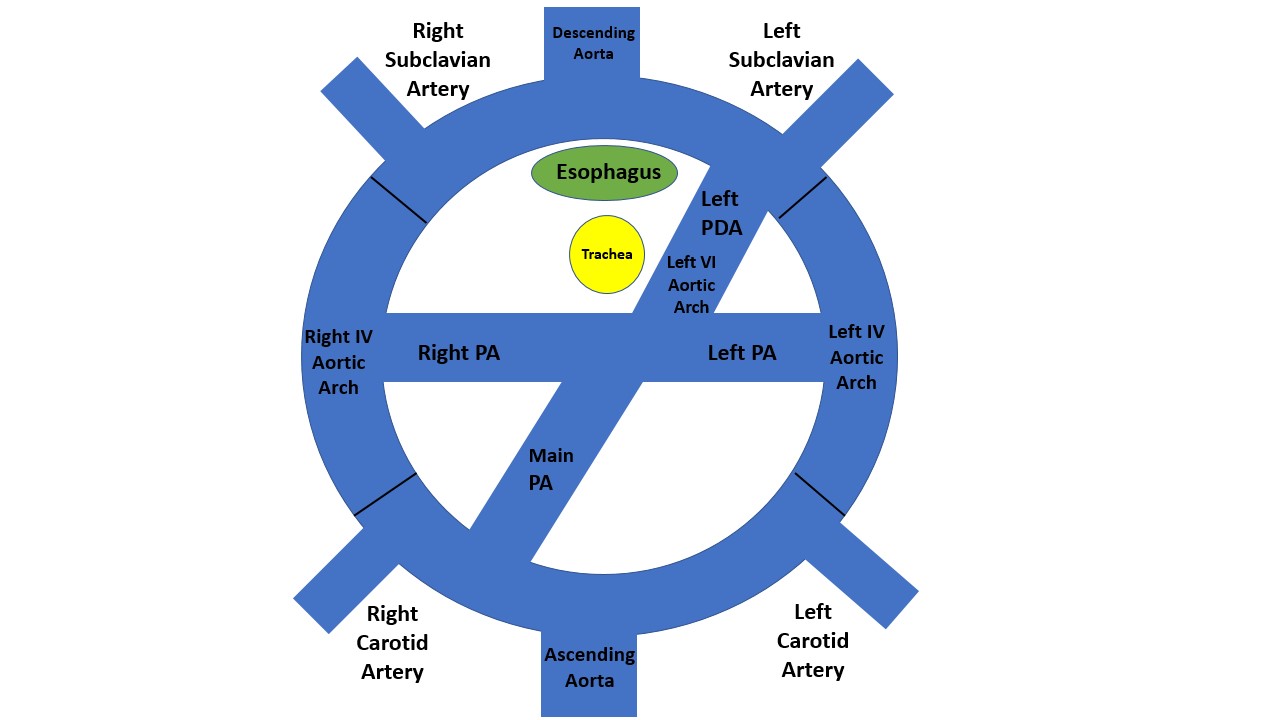
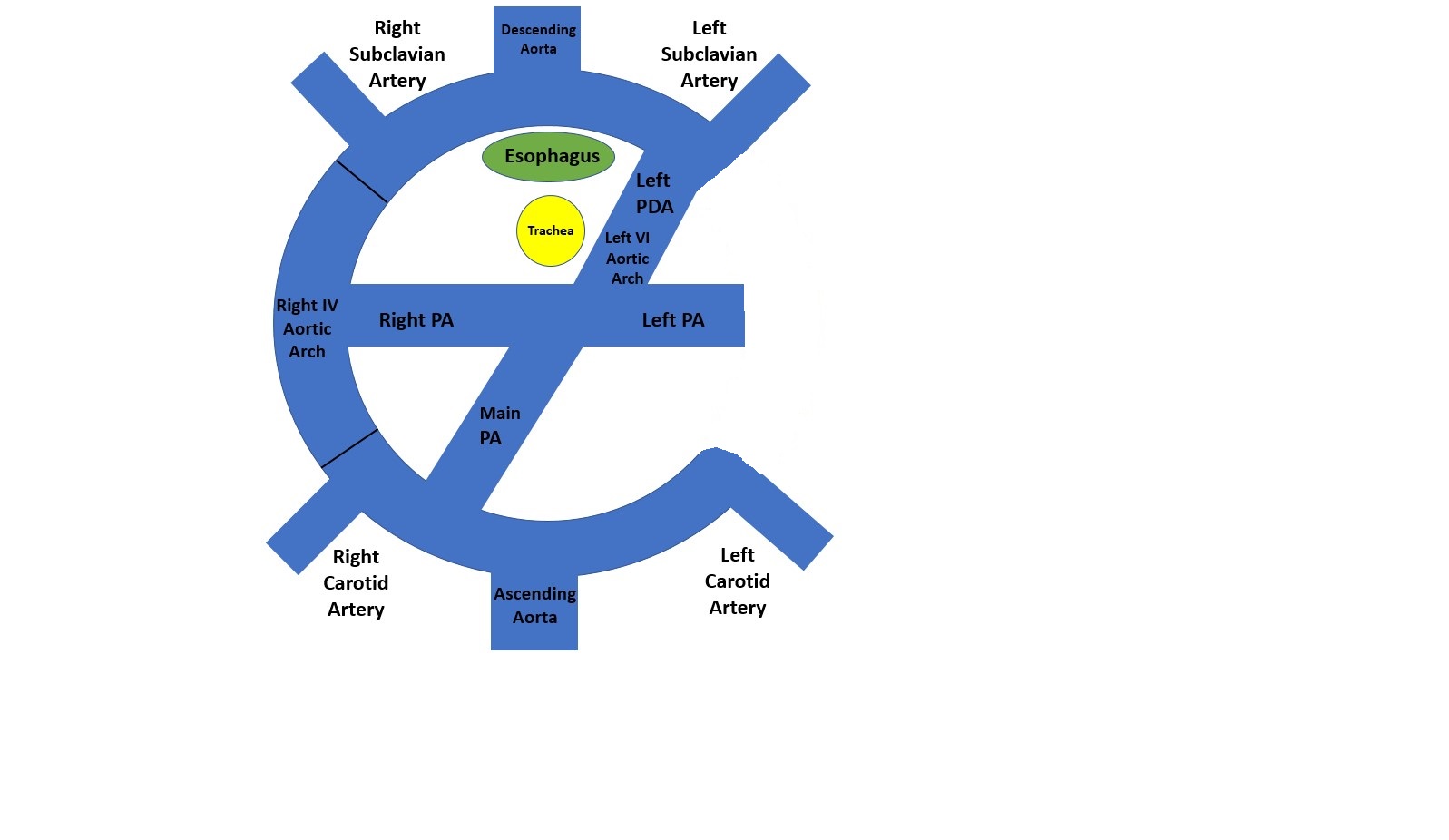
[1]
Backer CL, Mavroudis C. Congenital Heart Surgery Nomenclature and Database Project: vascular rings, tracheal stenosis, pectus excavatum. The Annals of thoracic surgery. 2000 Apr:69(4 Suppl):S308-18
[PubMed PMID: 10798437]
[2]
Kellenberger CJ. Aortic arch malformations. Pediatric radiology. 2010 Jun:40(6):876-84. doi: 10.1007/s00247-010-1607-9. Epub 2010 Mar 31
[PubMed PMID: 20354848]
[3]
Licari A, Manca E, Rispoli GA, Mannarino S, Pelizzo G, Marseglia GL. Congenital vascular rings: a clinical challenge for the pediatrician. Pediatric pulmonology. 2015 May:50(5):511-24. doi: 10.1002/ppul.23152. Epub 2015 Jan 20
[PubMed PMID: 25604054]
[4]
Türkvatan A, Büyükbayraktar FG, Olçer T, Cumhur T. Multidetector computed tomographic angiography of aberrant subclavian arteries. Vascular medicine (London, England). 2009 Feb:14(1):5-11. doi: 10.1177/1358863X08097903. Epub
[PubMed PMID: 19144774]
[5]
Tanaka A, Milner R, Ota T. Kommerell's diverticulum in the current era: a comprehensive review. General thoracic and cardiovascular surgery. 2015 May:63(5):245-59. doi: 10.1007/s11748-015-0521-3. Epub 2015 Jan 31
[PubMed PMID: 25636900]
[6]
McElhinney DB, Clark BJ 3rd, Weinberg PM, Kenton ML, McDonald-McGinn D, Driscoll DA, Zackai EH, Goldmuntz E. Association of chromosome 22q11 deletion with isolated anomalies of aortic arch laterality and branching. Journal of the American College of Cardiology. 2001 Jun 15:37(8):2114-9
[PubMed PMID: 11419896]
[7]
Alsenaidi K, Gurofsky R, Karamlou T, Williams WG, McCrindle BW. Management and outcomes of double aortic arch in 81 patients. Pediatrics. 2006 Nov:118(5):e1336-41
[PubMed PMID: 17000782]
[8]
Shirali GS, Geva T, Ott DA, Bricker JT. Double aortic arch and bilateral patent ducti arteriosi associated with transposition of the great arteries: missing clinical link in an embryologic theory. American heart journal. 1994 Feb:127(2):451-3
[PubMed PMID: 8296720]
[9]
Hastreiter AR, D'Cruz IA, Cantez T, Namin EP, Licata R. Right-sided aorta. I. Occurrence of right aortic arch in various types of congenital heart disease. II. Right aortic arch, right descending aorta, and associated anomalies. British heart journal. 1966 Nov:28(6):722-39
[PubMed PMID: 5332779]
[10]
Lowe GM, Donaldson JS, Backer CL. Vascular rings: 10-year review of imaging. Radiographics : a review publication of the Radiological Society of North America, Inc. 1991 Jul:11(4):637-46
[PubMed PMID: 1887119]
[11]
EDWARDS JE. Anomalies of the derivatives of the aortic arch system. The Medical clinics of North America. 1948 Jul:32():925-49
[PubMed PMID: 18877614]
[12]
Cui W, Patel D, Husayni TS, Roberson DA. Double aortic arch and d-transposition of the great arteries. Echocardiography (Mount Kisco, N.Y.). 2008 Jan:25(1):91-5. doi: 10.1111/j.1540-8175.2007.00554.x. Epub
[PubMed PMID: 18186786]
[13]
Yıldırım SV, Yıldırım A. Truncus arteriosus with double aortic arch: A rare association. The Turkish journal of pediatrics. 2017:59(2):221-223. doi: 10.24953/turkjped.2017.02.020. Epub
[PubMed PMID: 29276881]
[14]
Backer CL, Mongé MC, Popescu AR, Eltayeb OM, Rastatter JC, Rigsby CK. Vascular rings. Seminars in pediatric surgery. 2016 Jun:25(3):165-75. doi: 10.1053/j.sempedsurg.2016.02.009. Epub 2016 Feb 22
[PubMed PMID: 27301603]
[15]
Hunter L, Callaghan N, Patel K, Rinaldi L, Bellsham-Revell H, Sharland G. Prenatal echocardiographic diagnosis of double aortic arch. Ultrasound in obstetrics & gynecology : the official journal of the International Society of Ultrasound in Obstetrics and Gynecology. 2015 Apr:45(4):483-5. doi: 10.1002/uog.13408. Epub 2015 Mar 9
[PubMed PMID: 24817195]
[16]
Cantinotti M, Hegde S, Bell A, Razavi R. Diagnostic role of magnetic resonance imaging in identifying aortic arch anomalies. Congenital heart disease. 2008 Mar-Apr:3(2):117-23. doi: 10.1111/j.1747-0803.2008.00174.x. Epub
[PubMed PMID: 18380760]
[17]
Bisset GS 3rd, Strife JL, Kirks DR, Bailey WW. Vascular rings: MR imaging. AJR. American journal of roentgenology. 1987 Aug:149(2):251-6
[PubMed PMID: 3496746]
[18]
Lillehei CW, Colan S. Echocardiography in the preoperative evaluation of vascular rings. Journal of pediatric surgery. 1992 Aug:27(8):1118-20; discussion 1120-1
[PubMed PMID: 1403546]
[19]
Wychulis AR, Kincaid OW, Weidman WH, Danielson GK. Congenital vascular ring: surgical considerations and results of operation. Mayo Clinic proceedings. 1971 Mar:46(3):182-8
[PubMed PMID: 5553129]
[20]
Koontz CS, Bhatia A, Forbess J, Wulkan ML. Video-assisted thoracoscopic division of vascular rings in pediatric patients. The American surgeon. 2005 Apr:71(4):289-91
[PubMed PMID: 15943400]
[21]
Shah RK, Mora BN, Bacha E, Sena LM, Buonomo C, Del Nido P, Rahbar R. The presentation and management of vascular rings: an otolaryngology perspective. International journal of pediatric otorhinolaryngology. 2007 Jan:71(1):57-62
[PubMed PMID: 17034866]
Level 3 (low-level) evidence
[22]
Backer CL, Mongé MC, Russell HM, Popescu AR, Rastatter JC, Costello JM. Reoperation after vascular ring repair. Seminars in thoracic and cardiovascular surgery. Pediatric cardiac surgery annual. 2014:17(1):48-55. doi: 10.1053/j.pcsu.2014.01.001. Epub
[PubMed PMID: 24725717]
[23]
Schmidt AMS, Larsen SH, Hjortdal VE. Vascular ring: Early and long-term mortality and morbidity after surgical repair. Journal of pediatric surgery. 2018 Oct:53(10):1976-1979. doi: 10.1016/j.jpedsurg.2017.12.022. Epub 2018 Jan 3
[PubMed PMID: 29402450]
[24]
Saran N, Dearani J, Said S, Fatima B, Schaff H, Bower T, Pochettino A. Vascular Rings in Adults: Outcome of Surgical Management. The Annals of thoracic surgery. 2019 Oct:108(4):1217-1227. doi: 10.1016/j.athoracsur.2019.04.097. Epub 2019 Jun 20
[PubMed PMID: 31229482]
[25]
Tola H, Ozturk E, Yildiz O, Erek E, Haydin S, Turkvatan A, Ergul Y, Guzeltas A, Bakir I. Assessment of children with vascular ring. Pediatrics international : official journal of the Japan Pediatric Society. 2017 Feb:59(2):134-140. doi: 10.1111/ped.13101. Epub 2016 Oct 31
[PubMed PMID: 27454661]
[26]
Othersen HB Jr, Khalil B, Zellner J, Sade R, Handy J, Tagge EP, Smith CD. Aortoesophageal fistula and double aortic arch: two important points in management. Journal of pediatric surgery. 1996 Apr:31(4):594-5
[PubMed PMID: 8801321]
[27]
van Son JA, Julsrud PR, Hagler DJ, Sim EK, Pairolero PC, Puga FJ, Schaff HV, Danielson GK. Surgical treatment of vascular rings: the Mayo Clinic experience. Mayo Clinic proceedings. 1993 Nov:68(11):1056-63
[PubMed PMID: 8231269]
[28]
Anand R, Dooley KJ, Williams WH, Vincent RN. Follow-up of surgical correction of vascular anomalies causing tracheobronchial compression. Pediatric cardiology. 1994 Mar-Apr:15(2):58-61
[PubMed PMID: 7997414]
[29]
Chun K, Colombani PM, Dudgeon DL, Haller JA Jr. Diagnosis and management of congenital vascular rings: a 22-year experience. The Annals of thoracic surgery. 1992 Apr:53(4):597-602; discussion 602-3
[PubMed PMID: 1554267]
[30]
Fleck RJ, Pacharn P, Fricke BL, Ziegler MA, Cotton RT, Donnelly LF. Imaging findings in pediatric patients with persistent airway symptoms after surgery for double aortic arch. AJR. American journal of roentgenology. 2002 May:178(5):1275-9
[PubMed PMID: 11959745]