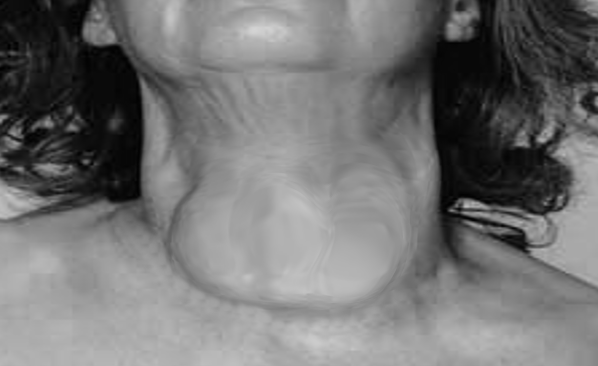
[2]
Swain M,Swain T,Mohanty BK, Autoimmune thyroid disorders-An update. Indian journal of clinical biochemistry : IJCB. 2005 Jan;
[PubMed PMID: 23105486]
[3]
Knobel M, Etiopathology, clinical features, and treatment of diffuse and multinodular nontoxic goiters. Journal of endocrinological investigation. 2016 Apr;
[PubMed PMID: 26392367]
[5]
Jereczek-Fossa BA,Alterio D,Jassem J,Gibelli B,Tradati N,Orecchia R, Radiotherapy-induced thyroid disorders. Cancer treatment reviews. 2004 Jun;
[PubMed PMID: 15145511]
[6]
Prabhakar BS,Bahn RS,Smith TJ, Current perspective on the pathogenesis of Graves' disease and ophthalmopathy. Endocrine reviews. 2003 Dec;
[PubMed PMID: 14671007]
Level 3 (low-level) evidence
[8]
Oertli D,Harder F,Oberholzer M,Staub JJ, [Hyperthyroidism and thyroid carcinoma--coincidence or association?]. Schweizerische medizinische Wochenschrift. 1998 Nov 28;
[PubMed PMID: 9879620]
[10]
Baskin HJ,Cobin RH,Duick DS,Gharib H,Guttler RB,Kaplan MM,Segal RL, American Association of Clinical Endocrinologists medical guidelines for clinical practice for the evaluation and treatment of hyperthyroidism and hypothyroidism. Endocrine practice : official journal of the American College of Endocrinology and the American Association of Clinical Endocrinologists. 2002 Nov-Dec;
[PubMed PMID: 15260011]
[11]
Chung JH, Antithyroid Drug Treatment in Graves' Disease. Endocrinology and metabolism (Seoul, Korea). 2021 Jun
[PubMed PMID: 34130446]
[12]
Allannic H,Fauchet R,Orgiazzi J,Madec AM,Genetet B,Lorcy Y,Le Guerrier AM,Delambre C,Derennes V, Antithyroid drugs and Graves' disease: a prospective randomized evaluation of the efficacy of treatment duration. The Journal of clinical endocrinology and metabolism. 1990 Mar;
[PubMed PMID: 1689737]
Level 1 (high-level) evidence
[13]
Abraham P,Avenell A,McGeoch SC,Clark LF,Bevan JS, Antithyroid drug regimen for treating Graves' hyperthyroidism. The Cochrane database of systematic reviews. 2010 Jan 20;
[PubMed PMID: 20091544]
Level 1 (high-level) evidence
[14]
Azizi F,Abdi H,Amouzegar A,Habibi Moeini AS, Long-term thionamide antithyroid treatment of Graves' disease. Best practice & research. Clinical endocrinology & metabolism. 2022 Feb 24
[PubMed PMID: 35440398]
[15]
Girgis CM,Champion BL,Wall JR, Current concepts in graves' disease. Therapeutic advances in endocrinology and metabolism. 2011 Jun
[PubMed PMID: 23148179]
Level 3 (low-level) evidence
[17]
Lavruk KZ,Dudiy PF,Skrypnyk NV,Mishchuk VH,Vytvytskiy ZY, Clinical-laboratory and ultrasound parallels of changes in the liver and thyroid gland in diffuse toxic goiter. Journal of medicine and life. 2022 Jan
[PubMed PMID: 35186140]