Introduction
The somatosensory nervous system maintains the sensation within the various dermatomes of sensation throughout the body. The somatosensory pathway serves as the conduit between the different sensory modalities within the body, sending information from the periphery to the postcentral gyrus and associated cortices to convey information from the surrounding environment. Peripheral mechanoreceptors, chemoreceptors, thermoreceptors, and nociceptors transmit information via specific nerve fibers that enter the central nervous system tracts towards the brain. Each carrying a unique modality of sensation, these senses are ultimately processed and integrated into the central nervous system to help individuals interact with their environmental stimuli. The ultimate destination of these sensory stimuli is the somatosensory cortex.
Structure and Function
Structure of the Somatosensory Cortex
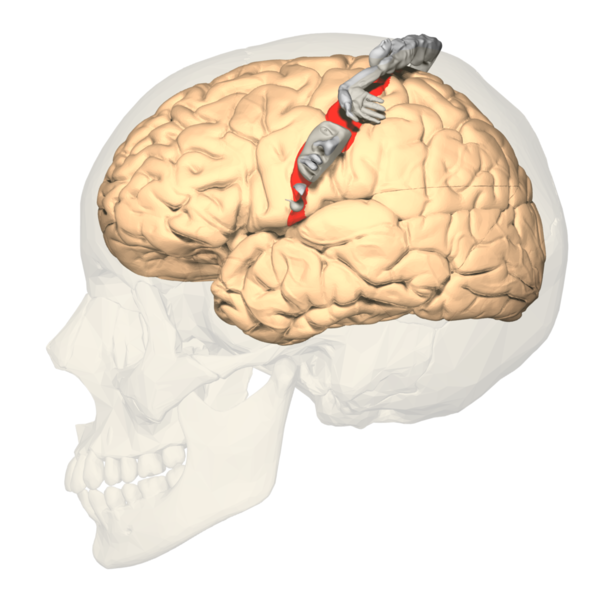
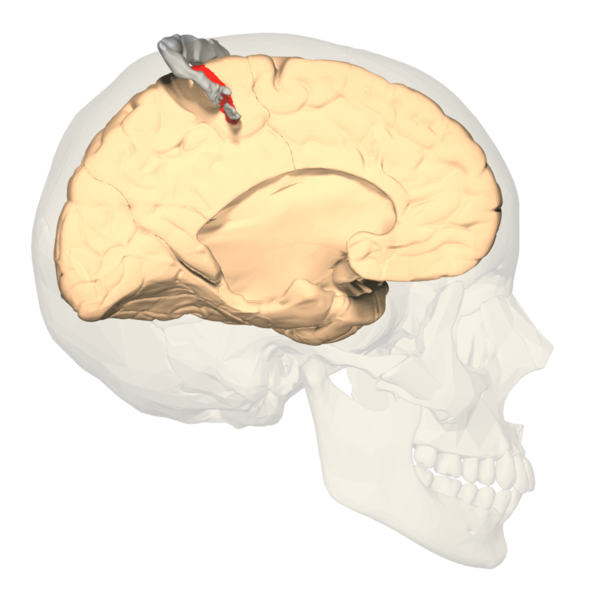
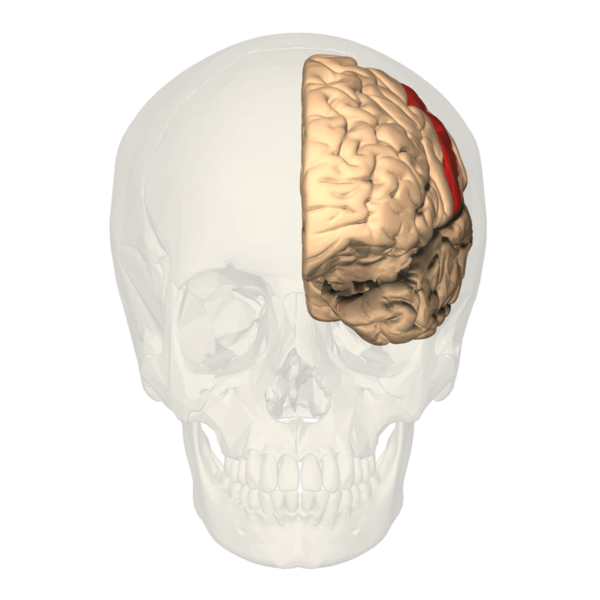
The primary somatosensory cortex (S1) and the secondary somatosensory cortex (S2) are in the parietal lobe of the brain (see Images. Primary Somatosensory Cortex Lateral View, Cortical Homunculus and Cortical Homunculus, Primary Somatosensory Cortex).
The primary somatosensory cortex is found just behind the central sulcus and receives sensory information from the VPL of the thalamus via the internal capsule and corona radiata. Often referred to as the postcentral gyrus, this area receives the sensory input from specific areas of the body, correlating to specific regions of the contralateral postcentral gyrus. This is known as somatotopy and is used to construct maps such as the sensory homunculus, as described in further detail below. Much like the rest of the neocortex, each primary cortex os paired association cortices to help interpret and assess the initial sensory information delivered. A larger association cortex is needed to assess more complex input. The S2 region helps to process the sensory information delivered to S1. The somatosensory cortex is organized into six layers, much like the rest of the neocortex. The sensory and motor layers tend to be more heterogeneous and differ in the sizes of layer four (internal granular) and layer 5 (internal pyramidal). Layer four is associated with more input, and layer five is associated with more output. Thus the primary somatosensory cortex receiving peripheral input will tend to have a larger fourth layer and smaller fifth layer. The opposite is true for the motor cortex. The association cortices tend to have a more homogenous organization in that all six layers are roughly the same size. While the somatosensory cortex is organized in the layers of similar cells described above, one should also note that the areas involved in similar functions are organized vertically in columns.[1][2][3]
Functional Neuroanatomy
The primary somatosensory cortex receives the peripheral sensory information but requires the secondary somatosensory cortex to store, process, and retain it (see image. Primary Somatosensory Cortex). The S2 region has links to the hippocampus and amygdala; this helps to receive information from our environments and make decisions on how to approach them by integrating our prior stored experience, factoring our current appraisal of the stimulus, and finally forming a reaction using other areas of the brain based on the former two aspects to aid higher-order processing and problem-solving. The S1 region would be associated with identifying the aspects of touch, such as shape, size, and texture. The S2 region would be associated with spatial and tactile memory associated with sensory experiences.
Sensory Homunculus
The concept of somatotopy refers to the mapping points of the cortex that correspond to its function in the body. In the postcentral gyrus, the sensory homunculus is a common way of representing which portion of the coronal cut of the postcentral gyrus corresponds with the sensation of in the respective part of the body. Often depicted in a coronal section of the postcentral gyrus, the medial portions of the homunculus tend to represent the hips and below along with the genitals while the more lateral sides of the coronal section constitute a larger area for the fingers, face, lips, eyes, and everything else that would require more sensory acuity. The sensory homunculus represents the concept by which we can use sensory deficits in the neurological physical exam to localize lesions in the sensory cortex.[4][5]
Embryology
The central nervous system comes from the neurectoderm, which gives rise to the neural tube and neural crest. The neural tube begins to form anatomical structures, induced by various transcription factors such as sonic the hedgehog, allowing proper migration, orientation, and patterning of cells. The rostral anterior and caudal posterior neuropores close when the neural tube forms. Problems with these neural tube closures result in neural tube defects with elevated levels of alpha-fetoprotein in prenatal testing. Following closure, the primary vesicles form. The primary vesicles include the prosencephalon, mesencephalon, and rhombencephalon. The primary vesicles will then give rise to the secondary vesicles, including the telencephalon, diencephalon, mesencephalon, metencephalon, and myelencephalon. The cerebral hemispheres, including the somatosensory cortex, arise from the telencephalon. The telencephalon is a secondary vesicle that comes from the prosencephalon.[6][7][2]
Blood Supply and Lymphatics
Arterial Blood Supply
The blood supply of the somatosensory cortex arises primarily from the anterior cerebral artery (ACA) and the middle cerebral artery (MCA). Both the anterior cerebral artery and the middle cerebral artery arise from the bifurcation of the internal carotid artery. The anterior cerebral artery supplies most of the medial part of the frontal lobe between the hemispheres. The middle cerebral artery supplies the largest portion of the brain compared to the other cerebral arteries and supplies the lateral sides of the cerebral hemispheres. The anterior cerebral artery supplies the superomedial part of the primary sensory cortex, while the inferolateral part receives supply from the middle cerebral artery - this is because the ACA moves upward between the hemispheres while the MCA courses between the temporal and frontal lobes. An area between the ACA and MCA on either side of the midline is known as the watershed region and tends to experience infarction during global cerebral ischemia. The ACA and the MCA supplying the somatosensory cortex arise from the circle of Willis, which involves both the ACAs, both internal carotid arteries, both posterior cerebral arteries, both posterior communicating arteries, and the anterior communicating artery.[8]
Venous Blood Supply
The venous drainage from the somatosensory cortex enters the venous sinus system contained within the dura. The adjacent arachnoid mater contains granulations that are outlets for cerebrospinal fluid (CSF) from the subarachnoid space. Cerebral veins and anastomosing veins drain deoxygenated blood, waste, and metabolites into the superior sagittal sinus, which drains towards the confluence of sinuses towards the occipital pole. From there, the blood moves down the transverse sinus, sigmoid sinus, then leaves the cranial vault via the internal jugular vein through the jugular foramen. Eventually, the internal jugular carries the blood back to the superior vena cava and back to the right heart via the subclavian and brachiocephalic veins.
Lymphatics
Despite previous suggestions that the brain did not contain the lymphatic flow, a recent paradigm shift used MRI to suggest that the lymphatic vessels exist within the dura mater. The lymphatic circulation serves to drain the subarachnoid space CSF and interstitial fluid within the brain using aquaporins.[9]
Nerves
Receptors Sending Information to the Somatosensory Cortex
The somatosensory nervous system begins with the input from peripheral receptors that eventually translate into meaningful representations in the brain. At the receptor level, various stimuli cause the sensory receptors to release receptor potentials that meet a sufficient threshold to travel to the central nervous system as action potentials. At the level of the specialized sensory receptor, the amplitude of the stimulus recognized by a receptor must reach a threshold before being communicated to the central nervous system (CNS) via interneurons and spinal tracts. The increase in amplitude is due to sodium channels that open in response to the specific stimulus. These specialized receptors communicate specific modalities of sensation such as touch, pain, vibration, temperature, and proprioception. When communicated beyond the level of the receptor, the action potential carries the signal via specific bundles of nerves carrying the same modality to the spinal cord within interneurons. When a sensory receptor receives a strong impulse, this translates to a higher amplitude at the level of the receptor potential then subsequently a higher frequency of action potentials via the nerves receiving information from these receptors given that threshold was attained. Another principle of sensory neuroscience to consider include the phenomena of adaptation. Different receptor types will experience different degrees of adaptation and extinction. Quickly-adapting receptors (phasic) tend to adjust more quickly and send signals more frequently with changes in intensity, such as during onset and offset of the stimulus. Phasic receptors are more important in vestibular senses, which tend to respond to changes in our body position for balance. In contrast, slowly-adapting (tonic) receptors tend to experience slower adaptation that allows continued signaling of an active stimulus of unwavering intensity. An important example of this would be the sensation of proprioception, which requires the body to maintain information about its current position in space.[10][11][12]
Pathway to the Somatosensory Cortex
The sensation throughout the surface of the skin is arranged in discrete segments that correlate to levels of the spinal cord. These segments are referred to as dermatomes. There is often an overlap of sensation between dermatomes, but certain points on the body may not exhibit these overlaps. By identifying the points specific to dermatomes, physicians can use these points in a neurological physical exam to identify the location of a spinal cord lesion or nerve root compression. The first-order neuron cell body is in the dorsal root ganglion.
Each pathway to the somatosensory cortex follows a three-ordered neuronal series from the sensory receptor in the periphery to the primary somatosensory cortex of the brain.
The dorsal root ganglion fibers enter the dorsal column of the spinal cord via the dorsal nerve roots and continue upward within the spinal cord white matter of the dorsal column. The fasciculus cuneatus is more lateral and carries information from T6 and above, and the fasciculus gracilis carries information from below T6. Together they travel up the dorsal tract and reach the nucleus cuneatus and gracilis, respectively. At the level of the lower medulla, they cross the midline and continue along the medial lemniscus to the VPL of the thalamus, which is the third-order neuron in the pathway. From the thalamus, the fibers move through the internal capsule to reach the primary somatosensory cortex. This tract is thus called the dorsal column medial lemniscus pathway (DCML) and carries information to the primary sensory cortex from the contralateral side of the body. The A-beta nerve fibers tend to carry the modalities of touch and pressure to the central nervous system and are myelinated fibers.
The sensory ascending tract would be the anterolateral spinothalamic tract (STT). This tract originates from receptors within the same dermatomal distribution, but the secondary neuron differs. In the spinothalamic tract, which carries information about pain, temperature, and crude touch, the secondary neuron is the dorsal horn of the spinal cord. The fibers from this neuron cross the anterior white commissure of the spinal cord and proceed anterolaterally to the VPL of the thalamus, finally terminating in the primary somatosensory cortex as well. Pain impulses t in specific fibers. These include A-delta pain fibers and C fibers. A-delta pain fibers tend to carry fast pain as they are myelinated, while C fibers tend to carry slow pain and are unmyelinated.[13][14]
Muscles
The receptors of proprioception exist within the skeletal muscles and tendons and assist in understanding the locations of the body in space. Proprioception helps to assist our balance, movement, and coordination and is particularly important in the context of maintaining balance in the absence of visual cues. Without proprioception, one may find difficulty walking in dimly-lit or dark environments, as they have trouble understanding where their limbs are when walking. The sensory receptors involved in proprioception include the muscle spindle and the Golgi tendon organ. The muscle spindle is in parallel with muscle fibers and contains a fusiform capsule in the center of the muscle and is innervated by gamma (A-gamma) motor neurons, which help communicate changes in the length of the muscle fibers during movement and in different positions. This information is communicated by type Ia and type II sensory fibers. The Golgi tendon organ is innervated by type Ib afferent neurons and is found in the tendons, continuously signaling muscle tension status. Together these components help create a sense of the human body in space (proprioception) and participate in reflex arcs.[15][2][16]
Physiologic Variants
The somatotopic arrangement of the sensory cortex and much of the other parts of the brain are subject to the concept of plasticity. The brain adapts to the sensory needs of the body and changes in shape based on usage. Thus the cortical area that corresponds to the sensation of different parts of the body depends on the amount of sensory input that area of the body receives relative to other areas. Even though the amount of cortical space dedicated to sensation can change, the overall distribution of the homunculus is conserved.
Surgical Considerations
There are concerns associated with neurosurgery that must merit careful consideration to determine whether the risks of performing invasive surgery is beneficial to a patient's prognosis. In the worst cases, a neurological decline can follow the postoperative period of neurosurgery. Common complications associated with surgery include:
- Intracranial bleeds
- Brain edema
- CSF leakage
- Infection
- Ischemia
- Hyperperfusion syndrome
- Embolism and stroke
- Cranial nerve palsy, particularly after the endarterectomy of the carotid
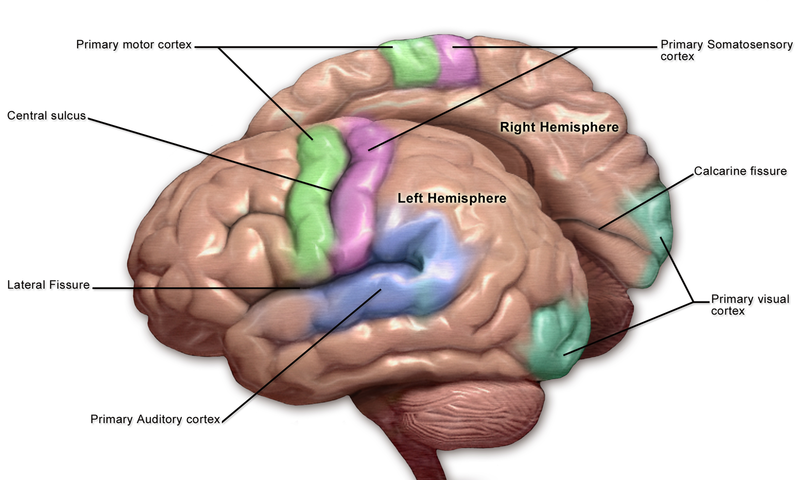
The sensory cortex can be affected by many of the aspects contributing to potential neurological decline after surgery. Any obstruction of the anterior cerebral artery (ACA) or middle cerebral artery (MCA) will cause associated sensory deficits along with other neurological deficits, as this arterial system supplies more than simply the sensory cortex. The surgery on the vasculature itself, including the circle of Willis, can cause embolization. Thromboemboli and atheroemboli (can arise after carotid endarterectomy) from surgical procedures can reach the ACA or MCA and result in the associated neurological symptoms. The obstruction of the ACA typically can cause sensory and motor deficits in the legs, while common manifestations of the MCA obstruction include sensory and motor deficits in the face and arms. See Image. Motor and Sensory Regions of the Cerebral Cortex. Both of these obstructions typically present unilaterally. Essentially, surgically manipulating the vasculature supplying the sensory cortex and the vessels that upstream can result in embolic infarcts. Further surgical considerations include hypotension from blood loss and internal carotid obstruction, resulting in global cerebral ischemia and watershed infarcts in the area between the ACA and MCA.[17][18]
Clinical Significance
The neurological physical exam evaluates light touch, pinprick, vibration using tuning forks, and proprioceptive senses in the extremities. This examination helps the physician to begin to create a differential diagnosis for any problems in the sensation while eventually narrowing down the site of the neurological lesion. The Romberg sign is a useful physical exam finding that can delineate ataxia due to cerebellar problems versus proprioceptive problems. Proprioception is important for maintaining balance; however, it may not present as poor balance when compensated by visual stimuli. Thus a positive Romberg sign indicating sensory proprioceptive deficit is elicited by asking the patient to stand erect with their feet together, assessing for imbalance or sway. The patient should then close their eyes for a full minute, and the examiner should evaluate for imbalance or sway. When the patient shows sway only when with their eyes closed, this is a positive sign. The initial assessment offered by a neurological physical exam gives a standardized way to appraise the sensory, neurological complaints before proceeding further.
Clinical signs and symptoms related to the sensory cortex often present with strokes. The incidence of stroke is higher in western societies due to an increasingly sedentary lifestyle and atherogenic diets. Based on the distribution of symptoms described earlier sensory deficits in various parts of the body can help to determine sites of potential obstruction due to loss of blood supply to their respective parts of the somatosensory cortex. For example, the ACA supplies the superomedial part of the somatosensory cortex. Considering that this area receives sensory input from the lower extremities, loss of sensation in the lower extremities can help localize the source of the stoke to the ACA. The motor supply of the precentral gyrus is supplied by the ACA and MCA as well and follows a similar distribution, causing an additional motor weakness in the corresponding areas as well. Since the mechanosensory modality carried in the dorsal column medial lemniscus pathway crosses the midline, the lesions produce a contralateral loss of sensation.
Watershed infarcts mentioned earlier tend to present with the FAST mnemonic, which stands for facial weakness, arm weakness, speech difficulty, and time to act. When a stroke does not receive a timely intervention, symptoms can lead to numbness and hyperreflexia in the affected area. The watershed strokes affect more than an ACA or MCA stroke and can present with additional deficits of bilateral vision loss or even proximal limb weakness that does not affect the face, hands, or feet. Watershed infarcts can be caused by congestive heart failure, atherosclerosis, thrombi, or carotid artery stenosis, to name a few.
Direct lesions of the parietal lobe can result in a wide variety of somatosensory defects as well. Integrative sensorimotor functions can be affected and lead to presentations based on the severity of the lesion. An example of a loss of integrative sensorimotor function from parietal defects involving the somatosensory cortex is tactile apraxia. Tactile apraxia is a defect in active touch feedback when manipulating objects with our hands that integrate the sensory aspect of digital palpation and spatial features of objects with the brain's motor aspect to manipulate them. It is important to note that movements of the hand without the use of an object, are intact. Motor function without tactile feedback is intact. Lesions that affect the primary somatosensory cortex can result in a loss in the ability to discriminate objects using the touch modality only, such as shape, size, or texture. Lesions that affect the secondary somatosensory cortex affect the recognition of objects based on tactile appraisal over sensory discrimination. An individual with such a lesion will have trouble recognizing an object based on touch, known as tactile apraxia unilaterally and astereognosis bilaterally. It is important to understand that the somatosensory nervous system interconnects with other areas of the brain that require intact sensory input to interact with surrounding stimuli.[18][19][3][20][21]