajramani G, Sparrow O. Comparison of first-time microvascular decompression with percutaneous surgery for trigeminal neuralgia: long-term outcomes and prognostic factors. Acta neurochirurgica. 2021 Jun:163(6):1623-1634. doi: 10.1007/s00701-021-04793-4. Epub 2021 Mar 22
[PubMed PMID: 33751217]
[28]
Hannan C, Shoakazemi A, Quigley G. Microvascular Decompression for Trigeminal Neuralgia: A regional unit's experience. The Ulster medical journal. 2018 Jan:87(1):30-33
[PubMed PMID: 29588554]
[29]
Yuan H, Spare NM, Silberstein SD. Targeting CGRP for the Prevention of Migraine and Cluster Headache: A Narrative Review. Headache. 2019 Jul:59 Suppl 2():20-32. doi: 10.1111/head.13583. Epub
[PubMed PMID: 31291020]
Level 3 (low-level) evidence
[30]
Goadsby PJ. Pathophysiology of cluster headache: a trigeminal autonomic cephalgia. The Lancet. Neurology. 2002 Aug:1(4):251-7
[PubMed PMID: 12849458]
[31]
Theil D, Derfuss T, Paripovic I, Herberger S, Meinl E, Schueler O, Strupp M, Arbusow V, Brandt T. Latent herpesvirus infection in human trigeminal ganglia causes chronic immune response. The American journal of pathology. 2003 Dec:163(6):2179-84
[PubMed PMID: 14633592]
[32]
Fredeking AE, Silverman RA. Successful treatment of trigeminal trophic syndrome in a 6-year-old boy with negative pressure wound therapy. Archives of dermatology. 2008 Aug:144(8):984-6. doi: 10.1001/archderm.144.8.984. Epub
[PubMed PMID: 18711069]