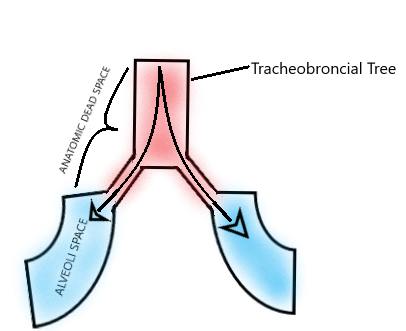
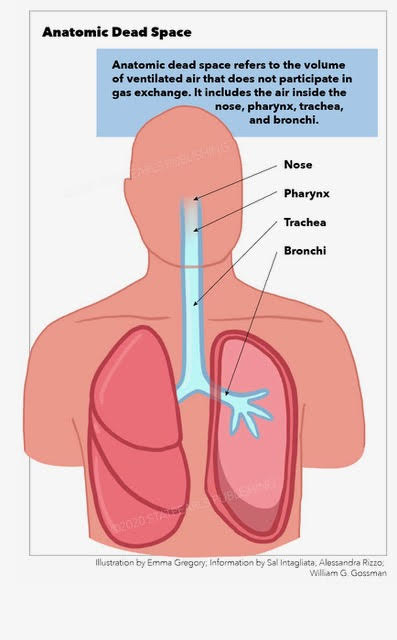
[1]
Robertson HT, Dead space: the physiology of wasted ventilation. The European respiratory journal. 2015 Jun
[PubMed PMID: 25395032]
[2]
Nuckton TJ,Alonso JA,Kallet RH,Daniel BM,Pittet JF,Eisner MD,Matthay MA, Pulmonary dead-space fraction as a risk factor for death in the acute respiratory distress syndrome. The New England journal of medicine. 2002 Apr 25
[PubMed PMID: 11973365]
[3]
Doorduin J,Nollet JL,Vugts MP,Roesthuis LH,Akankan F,van der Hoeven JG,van Hees HW,Heunks LM, Assessment of dead-space ventilation in patients with acute respiratory distress syndrome: a prospective observational study. Critical care (London, England). 2016 May 5
[PubMed PMID: 27145818]
Level 2 (mid-level) evidence
[4]
Verscheure S,Massion PB,Verschuren F,Damas P,Magder S, Volumetric capnography: lessons from the past and current clinical applications. Critical care (London, England). 2016 Jun 23;
[PubMed PMID: 27334879]
[5]
Hedenstierna G,Sandhagen B, Assessing dead space. A meaningful variable? Minerva anestesiologica. 2006 Jun
[PubMed PMID: 16682925]
[6]
Farkas EA,Detterbeck FC, Airway complications after pulmonary resection. Thoracic surgery clinics. 2006 Aug
[PubMed PMID: 17004552]
[7]
Bourgoin P,Baudin F,Brossier D,Emeriaud G,Wysocki M,Jouvet P, Assessment of Bohr and Enghoff Dead Space Equations in Mechanically Ventilated Children. Respiratory care. 2017 Apr
[PubMed PMID: 28223465]
[8]
Keck T,Leiacker R,Heinrich A,Kühnemann S,Rettinger G, Humidity and temperature profile in the nasal cavity. Rhinology. 2000 Dec
[PubMed PMID: 11190750]
[9]
Plotnikow GA,Accoce M,Navarro E,Tiribelli N, Humidification and heating of inhaled gas in patients with artificial airway. A narrative review. Revista Brasileira de terapia intensiva. 2018 Mar
[PubMed PMID: 29742220]
Level 3 (low-level) evidence
[10]
Gehlbach JA,Miller AG,Hornik CP,Cheifetz IM, Dead Space to Tidal Volume Ratio Is Associated With Higher Postextubation Support in Children. Respiratory care. 2020 Nov;
[PubMed PMID: 32606073]
[11]
Smith LJ,Macleod KA,Collier GJ,Horn FC,Sheridan H,Aldag I,Taylor CJ,Cunningham S,Wild JM,Horsley A, Supine posture changes lung volumes and increases ventilation heterogeneity in cystic fibrosis. PloS one. 2017
[PubMed PMID: 29176899]
[12]
Biselli P,Fricke K,Grote L,Braun AT,Kirkness J,Smith P,Schwartz A,Schneider H, Reductions in dead space ventilation with nasal high flow depend on physiological dead space volume: metabolic hood measurements during sleep in patients with COPD and controls. The European respiratory journal. 2018 May
[PubMed PMID: 29724917]
[13]
Mündel T,Feng S,Tatkov S,Schneider H, Mechanisms of nasal high flow on ventilation during wakefulness and sleep. Journal of applied physiology (Bethesda, Md. : 1985). 2013 Apr
[PubMed PMID: 23412897]
[14]
Tang Y,Turner MJ,Baker AB, Effects of lung time constant, gas analyser delay and rise time on measurements of respiratory dead-space. Physiological measurement. 2005 Dec
[PubMed PMID: 16311457]
[15]
Blanch L,Romero PV,Lucangelo U, Volumetric capnography in the mechanically ventilated patient. Minerva anestesiologica. 2006 Jun
[PubMed PMID: 16682932]
[16]
Miller DM,Adams AP,Light D, Dead space and paediatric anaesthetic equipment: a physical lung model study. Anaesthesia. 2004 Jun
[PubMed PMID: 15144302]
[17]
Chuang ML, Combining Dynamic Hyperinflation with Dead Space Volume during Maximal Exercise in Patients with Chronic Obstructive Pulmonary Disease. Journal of clinical medicine. 2020 Apr 15;
[PubMed PMID: 32326507]
[18]
Romero PV,Rodriguez B,de Oliveira D,Blanch L,Manresa F, Volumetric capnography and chronic obstructive pulmonary disease staging. International journal of chronic obstructive pulmonary disease. 2007;
[PubMed PMID: 18229577]
[19]
Raurich JM,Vilar M,Colomar A,Ibáñez J,Ayestarán I,Pérez-Bárcena J,Llompart-Pou JA, Prognostic value of the pulmonary dead-space fraction during the early and intermediate phases of acute respiratory distress syndrome. Respiratory care. 2010 Mar
[PubMed PMID: 20196876]
[20]
Kline JA,Israel EG,Michelson EA,O'Neil BJ,Plewa MC,Portelli DC, Diagnostic accuracy of a bedside D-dimer assay and alveolar dead-space measurement for rapid exclusion of pulmonary embolism: a multicenter study. JAMA. 2001 Feb 14
[PubMed PMID: 11176914]
Level 2 (mid-level) evidence
[21]
Verschuren F,Heinonen E,Clause D,Roeseler J,Thys F,Meert P,Marion E,El Gariani A,Col J,Reynaert M,Liistro G, Volumetric capnography as a bedside monitoring of thrombolysis in major pulmonary embolism. Intensive care medicine. 2004 Nov
[PubMed PMID: 15378240]
[22]
Hinkson CR,Benson MS,Stephens LM,Deem S, The effects of apparatus dead space on P(aCO2) in patients receiving lung-protective ventilation. Respiratory care. 2006 Oct;
[PubMed PMID: 17005059]
[23]
Tusman G,Gogniat E,Madorno M,Otero P,Dianti J,Ceballos IF,Ceballos M,Verdier N,Böhm SH,Rodriguez PO,San Roman E, Effect of PEEP on Dead Space in an Experimental Model of ARDS. Respiratory care. 2020 Jan
[PubMed PMID: 31615922]
[24]
González-Castro A,Suárez-Lopez V,Gómez-Marcos V,González-Fernandez C,Iglesias-Posadilla D,Burón-Mediavilla J,Rodríguez-Borregan JC,Miñambres E,Llorca J, [Utility of the dead space fraction (Vd/Vt) as a predictor of extubation success]. Medicina intensiva. 2011 Dec;
[PubMed PMID: 21782289]
[25]
Hubble CL,Gentile MA,Tripp DS,Craig DM,Meliones JN,Cheifetz IM, Deadspace to tidal volume ratio predicts successful extubation in infants and children. Critical care medicine. 2000 Jun
[PubMed PMID: 10890660]
[26]
Dassios T,Dixon P,Hickey A,Fouzas S,Greenough A, Physiological and anatomical dead space in mechanically ventilated newborn infants. Pediatric pulmonology. 2018 Jan
[PubMed PMID: 29152912]
[27]
Praetel C,Banner MJ,Monk T,Gabrielli A, Isoflurane inhalation enhances increased physiologic deadspace volume associated with positive pressure ventilation and compromises arterial oxygenation. Anesthesia and analgesia. 2004 Oct
[PubMed PMID: 15385359]
[28]
Spoletini G,Alotaibi M,Blasi F,Hill NS, Heated Humidified High-Flow Nasal Oxygen in Adults: Mechanisms of Action and Clinical Implications. Chest. 2015 Jul;
[PubMed PMID: 25742321]
[29]
Möller W,Feng S,Domanski U,Franke KJ,Celik G,Bartenstein P,Becker S,Meyer G,Schmid O,Eickelberg O,Tatkov S,Nilius G, Nasal high flow reduces dead space. Journal of applied physiology (Bethesda, Md. : 1985). 2017 Jan 1
[PubMed PMID: 27856714]