Introduction
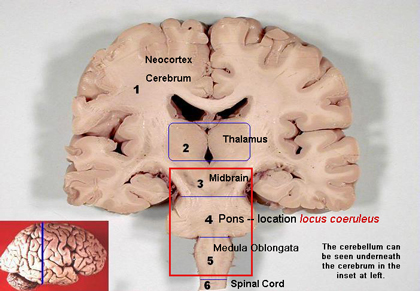
The locus coeruleus (LC) is one component of the reticular activating system (RAS). See Image. Locus Coeruleus. The RAS is composed of the reticular formation, raphe nuclei, and locus coeruleus. It is involved in many important functions; normally, the RAS mediates consciousness, wakefulness, and attentiveness. The loss of muscle tone during REM sleep is influenced by the caudal third of the locus coeruleus, although the locus coeruleus appears to be mostly inactive during REM sleep in some studies. The locus coeruleus primarily functions through the transmission of norepinephrine via a g protein-coupled receptor pathway. Arousal, sleep-wake cycle, memory, emotions, and stress are all influenced by the locus coeruleus.[1][2][3]
Structure and Function
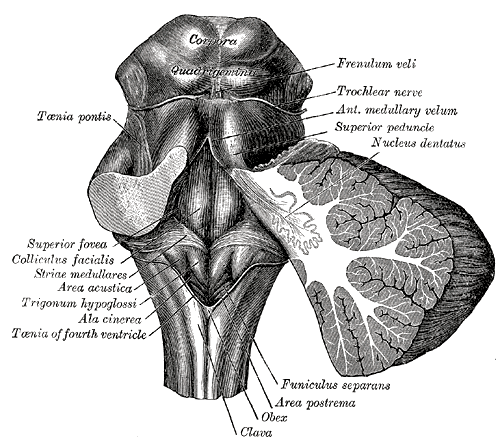
The locus coeruleus is a nucleus in the lateral floor of the fourth ventricle and upper dorsolateral pons. Norepinephrine, also called noradrenaline, is a neurotransmitter predominantly released from the locus coeruleus. Norepinephrine helps promote the formation of memories and strengthens memories, especially those created in stressful situations. Noradrenaline released from the locus coeruleus can act on adrenergic receptors (alpha and beta receptors) present on neurons and glial cells (oligodendrocytes, astrocytes, ependymal cells, Schwann cells, microglia, and satellite cells) via adenylyl cyclase and phospholipase C pathways. Adenylyl cyclase converts ATP to cAMP; cAMP then phosphorylates protein kinase A (PKA), which in turn has many functions within the central nervous system. Phospholipase C (PLC) is an enzyme that cleaves phospholipids resulting in the formation of diacylglycerol (DAG) and inositol 1,4,5-trisphosphate (IP3) that contribute to signal transduction pathways. Noradrenaline can also activate glycogen metabolism and calcium signaling in astrocytes as well as regulate blood flow and blood-brain barrier (BBB) permeability.[4]
Embryology
Studies on the embryologic development of the locus coeruleus within humans are limited. As the neural tube forms during embryonic development, it divides into segments called rhombomeres in the region of the hindbrain (see Image. The Hindbrain or Rhombencephalon). In studies performed on mice, researchers discovered that the locus coeruleus to originate from the dorsal alar rhombomere 1 (R1). The dorsal subcoeruleus (SubC) also predominantly arises from dorsal alar R1 (Oliveira et al., 2017). Brain development requires regulation via homeobox genes. A homeobox contains a DNA sequence that regulates embryological development and is involved in the segmental organization of the embryo. Engrailed (En) 1 is a homeobox gene that, when expressed, allows the migration of locus coeruleus and SubC to move from ventrocaudal to the basolateral position, placing the structures close to the fourth ventricle.
Blood Supply and Lymphatics
Some studies suggest that activation of the locus coeruleus may lead to increased cerebral blood flow to certain areas of the brain. Norepinephrine (NE) released from the locus coeruleus helps maintain the blood-brain barrier; it leads to vasoconstriction of vessels and may have a role in redistributing blood flow to more active areas of the brain. The brain receives blood supply from arteries that branch from the circle of Willis. The circle of Willis is comprised of anastomoses of arteries located at the base of the brain. The internal carotid artery and basilar artery are two vessels that are part of the circle of Willis. Branches of the internal carotid artery (anterior, middle, and posterior cerebral arteries) supply most of the brain except for the cerebellum, brainstem, and medial occipital lobe, which are supplied by the basilar artery. There has been a long-held belief that the brain does not have lymphatic vasculature. Recently research has determined that the central nervous system undergoes immune regulation that may take place within the meninges. The discovery of lymphatic vessels or structures within the brain is limited; therefore, further research is necessary.
Nerves
The neurons of the locus coeruleus are mostly branched and not myelinated. Some of the areas that provide input to the LC are the medial prefrontal cortex, hypothalamus, paragigantocellularis, and nucleus prepositus. The locus coeruleus provides output to the hippocampus, spinal cord, hypothalamus, and amygdala. The noradrenergic neurons that innervate areas of the brain that help maintain functions such as memory, attention, and the sleep-wake cycle.[5][6][7]
Muscles
Rapid eye movement sleep (REM sleep) occurs in about 90-minute intervals and is predominantly a parasympathetic state. REM sleep is also known as paradoxical sleep because beta waves appear on an electroencephalogram (EEG). Beta waves are also seen on EEG when an individual is awake. Normally during REM, there is a loss of muscle tone (atonia or tonic muscular inhibition). There is a theory that tonic muscular inhibition is under the control of the caudal part of the locus coeruleus. This decreased muscle tone may protect an individual from inadvertently harming themselves by preventing them from "acting out" their dreams. Lesions of the locus coeruleus and subcoeruleus have been theorized to be one possible cause of REM sleep behavior disorder. REM sleep behavior disorder (RBD) occurs due to the loss of tonic muscular inhibition during REM, leading an individual to "act out" dreams. Depending on the area of the lesion, symptoms can range anywhere from simple limb twitches to complex behaviors such as screaming, laughing, punching, kicking, or running.
Physiologic Variants
Abnormalities of the locus coeruleus have implications as contributing to neuropsychiatric and neurodegenerative processes. Individuals suffering from neuropsychiatric processes may be affected due to abnormal norepinephrine regulation leading to the formation of clinical depression, post-traumatic stress disorder (PTSD), panic attacks, and anxiety. When norepinephrine becomes elevated, there is an increase in anxiety and mania. Depression may be apparent when norepinephrine decreases. The degeneration of neurons in the locus coeruleus has been postulated as contributing to Parkinson disease and Alzheimer disease. In studies performed on mice, researchers induced the degeneration of locus coeruleus neurons, which resulted in increased deposits of amyloid-beta protein in the brain. Amyloid-beta protein is a component of amyloid plaques. Amyloid plaques are involved in the pathology of Alzheimer disease.
Surgical Considerations
Benefits and risks always merit consideration when contemplating neurosurgery. The locus coeruleus may be damaged in the process of neurosurgery. The tectum is a region of the brainstem (midbrain) that consists of the inferior and superior colliculi. The superior colliculus functions to process visual information and control eye movement. The inferior colliculus functions to process auditory information. Benign tumors may form in the tectal region of the brain. Neurosurgery for the removal of tumors found in the tectal region is challenging and may contribute to iatrogenic lesions of the locus coeruleus. Damage to the locus coeruleus may result in depression, REM sleep disorder, cognitive difficulties, and more.[8]
Clinical Significance
Locus coeruleus involvement is implicated in processes such as depression, anxiety, Alzheimer disease, Parkinson disease, and even opiate withdrawal. Medications such as norepinephrine reuptake inhibitors (NRI), norepinephrine dopamine reuptake inhibitors (NDRI), serotonin-norepinephrine reuptake inhibitors (SSRI), and alpha-2 agonists act on neurons in the locus coeruleus. Understanding the locus coeruleus can help in finding the best treatment option for many neurological disorders. Bipolar depression is one of many psychiatric illnesses that are difficult to treat. One study observed that when fluoxetine (an SSRI) was administered independently, the LC decreased firing. Administering olanzapine (an atypical antipsychotic) with fluoxetine increased the firing of the locus coeruleus and the levels of norepinephrine release, which helped reduce the overall symptoms of bipolar depression. Increased LC activation has implications in the process of opiate withdrawal. Clonidine is an alpha-2 agonist that acts on receptors within the LC and decreases the activity of noradrenergic neurons. This decrease in activation of the LC can help ameliorate the symptoms of withdrawal. Other neural mechanisms may be involved in opioid withdrawal; however, more research is necessary. There have been many strides towards the advancement in medicine and understanding neural mechanisms. Medicine is a continually evolving field, and further research is required to gain an understanding of LC involvement in neuropsychiatric and neurodegenerative disorders. Understanding the structure and function of the LC helps determine the best treatment of many neuropsychiatric and neurodegenerative diseases that afflict the general population.
Other Issues
The use of MRIs allows non-invasive imaging of the human brain; however, neuroanatomy is quite complex. Many studies to understand pathological processes and the effects of medication are performed using mice or other animals. The mouse brain is similar to that of a human. However, mice may show different physiologic responses in a study. Mice may not experience certain side effects of a medication that a human would and vice versa.