Continuing Education Activity
Pulsus alternans is an arterial pulse with alternating strong and weak beats. It is found in the setting of severe ventricular dysfunction and other forms of cardiac pathology. The treatment and clinical outcomes for pulsus alternans vary depending upon the underlying causative etiology, though its presence generally suggests a poor prognostic outlook. This activity reviews the evaluation and treatment of pulsus alternans and highlights the role of the interprofessional team in evaluating and treating patients with this condition.
Objectives:
- Describe the pathophysiology of pulsus alternans.
- Review the common findings associated with pulsus alternans.
- Outline the treatment considerations for patients with pulsus alternans.
- Explain the importance of collaboration and communication among the interprofessional team to enhance the delivery of care for patients with pulsus alternans.
Introduction
Pulsus alternans (PA), not to be confused with pulsus paradoxus or electrical alternans, is an arterial pulse with alternating strong and weak beats. It is a hemodynamic phenomenon marked by a beat-to-beat alternation in the amplitude of the pulse resulting in a clinically identifiable pulse half of the electrical rate. In 1872, Ludwig Traube first described this phenomenon.[1] Systolic pressures will vary from beat to beat as identified on physical exam and echocardiogram. Pulsus alternans is found in the setting of severe ventricular dysfunction and other forms of cardiac pathology. Variations of pulsus alternans include left ventricular alternans, right ventricular alternans, and biventricular alternans. The treatment and clinical outcomes for pulsus alternans vary depending upon the underlying causative etiology, though its presence generally suggests a poor prognostic outlook.[2]
Pulsus alternans usually occurs in association with severe heart disease and can eventually lead to pulseless electrical activity.[3][4][5]
Etiology
Pulsus alternans occurs as a result of underlying cardiac distress and dysfunction. Pulsus alternans has been identified as left ventricular, right ventricular, or biventricular, and the etiology of pulsus alternans varies depending on the causative pathology. Left ventricular alternans occurs in the setting of severe left ventricular dysfunction. This includes cardiomyopathy, aortic stenosis, and coronary artery disease. Right ventricular alternans occurs as a result of right ventricular strain, often precipitated by a pulmonary embolus or pulmonary hypertension. Other potential etiologies of right ventricular alternans include reactive airway disease, mitral stenosis, or left-sided heart failure.[2] Biventricular alternans is very rare, only being documented in a small number of case reports. Case reports have shown biventricular alternans to be caused by severe left ventricular dysfunction and left anterior descending coronary artery disease.[4]
Another possible etiology for PA is pericardial effusion. Different mechanisms may provoke PA, such as hemodynamic and inotropic impairment.[6][7][8]
Epidemiology
The patient population in which pulsus alternans occurs is reflected by the underlying pathophysiology. While pulsus alternans is a relatively rare documented occurrence, it is most commonly associated with left ventricular dysfunction.[2] Therefore, patients with heart failure, cardiomyopathy, or coronary artery disease are at the greatest risk for developing pulsus alternans. Cardiac risk factors may include a previous history of coronary artery disease, congestive heart failure, hypertension, hyperlipidemia, diabetes, peripheral vascular disease, prior stroke, or tobacco use. At the time of this publication, larger epidemiological studies have not been performed.
Pathophysiology
The exact mechanism of pulsus alternans has never been clearly determined. However, two different mechanisms have been theorized to explain the pathophysiology. The first involves the Frank-Starling curve, where the alternating contractile force is a result of the variation of the end-diastolic volume.[9] The poorly contracting left ventricle has a reduced stroke volume, and this increases the end-diastolic volume for the next contraction.[7] The second mechanism involves diseased cardiac myocytes with abnormal calcium cycling, possibly due to the over-expression of calsequestrin.[2] A previous study demonstrated alterations of the calcium amplitude in conjunction with the force of contraction in right ventricular papillary muscles, thereby strengthening this theory.[10]
History and Physical
A thorough cardiovascular exam should be completed for all patients identified as having pulsus alternans. Given the common underlying etiologies, patients with pulsus alternans will often present with overt signs of decompensated heart failure. This includes elevated jugular venous pressure, bibasilar rales, and peripheral edema. Depending upon the degree of cardiac decompensation, the patient may also be tachycardic, tachypneic, or hypotensive. The presence of an aortic stenosis murmur, S3, or S4, is another exam finding for which one should look. A more specific physical exam finding classically associated with pulsus alternans would be alternating strong and soft pulses of the radial artery.[11]
Pulsus alternans can be identified by palpating the artery, using a sphygmomanometer, and examining arterial and plethysmographic waveforms.[12] Pulsus alternans becomes difficult to assess by the palpatory method when there is a greater than 20 mmHg difference between large and small systolic beats. This difference is best detected by the palpation of the femoral pulses instead of the brachial, radial, or carotid pulses. In order to detect with a sphygmomanometer, the cuff is first inflated higher than peak systolic pressure. It is then gradually deflated while the operator focuses on listening to the first audible systolic beat (first pressure). At this point, only strong pulses are audible. The cuff is then slowly deflated until all systolic beats are audible (second pressure). The severity of pulsus alternans can be assessed by subtracting the first pressure from the second pressure.
Evaluation
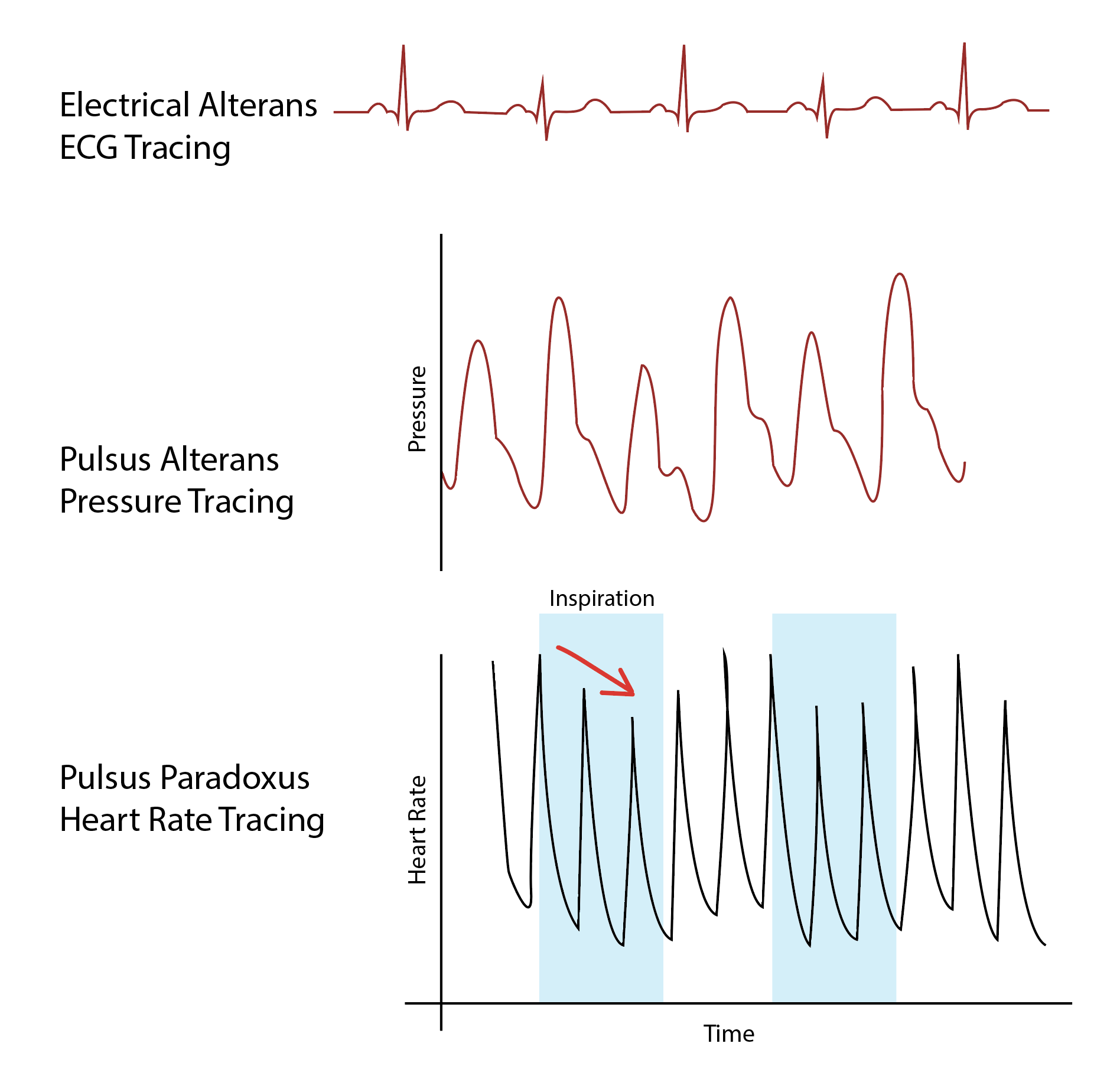
Whenever pulsus alternans is suspected, the appropriate cardiac workup should be completed, including laboratory work, chest X-ray, electrocardiogram, and echocardiogram. Objective laboratory findings include elevated pro-BNP and pulmonary vascular congestion on chest X-ray. Electrocardiogram will often show normal sinus rhythm or sinus tachycardia.[2][13][14] Notably, there will typically be no electrical alternans on the electrocardiogram, as this is found with electrical alternans and not pulsus alternans.[15] An echocardiogram will show alternating beat-to-beat arterial waveforms, the pathognomonic finding for pulsus alternans.[4]
Timing of events and the series of occurrences are important details one should pay attention to. Rarely, there can be a discrepancy between the electrical and mechanical activities of the heart, and echocardiography is an important tool to identify such inconsistency. Furthermore, there may be a discrepancy between the efficiency of the heart and the mechanical activation of the heart, the production of cardiac output, and in such cases, echocardiography can play a crucial role in diagnosing such a condition. Combining information from 2D and tissue Doppler evaluation of contractility, ECG tracing, and spectral tracing across the left ventricular outflow tract can provide an assessment of the mechanical activation of the heart, electrical activation of the heart muscle, and the resulting “success” of these efforts in producing cardiac output.
Treatment / Management
As pulsus alternans is a sign of underlying cardiac dysfunction or disease, treatment involves the management of the underlying pathology. Treatment modalities for left ventricular alternans may include diuresis (for left-sided heart failure) or replacement of the aortic valve (for severe aortic stenosis).[16] Both left ventricular alternans, and biventricular alternans have been associated with coronary artery disease, particularly of the left anterior descending coronary artery. Ischemic evaluation should be considered in this patient population. Treatment modalities for right ventricular alternans may include anticoagulation (for pulmonary embolism), pulmonary antihypertensives (for primary pulmonary hypertension), continuous positive airway pressure (CPAP), or bilevel positive airway pressure (BiPAP) used for sleep apnea or obesity hypoventilation syndrome.
Differential Diagnosis
The differential diagnosis for pulsus alternans is varied depending upon the potential underlying etiology. Pulsus alternans of the left ventricle should raise concern for reduced ejection fraction, cardiomyopathy, aortic stenosis, coronary artery disease, or cardiac ischemia. Pulsus alternans of the right ventricle should raise concern for pulmonary embolus, pulmonary hypertension, reactive airway disease, or mitral stenosis.[4] Biventricular alternans is rare, though it should raise concern for possible ischemia of the left anterior descending coronary artery.[2] Of importance, pulsus alternans should be distinguished from electrical alternans, an electrocardiogram finding demonstrating alternating levels of the amplitude of the QRS complex. Electrical alternans is a sign commonly found in the setting of pericardial effusion.[15]
Prognosis
The prognosis for a patient with pulsus alternans will vary dependent upon the underlying etiology. However, the presence of pulsus alternans is generally considered a poor prognostic sign, as pulsus alternans is a feature of severe ventricular dysfunction.[17] Correction of the causative left ventricular or right ventricular pathology will often resolve the findings of pulsus alternans on physical exam and echocardiogram. For example, a previous study demonstrated the resolution of pulsus alternans after aortic valve replacement in the setting of severe aortic stenosis.[16] Patients with irreversible cardiomyopathies are at increased risk for worse outcomes due to the inability to correct the underlying pathology.
Complications
Potential complications in the setting of pulsus alternans are varied. Of chief concern are life-threatening cardiac complications, including myocardial infarction and ventricular arrhythmias. While the risk for developing ventricular arrhythmias in the setting of pulsus alternans is unknown, there is theoretically increased risk due to potential changes in cardiac conduction.[7] Other studies have shown an experimental association of pulsus alternans with cardiac arrhythmias and myocardial ischemia.[18] Atrial fibrillation and atrial flutter are other common arrhythmias that may occur in settings of cardiac distress and decompensation. Severe decompensation in the setting of congestive heart failure may also lead to hemodynamic instability, respiratory distress, hypoxia, hepatic congestion, and acute kidney injury.
Deterrence and Patient Education
Pulsus alternans indicate cardiac pathology, particularly left ventricular dysfunction, and is considered a poor prognostic indicator. When identified, pulsus alternans should elicit prompt treatment of the underlying causative process.
Pearls and Other Issues
Pulsus alternans is not to be confused with electrical alternans or pulsus paradoxus. Pulsus alternans is beat-to-beat variability of the arterial pressure waveform as identified on physical exam and echocardiogram. Electrical alternans is beat-to-beat variability of the QRS complex on electrocardiogram, often found in the setting of pericardial effusion.[15] Pulsus paradoxus is a drop of systolic blood pressure greater than 10 mmHg with inspiration and is associated with cardiac tamponade.[19] Although the nomenclature is similar, it is important to clarify that both electrical alternans and pulsus paradoxus are not related to pulsus alternans and have different underlying pathophysiology.
Enhancing Healthcare Team Outcomes
Management of pulsus alternans secondary to underlying cardiac pathology will require an interprofessional approach. Prompt evaluation with cardiology should be an important component of any initial assessment for pulsus alternans. If the patient has associated respiratory distress, coordination with respiratory therapy will be necessary for the treatment plan. Nursing staff will need to closely monitor the patient’s vitals and perform strict monitoring of the patient’s intake and output. If ischemic heart disease or severe valvulopathy is suspected, consultation with interventional cardiology would also be warranted. If a large pulmonary embolism is identified, coordination with interventional radiology for catheter-directed anticoagulation may be considered. Depending on the degree of cardiac dysfunction, a surveillance echocardiogram may be a consideration. Given the cardiac dysfunction associated with pulsus alternans, a multidisciplinary approach is of great importance to monitor and treat patients with complex cardiac disease.