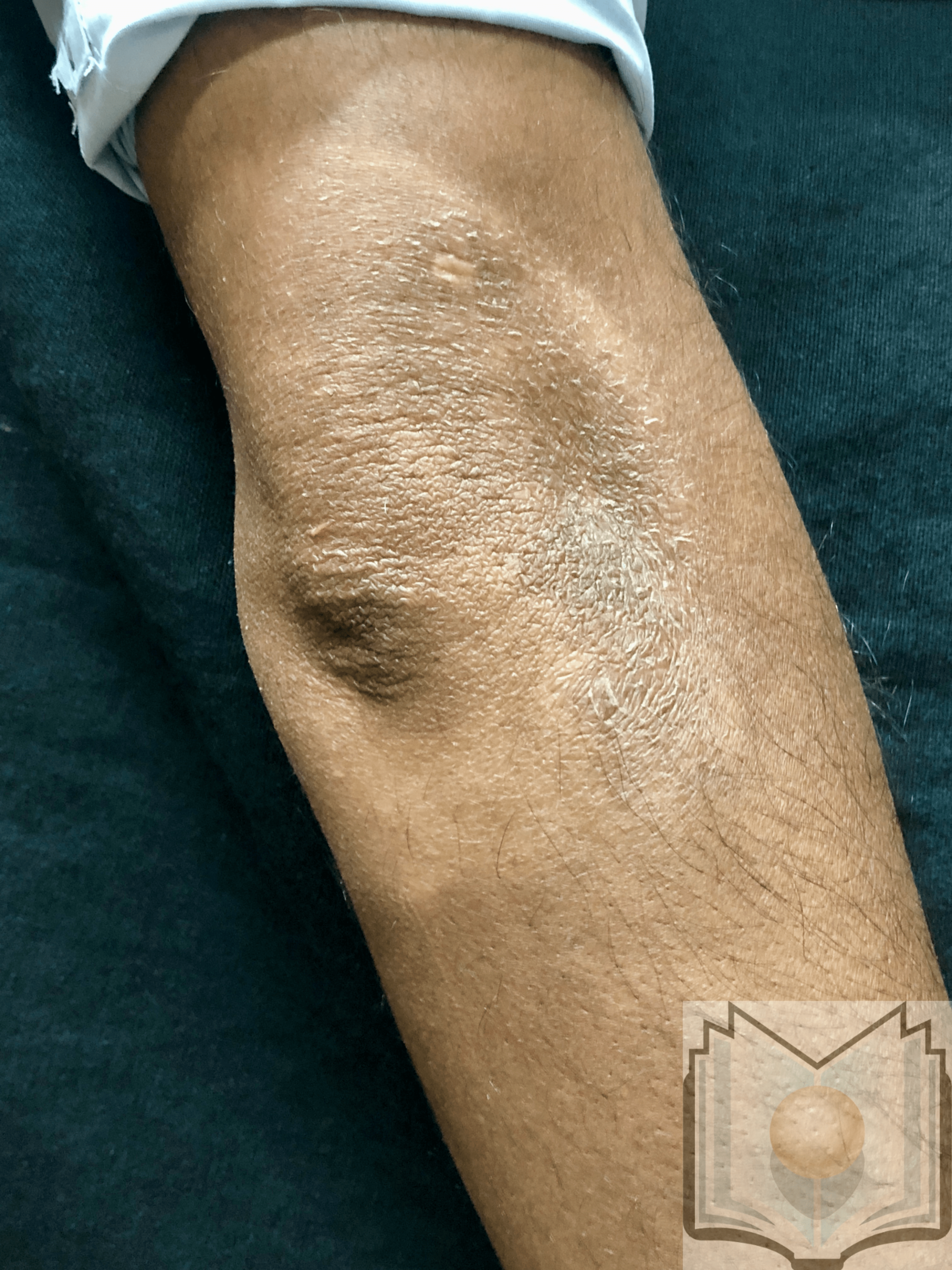
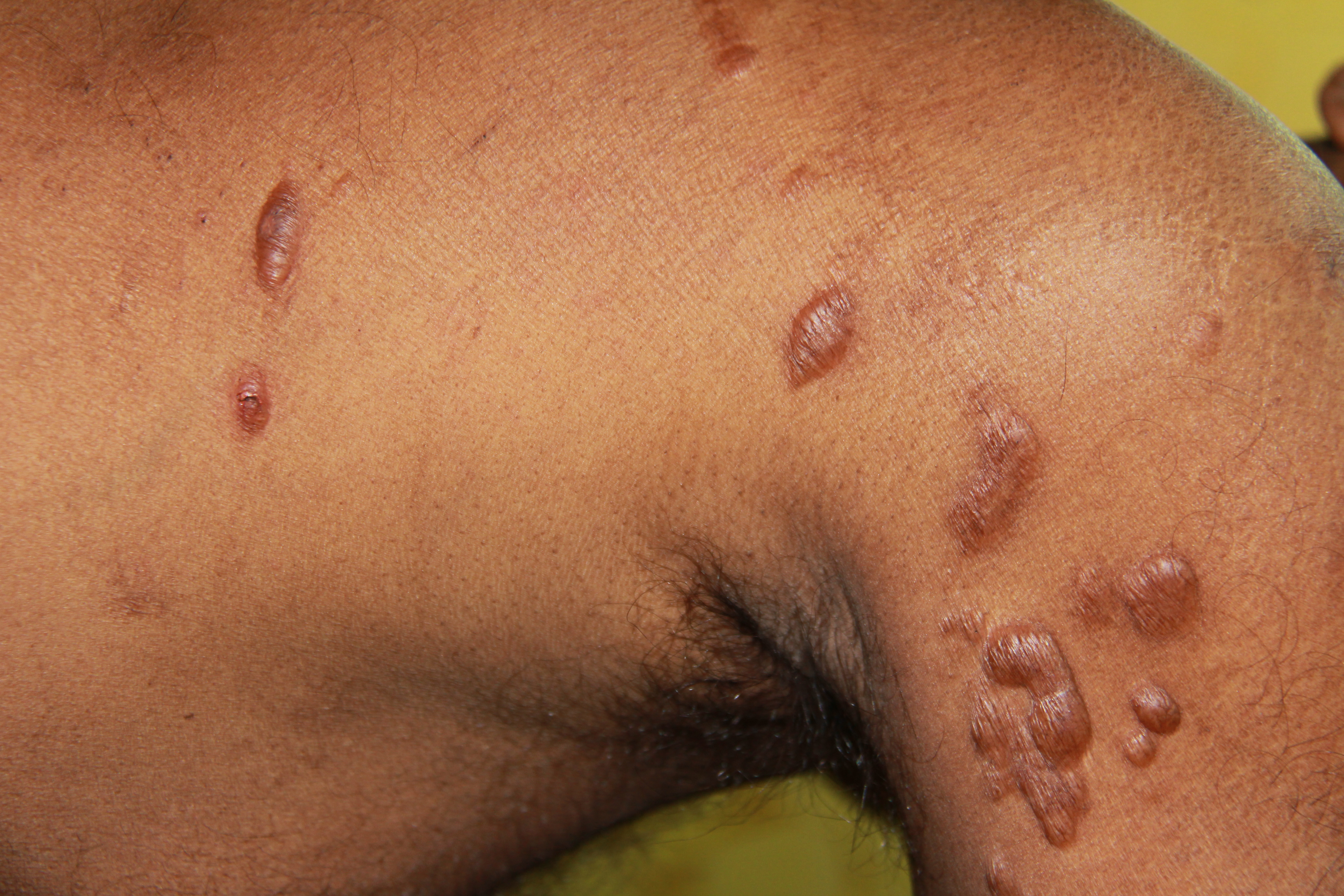
[1]
Singh P, Benjak A, Schuenemann VJ, Herbig A, Avanzi C, Busso P, Nieselt K, Krause J, Vera-Cabrera L, Cole ST. Insight into the evolution and origin of leprosy bacilli from the genome sequence of Mycobacterium lepromatosis. Proceedings of the National Academy of Sciences of the United States of America. 2015 Apr 7:112(14):4459-64. doi: 10.1073/pnas.1421504112. Epub 2015 Mar 23
[PubMed PMID: 25831531]
[2]
Han XY, Sizer KC, Thompson EJ, Kabanja J, Li J, Hu P, Gómez-Valero L, Silva FJ. Comparative sequence analysis of Mycobacterium leprae and the new leprosy-causing Mycobacterium lepromatosis. Journal of bacteriology. 2009 Oct:191(19):6067-74. doi: 10.1128/JB.00762-09. Epub 2009 Jul 24
[PubMed PMID: 19633074]
Level 2 (mid-level) evidence
[3]
Moschella SL. An update on the diagnosis and treatment of leprosy. Journal of the American Academy of Dermatology. 2004 Sep:51(3):417-26
[PubMed PMID: 15337986]
[4]
Scollard DM, Adams LB, Gillis TP, Krahenbuhl JL, Truman RW, Williams DL. The continuing challenges of leprosy. Clinical microbiology reviews. 2006 Apr:19(2):338-81
[PubMed PMID: 16614253]
[7]
Valverde CR, Canfield D, Tarara R, Esteves MI, Gormus BJ. Spontaneous leprosy in a wild-caught cynomolgus macaque. International journal of leprosy and other mycobacterial diseases : official organ of the International Leprosy Association. 1998 Jun:66(2):140-8
[PubMed PMID: 9728446]
[8]
Cole ST, Eiglmeier K, Parkhill J, James KD, Thomson NR, Wheeler PR, Honoré N, Garnier T, Churcher C, Harris D, Mungall K, Basham D, Brown D, Chillingworth T, Connor R, Davies RM, Devlin K, Duthoy S, Feltwell T, Fraser A, Hamlin N, Holroyd S, Hornsby T, Jagels K, Lacroix C, Maclean J, Moule S, Murphy L, Oliver K, Quail MA, Rajandream MA, Rutherford KM, Rutter S, Seeger K, Simon S, Simmonds M, Skelton J, Squares R, Squares S, Stevens K, Taylor K, Whitehead S, Woodward JR, Barrell BG. Massive gene decay in the leprosy bacillus. Nature. 2001 Feb 22:409(6823):1007-11
[PubMed PMID: 11234002]
[9]
Eiglmeier K, Parkhill J, Honoré N, Garnier T, Tekaia F, Telenti A, Klatser P, James KD, Thomson NR, Wheeler PR, Churcher C, Harris D, Mungall K, Barrell BG, Cole ST. The decaying genome of Mycobacterium leprae. Leprosy review. 2001 Dec:72(4):387-98
[PubMed PMID: 11826475]
[11]
Truman RW, Singh P, Sharma R, Busso P, Rougemont J, Paniz-Mondolfi A, Kapopoulou A, Brisse S, Scollard DM, Gillis TP, Cole ST. Probable zoonotic leprosy in the southern United States. The New England journal of medicine. 2011 Apr 28:364(17):1626-33. doi: 10.1056/NEJMoa1010536. Epub
[PubMed PMID: 21524213]
[12]
Domozych R, Kim E, Hart S, Greenwald J. Increasing incidence of leprosy and transmission from armadillos in Central Florida: A case series. JAAD case reports. 2016 May:2(3):189-92. doi: 10.1016/j.jdcr.2016.03.004. Epub 2016 May 12
[PubMed PMID: 27222885]
Level 2 (mid-level) evidence
[13]
Sharma R, Singh P, Loughry WJ, Lockhart JM, Inman WB, Duthie MS, Pena MT, Marcos LA, Scollard DM, Cole ST, Truman RW. Zoonotic Leprosy in the Southeastern United States. Emerging infectious diseases. 2015 Dec:21(12):2127-34. doi: 10.3201/eid2112.150501. Epub
[PubMed PMID: 26583204]
[14]
Abraham S, Mozhi NM, Joseph GA, Kurian N, Rao PS, Job CK. Epidemiological significance of first skin lesion in leprosy. International journal of leprosy and other mycobacterial diseases : official organ of the International Leprosy Association. 1998 Jun:66(2):131-9
[PubMed PMID: 9728445]
Level 2 (mid-level) evidence
[15]
Araujo S, Freitas LO, Goulart LR, Goulart IM. Molecular Evidence for the Aerial Route of Infection of Mycobacterium leprae and the Role of Asymptomatic Carriers in the Persistence of Leprosy. Clinical infectious diseases : an official publication of the Infectious Diseases Society of America. 2016 Dec 1:63(11):1412-1420
[PubMed PMID: 27558564]
[17]
Lahiri R, Krahenbuhl JL. The role of free-living pathogenic amoeba in the transmission of leprosy: a proof of principle. Leprosy review. 2008 Dec:79(4):401-9
[PubMed PMID: 19274986]
[18]
Paling S, Wahyuni R, Ni'matuzahroh, Winarni D, Iswahyudi, Astari L, Adriaty D, Agusni I, Izumi S. ACANTHAMOEBA SP.S-11 PHAGOCYTOTIC ACTIVITY ON MYCOBACTERIUM LEPRAE IN DIFFERENT NUTRIENT CONDITIONS. African journal of infectious diseases. 2018:12(1 Suppl):44-48. doi: 10.2101/Ajid.12v1S.5. Epub 2018 Mar 7
[PubMed PMID: 29619429]
[19]
Wheat WH, Casali AL, Thomas V, Spencer JS, Lahiri R, Williams DL, McDonnell GE, Gonzalez-Juarrero M, Brennan PJ, Jackson M. Long-term survival and virulence of Mycobacterium leprae in amoebal cysts. PLoS neglected tropical diseases. 2014 Dec:8(12):e3405. doi: 10.1371/journal.pntd.0003405. Epub 2014 Dec 18
[PubMed PMID: 25521850]
[20]
Moss C, Burn J. Genetic counselling in hypomelanosis of Ito: case report and review. Clinical genetics. 1988 Aug:34(2):109-15
[PubMed PMID: 3056640]
Level 3 (low-level) evidence
[21]
van Beers SM, Hatta M, Klatser PR. Patient contact is the major determinant in incident leprosy: implications for future control. International journal of leprosy and other mycobacterial diseases : official organ of the International Leprosy Association. 1999 Jun:67(2):119-28
[PubMed PMID: 10472363]
[22]
Moet FJ, Pahan D, Schuring RP, Oskam L, Richardus JH. Physical distance, genetic relationship, age, and leprosy classification are independent risk factors for leprosy in contacts of patients with leprosy. The Journal of infectious diseases. 2006 Feb 1:193(3):346-53
[PubMed PMID: 16388481]
[23]
Mira MT, Alcaïs A, Nguyen VT, Moraes MO, Di Flumeri C, Vu HT, Mai CP, Nguyen TH, Nguyen NB, Pham XK, Sarno EN, Alter A, Montpetit A, Moraes ME, Moraes JR, Doré C, Gallant CJ, Lepage P, Verner A, Van De Vosse E, Hudson TJ, Abel L, Schurr E. Susceptibility to leprosy is associated with PARK2 and PACRG. Nature. 2004 Feb 12:427(6975):636-40
[PubMed PMID: 14737177]
[24]
Trindade MA, Palermo ML, Pagliari C, Valente N, Naafs B, Massarollo PC, D'Albuquerque LA, Benard G. Leprosy in transplant recipients: report of a case after liver transplantation and review of the literature. Transplant infectious disease : an official journal of the Transplantation Society. 2011 Feb:13(1):63-9. doi: 10.1111/j.1399-3062.2010.00549.x. Epub
[PubMed PMID: 20678090]
Level 3 (low-level) evidence
[25]
Martiniuk F, Rao SD, Rea TH, Glickman MS, Giovinazzo J, Rom WN, Cabrera A, Levis WR. Leprosy as immune reconstitution inflammatory syndrome in HIV-positive persons. Emerging infectious diseases. 2007 Sep:13(9):1438-40. doi: 10.3201/eid1309.070301. Epub
[PubMed PMID: 18252138]
[26]
Scollard DM, Joyce MP, Gillis TP. Development of leprosy and type 1 leprosy reactions after treatment with infliximab: a report of 2 cases. Clinical infectious diseases : an official publication of the Infectious Diseases Society of America. 2006 Jul 15:43(2):e19-22
[PubMed PMID: 16779736]
Level 3 (low-level) evidence
[27]
Massone C, Belachew WA, Schettini A. Histopathology of the lepromatous skin biopsy. Clinics in dermatology. 2015 Jan-Feb:33(1):38-45. doi: 10.1016/j.clindermatol.2014.10.003. Epub
[PubMed PMID: 25432809]
[28]
Ridley DS, Jopling WH. Classification of leprosy according to immunity. A five-group system. International journal of leprosy and other mycobacterial diseases : official organ of the International Leprosy Association. 1966 Jul-Sep:34(3):255-73
[PubMed PMID: 5950347]
[29]
Pardillo FE, Fajardo TT, Abalos RM, Scollard D, Gelber RH. Methods for the classification of leprosy for treatment purposes. Clinical infectious diseases : an official publication of the Infectious Diseases Society of America. 2007 Apr 15:44(8):1096-9
[PubMed PMID: 17366457]
[30]
Elinav H, Palladas L, Applbaum YH, Gilead L, Moses AE, Cohen-Poradosu R. Plantar ulcers and eyebrow-hair paucity. Clinical infectious diseases : an official publication of the Infectious Diseases Society of America. 2006 Mar 1:42(5):684-5, 722-4
[PubMed PMID: 16450473]
[31]
Lane JE, Balagon MV, Dela Cruz EC, Abalos RM, Tan EV, Cellona RV, Sadaya PG, Walsh GP, Walsh DS. Mycobacterium leprae in untreated lepromatous leprosy: more than skin deep. Clinical and experimental dermatology. 2006 May:31(3):469-70
[PubMed PMID: 16681612]
[32]
Kumar B, Rai R, Kaur I. Systemic involvement in leprosy and its significance. Indian journal of leprosy. 2000 Jan-Mar:72(1):123-42
[PubMed PMID: 10935191]
[33]
Fleury RN, Duerksen F. Emergency in leprosy: involvement of the larynx. Leprosy review. 2007 Jun:78(2):148-50
[PubMed PMID: 17824485]
[34]
Saunderson P, Bizuneh E, Leekassa R. Neuropathic pain in people treated for multibacillary leprosy more than ten years previously. Leprosy review. 2008 Sep:79(3):270-6
[PubMed PMID: 19009976]
[35]
Lasry-Levy E, Hietaharju A, Pai V, Ganapati R, Rice AS, Haanpää M, Lockwood DN. Neuropathic pain and psychological morbidity in patients with treated leprosy: a cross-sectional prevalence study in Mumbai. PLoS neglected tropical diseases. 2011 Mar 8:5(3):e981. doi: 10.1371/journal.pntd.0000981. Epub 2011 Mar 8
[PubMed PMID: 21408111]
Level 2 (mid-level) evidence
[36]
Wilder-Smith EP, Van Brakel WH. Nerve damage in leprosy and its management. Nature clinical practice. Neurology. 2008 Dec:4(12):656-63. doi: 10.1038/ncpneuro0941. Epub 2008 Nov 11
[PubMed PMID: 19002133]
[37]
Meima A, Saunderson PR, Gebre S, Desta K, Habbema JD. Dynamics of impairment during and after treatment: the AMFES cohort. Leprosy review. 2001 Jun:72(2):158-70
[PubMed PMID: 11495447]
[38]
Richardus JH, Nicholls PG, Croft RP, Withington SG, Smith WC. Incidence of acute nerve function impairment and reactions in leprosy: a prospective cohort analysis after 5 years of follow-up. International journal of epidemiology. 2004 Apr:33(2):337-43
[PubMed PMID: 15082636]
[39]
Saunderson P, Gebre S, Desta K, Byass P, Lockwood DN. The pattern of leprosy-related neuropathy in the AMFES patients in Ethiopia: definitions, incidence, risk factors and outcome. Leprosy review. 2000 Sep:71(3):285-308
[PubMed PMID: 11105488]
[40]
van Brakel WH, Nicholls PG, Wilder-Smith EP, Das L, Barkataki P, Lockwood DN, INFIR Study Group. Early diagnosis of neuropathy in leprosy--comparing diagnostic tests in a large prospective study (the INFIR cohort study). PLoS neglected tropical diseases. 2008 Apr 2:2(4):e212. doi: 10.1371/journal.pntd.0000212. Epub 2008 Apr 2
[PubMed PMID: 18382604]
[42]
Scollard DM. Time and change: new dimensions in the immunopathologic spectrum of leprosy. Annales de la Societe belge de medecine tropicale. 1993:73 Suppl 1():5-11
[PubMed PMID: 8129479]
[43]
Lockwood DN, Lucas SB, Desikan KV, Ebenezer G, Suneetha S, Nicholls P. The histological diagnosis of leprosy type 1 reactions: identification of key variables and an analysis of the process of histological diagnosis. Journal of clinical pathology. 2008 May:61(5):595-600. doi: 10.1136/jcp.2007.053389. Epub 2008 Mar 6
[PubMed PMID: 18326022]
[44]
Ranque B, Nguyen VT, Vu HT, Nguyen TH, Nguyen NB, Pham XK, Schurr E, Abel L, Alcaïs A. Age is an important risk factor for onset and sequelae of reversal reactions in Vietnamese patients with leprosy. Clinical infectious diseases : an official publication of the Infectious Diseases Society of America. 2007 Jan 1:44(1):33-40
[PubMed PMID: 17143812]
[45]
Walker SL, Sales AM, Butlin CR, Shah M, Maghanoy A, Lambert SM, Darlong J, Rozario BJ, Pai VV, Balagon M, Doni SN, Hagge DA, Nery JAC, Neupane KD, Baral S, Sangma BA, Alembo DT, Yetaye AM, Hassan BA, Shelemo MB, Nicholls PG, Lockwood DNJ, Erythema Nodosum Leprosum International STudy Group. A leprosy clinical severity scale for erythema nodosum leprosum: An international, multicentre validation study of the ENLIST ENL Severity Scale. PLoS neglected tropical diseases. 2017 Jul:11(7):e0005716. doi: 10.1371/journal.pntd.0005716. Epub 2017 Jul 3
[PubMed PMID: 28671966]
Level 1 (high-level) evidence
[46]
Rea TH, Jerskey RS. Clinical and histologic variations among thirty patients with Lucio's phenomenon and pure and primitive diffuse lepromatosis (Latapi's lepromatosis). International journal of leprosy and other mycobacterial diseases : official organ of the International Leprosy Association. 2005 Sep:73(3):169-88
[PubMed PMID: 16830639]
[47]
White C, Franco-Paredes C. Leprosy in the 21st century. Clinical microbiology reviews. 2015 Jan:28(1):80-94. doi: 10.1128/CMR.00079-13. Epub
[PubMed PMID: 25567223]
[48]
Negera E, Bobosha K, Walker SL, Endale B, Howe R, Aseffa A, Dockrell HM, Lockwood DN. New Insight into the Pathogenesis of Erythema Nodosum Leprosum: The Role of Activated Memory T-Cells. Frontiers in immunology. 2017:8():1149. doi: 10.3389/fimmu.2017.01149. Epub 2017 Sep 15
[PubMed PMID: 28966621]
[49]
Negera E, Walker SL, Bekele Y, Dockrell HM, Lockwood DN. Increased activated memory B-cells in the peripheral blood of patients with erythema nodosum leprosum reactions. PLoS neglected tropical diseases. 2017 Dec:11(12):e0006121. doi: 10.1371/journal.pntd.0006121. Epub 2017 Dec 18
[PubMed PMID: 29253897]
[50]
Polycarpou A, Walker SL, Lockwood DN. A Systematic Review of Immunological Studies of Erythema Nodosum Leprosum. Frontiers in immunology. 2017:8():233. doi: 10.3389/fimmu.2017.00233. Epub 2017 Mar 13
[PubMed PMID: 28348555]
Level 1 (high-level) evidence
[51]
Silva EA, Iyer A, Ura S, Lauris JR, Naafs B, Das PK, Vilani-Moreno F. Utility of measuring serum levels of anti-PGL-I antibody, neopterin and C-reactive protein in monitoring leprosy patients during multi-drug treatment and reactions. Tropical medicine & international health : TM & IH. 2007 Dec:12(12):1450-8
[PubMed PMID: 18076551]
[52]
Gillis T, Vissa V, Matsuoka M, Young S, Richardus JH, Truman R, Hall B, Brennan P, Ideal Consortium Partners. Characterisation of short tandem repeats for genotyping Mycobacterium leprae. Leprosy review. 2009 Sep:80(3):250-60
[PubMed PMID: 19994470]
[53]
Brennan PJ. Skin test development in leprosy: progress with first-generation skin test antigens, and an approach to the second generation. Leprosy review. 2000 Dec:71 Suppl():S50-4
[PubMed PMID: 11201887]
[54]
Mitra DK, Joshi B, Dinda AK, Rai AK, Girdhar BK, Katoch K, Bindra MS, Sengupta U. Induction of lepromin reactivity in cured lepromatous leprosy patients: impaired chemokine response dissociates protective immunity from delayed type hypersensitivity. Microbes and infection. 2009 Dec:11(14-15):1122-30. doi: 10.1016/j.micinf.2009.08.006. Epub 2009 Aug 22
[PubMed PMID: 19703581]
[55]
Kaplan G, Laal S, Sheftel G, Nusrat A, Nath I, Mathur NK, Mishra RS, Cohn ZA. The nature and kinetics of a delayed immune response to purified protein derivative of tuberculin in the skin of lepromatous leprosy patients. The Journal of experimental medicine. 1988 Nov 1:168(5):1811-24
[PubMed PMID: 3053962]
[56]
Butlin CR, Soares D, Neupane KD, Failbus SS, Roche PW. IgM anti-phenolic glycolipid-I antibody measurements from skin-smear sites: correlation with venous antibody levels and the bacterial index. International journal of leprosy and other mycobacterial diseases : official organ of the International Leprosy Association. 1997 Dec:65(4):465-8
[PubMed PMID: 9465156]
[57]
Bührer SS, Smits HL, Gussenhoven GC, van Ingen CW, Klatser PR. A simple dipstick assay for the detection of antibodies to phenolic glycolipid-I of Mycobacterium leprae. The American journal of tropical medicine and hygiene. 1998 Feb:58(2):133-6
[PubMed PMID: 9502593]
[58]
Oskam L, Slim E, Bührer-Sékula S. Serology: recent developments, strengths, limitations and prospects: a state of the art overview. Leprosy review. 2003 Sep:74(3):196-205
[PubMed PMID: 14577464]
Level 3 (low-level) evidence
[59]
Douglas JT, Celona RV, Abalos RM, Madarang MG, Fajardo T. Serological reactivity and early detection of leprosy among contacts of lepromatous patients in Cebu, the Philippines. International journal of leprosy and other mycobacterial diseases : official organ of the International Leprosy Association. 1987 Dec:55(4):718-21
[PubMed PMID: 3323372]
[60]
Ulrich M, Smith PG, Sampson C, Zuniga M, Centeno M, Garcia V, Manrique X, Salgado A, Convit J. IgM antibodies to native phenolic glycolipid-I in contacts of leprosy patients in Venezuela: epidemiological observations and a prospective study of the risk of leprosy. International journal of leprosy and other mycobacterial diseases : official organ of the International Leprosy Association. 1991 Sep:59(3):405-15
[PubMed PMID: 1890364]
Level 2 (mid-level) evidence
[61]
Chanteau S, Glaziou P, Plichart C, Luquiaud P, Plichart R, Faucher JF, Cartel JL. Low predictive value of PGL-I serology for the early diagnosis of leprosy in family contacts: results of a 10-year prospective field study in French Polynesia. International journal of leprosy and other mycobacterial diseases : official organ of the International Leprosy Association. 1993 Dec:61(4):533-41
[PubMed PMID: 8151183]
[62]
Aseffa A, Brennan P, Dockrell H, Gillis T, Hussain R, Oskam L, Richardus JH, Ideal Consortium. Report on the first meeting of the IDEAL (Initiative for Diagnostic and Epidemiological Assays for Leprosy) consortium held at Armauer Hansen Research Institute, ALERT, Addis Ababa, Ethiopia on 24-27 October 2004. Leprosy review. 2005 Jun:76(2):147-59
[PubMed PMID: 16038248]
Level 2 (mid-level) evidence
[63]
Dean DA, Davidson AL, Nikaido H. The role of ATP as the energy source for maltose transport in Escherichia coli. Research in microbiology. 1990 Mar-Apr:141(3):348-52
[PubMed PMID: 2177914]
[64]
Ji B. Why multidrug therapy for multibacillary leprosy can be shortened to 12 months. Leprosy review. 1998 Jun:69(2):106-9
[PubMed PMID: 9715593]
[65]
Freerksen E, Rosenfeld M, Depasquale G, Bonnici E, Gatt P. The Malta Project--a country freed itself of leprosy. A 27-year progress study (1972-1999) of the first successful eradication of leprosy. Chemotherapy. 2001 Sep-Oct:47(5):309-31
[PubMed PMID: 11561134]
[66]
Sotiriou MC, Stryjewska BM, Hill C. Two Cases of Leprosy in Siblings Caused by Mycobacterium lepromatosis and Review of the Literature. The American journal of tropical medicine and hygiene. 2016 Sep 7:95(3):522-7. doi: 10.4269/ajtmh.16-0076. Epub 2016 Jul 11
[PubMed PMID: 27402522]
Level 3 (low-level) evidence
[67]
Scollard DM. Infection with Mycobacterium lepromatosis. The American journal of tropical medicine and hygiene. 2016 Sep 7:95(3):500-501. doi: 10.4269/ajtmh.16-0473. Epub 2016 Jul 18
[PubMed PMID: 27430540]
[68]
FAGET GH, POGGE RC, JOHANSEN FA. Promizole treatment of leprosy. Public health reports (Washington, D.C. : 1896). 1946 Jun 28:61():957-60
[PubMed PMID: 20990604]
[69]
LOWE J. Treatment of leprosy with diamino-diphenyl sulphone by mouth. Lancet (London, England). 1950 Jan 28:1(6596):145-50
[PubMed PMID: 15401496]
[70]
PETTIT JH, REES RJ. SULPHONE RESISTANCE IN LEPROSY. AN EXPERIMENTAL AND CLINICAL STUDY. Lancet (London, England). 1964 Sep 26:2(7361):673-4
[PubMed PMID: 14188912]
[71]
. Chemotherapy of leprosy for control programmes. World Health Organization technical report series. 1982:675():1-33
[PubMed PMID: 6806990]
[72]
Levy L, Ng H, Evans MJ, Krahenbuhl JL. Susceptibility of thymectomized and irradiated mice to challenge with several organisms and the effect of dapsone on infection with Mycobacterium leprae. Infection and immunity. 1975 May:11(5):1122-32
[PubMed PMID: 804443]
[73]
Holmes IB, Banerjee DK, Hilson GR. Effect of rifampin, clofazimine, and B1912 on the viability of Mycobacterium leprae in established mouse footpad infection. Proceedings of the Society for Experimental Biology and Medicine. Society for Experimental Biology and Medicine (New York, N.Y.). 1976 Apr:151(4):637-41
[PubMed PMID: 772686]
[74]
Franzblau SG. Drug susceptibility testing of Mycobacterium leprae in the BACTEC 460 system. Antimicrobial agents and chemotherapy. 1989 Dec:33(12):2115-7
[PubMed PMID: 2694952]
[75]
Dacso MM, Jacobson RR, Scollard DM, Stryjewska BM, Prestigiacomo JF. Evaluation of multi-drug therapy for leprosy in the United States using daily rifampin. Southern medical journal. 2011 Oct:104(10):689-94. doi: 10.1097/SMJ.0b013e31822d6014. Epub
[PubMed PMID: 21941157]
[77]
Landaas S. Inhibition of branched-chain amino acid degradation by ketone bodies. Scandinavian journal of clinical and laboratory investigation. 1977 Sep:37(5):411-8
[PubMed PMID: 929096]
[78]
Ji B, Jamet P, Sow S, Perani EG, Traore I, Grosset JH. High relapse rate among lepromatous leprosy patients treated with rifampin plus ofloxacin daily for 4 weeks. Antimicrobial agents and chemotherapy. 1997 Sep:41(9):1953-6
[PubMed PMID: 9303392]
[79]
Katoch K, Natarajan M, Bagga A, Katoch VM. Clinical and bacteriological progress of highly bacillated BL-LL patients discontinuing treatment after different periods of MDT. International journal of leprosy and other mycobacterial diseases : official organ of the International Leprosy Association. 1991 Jun:59(2):248-54
[PubMed PMID: 2071982]
[80]
Franzblau SG. In vitro activities of aminoglycosides, lincosamides, and rifamycins against Mycobacterium leprae. Antimicrobial agents and chemotherapy. 1991 Jun:35(6):1232-4
[PubMed PMID: 1929269]
[81]
Kamath S, Vaccaro SA, Rea TH, Ochoa MT. Recognizing and managing the immunologic reactions in leprosy. Journal of the American Academy of Dermatology. 2014 Oct:71(4):795-803. doi: 10.1016/j.jaad.2014.03.034. Epub 2014 Apr 24
[PubMed PMID: 24767732]
[82]
Walker SL, Lockwood DN. Leprosy type 1 (reversal) reactions and their management. Leprosy review. 2008 Dec:79(4):372-86
[PubMed PMID: 19274984]
[83]
Rao PS, Sugamaran DS, Richard J, Smith WC. Multi-centre, double blind, randomized trial of three steroid regimens in the treatment of type-1 reactions in leprosy. Leprosy review. 2006 Mar:77(1):25-33
[PubMed PMID: 16715687]
Level 1 (high-level) evidence
[84]
Smith WC, Anderson AM, Withington SG, van Brakel WH, Croft RP, Nicholls PG, Richardus JH. Steroid prophylaxis for prevention of nerve function impairment in leprosy: randomised placebo controlled trial (TRIPOD 1). BMJ (Clinical research ed.). 2004 Jun 19:328(7454):1459
[PubMed PMID: 15159285]
Level 1 (high-level) evidence
[85]
Biosca G, Casallo S, López-Vélez R. Methotrexate treatment for type 1 (reversal) leprosy reactions. Clinical infectious diseases : an official publication of the Infectious Diseases Society of America. 2007 Jul 1:45(1):e7-9
[PubMed PMID: 17554691]
[86]
Marlowe SN, Leekassa R, Bizuneh E, Knuutilla J, Ale P, Bhattarai B, Sigdel H, Anderson A, Nicholls PG, Johnston A, Holt D, Lockwood DN. Response to ciclosporin treatment in Ethiopian and Nepali patients with severe leprosy Type 1 reactions. Transactions of the Royal Society of Tropical Medicine and Hygiene. 2007 Oct:101(10):1004-12
[PubMed PMID: 17669450]
[87]
Sena CB, Salgado CG, Tavares CM, Da Cruz CA, Xavier MB, Do Nascimento JL. Cyclosporine A treatment of leprosy patients with chronic neuritis is associated with pain control and reduction in antibodies against nerve growth factor. Leprosy review. 2006 Jun:77(2):121-9
[PubMed PMID: 16895068]
[88]
Van Veen NH, Lockwood DN, Van Brakel WH, Ramirez J Jr, Richardus JH. Interventions for erythema nodosum leprosum. A Cochrane review. Leprosy review. 2009 Dec:80(4):355-72
[PubMed PMID: 20306635]
[89]
SHESKIN J. THALIDOMIDE IN THE TREATMENT OF LEPRA REACTIONS. Clinical pharmacology and therapeutics. 1965 May-Jun:6():303-6
[PubMed PMID: 14296027]
[90]
Ramien ML, Wong A, Keystone JS. Severe refractory erythema nodosum leprosum successfully treated with the tumor necrosis factor inhibitor etanercept. Clinical infectious diseases : an official publication of the Infectious Diseases Society of America. 2011 Mar 1:52(5):e133-5. doi: 10.1093/cid/ciq213. Epub
[PubMed PMID: 21292656]
[91]
Faber WR, Jensema AJ, Goldschmidt WF. Treatment of recurrent erythema nodosum leprosum with infliximab. The New England journal of medicine. 2006 Aug 17:355(7):739
[PubMed PMID: 16914716]
[92]
Nathan C, Squires K, Griffo W, Levis W, Varghese M, Job CK, Nusrat AR, Sherwin S, Rappoport S, Sanchez E. Widespread intradermal accumulation of mononuclear leukocytes in lepromatous leprosy patients treated systemically with recombinant interferon gamma. The Journal of experimental medicine. 1990 Nov 1:172(5):1509-12
[PubMed PMID: 2121891]
[93]
Kaplan G. Recent advances in cytokine therapy in leprosy. The Journal of infectious diseases. 1993 Mar:167 Suppl 1():S18-22
[PubMed PMID: 8433015]
Level 3 (low-level) evidence
[94]
Kaplan G, Britton WJ, Hancock GE, Theuvenet WJ, Smith KA, Job CK, Roche PW, Molloy A, Burkhardt R, Barker J. The systemic influence of recombinant interleukin 2 on the manifestations of lepromatous leprosy. The Journal of experimental medicine. 1991 Apr 1:173(4):993-1006
[PubMed PMID: 2007862]
[95]
Kaplan G, Mathur NK, Job CK, Nath I, Cohn ZA. Effect of multiple interferon gamma injections on the disposal of Mycobacterium leprae. Proceedings of the National Academy of Sciences of the United States of America. 1989 Oct:86(20):8073-7
[PubMed PMID: 2510159]
[96]
Sampaio EP, Moreira AL, Sarno EN, Malta AM, Kaplan G. Prolonged treatment with recombinant interferon gamma induces erythema nodosum leprosum in lepromatous leprosy patients. The Journal of experimental medicine. 1992 Jun 1:175(6):1729-37
[PubMed PMID: 1588290]
[97]
Stefani MM, Guerra JG, Sousa AL, Costa MB, Oliveira ML, Martelli CT, Scollard DM. Potential plasma markers of Type 1 and Type 2 leprosy reactions: a preliminary report. BMC infectious diseases. 2009 May 27:9():75. doi: 10.1186/1471-2334-9-75. Epub 2009 May 27
[PubMed PMID: 19473542]
[98]
Scollard DM, Chaduvula MV, Martinez A, Fowlkes N, Nath I, Stryjewska BM, Kearney MT, Williams DL. Increased CXC ligand 10 levels and gene expression in type 1 leprosy reactions. Clinical and vaccine immunology : CVI. 2011 Jun:18(6):947-53. doi: 10.1128/CVI.00042-11. Epub 2011 Apr 20
[PubMed PMID: 21508169]
[100]
Cambau E, Saunderson P, Matsuoka M, Cole ST, Kai M, Suffys P, Rosa PS, Williams D, Gupta UD, Lavania M, Cardona-Castro N, Miyamoto Y, Hagge D, Srikantam A, Hongseng W, Indropo A, Vissa V, Johnson RC, Cauchoix B, Pannikar VK, Cooreman EAWD, Pemmaraju VRR, Gillini L, WHO surveillance network of antimicrobial resistance in leprosy. Antimicrobial resistance in leprosy: results of the first prospective open survey conducted by a WHO surveillance network for the period 2009-15. Clinical microbiology and infection : the official publication of the European Society of Clinical Microbiology and Infectious Diseases. 2018 Dec:24(12):1305-1310. doi: 10.1016/j.cmi.2018.02.022. Epub 2018 Mar 1
[PubMed PMID: 29496597]
Level 3 (low-level) evidence
[101]
. Randomised controlled trial of single BCG, repeated BCG, or combined BCG and killed Mycobacterium leprae vaccine for prevention of leprosy and tuberculosis in Malawi. Karonga Prevention Trial Group. Lancet (London, England). 1996 Jul 6:348(9019):17-24
[PubMed PMID: 8691924]
Level 1 (high-level) evidence
[102]
Bertolli J, Pangi C, Frerichs R, Halloran ME. A case-control study of the effectiveness of BCG vaccine for preventing leprosy in Yangon, Myanmar. International journal of epidemiology. 1997 Aug:26(4):888-96
[PubMed PMID: 9279624]
Level 2 (mid-level) evidence
[103]
Düppre NC, Camacho LA, da Cunha SS, Struchiner CJ, Sales AM, Nery JA, Sarno EN. Effectiveness of BCG vaccination among leprosy contacts: a cohort study. Transactions of the Royal Society of Tropical Medicine and Hygiene. 2008 Jul:102(7):631-8. doi: 10.1016/j.trstmh.2008.04.015. Epub 2008 Jun 2
[PubMed PMID: 18514242]
[104]
Gillini L, Cooreman E, Wood T, Pemmaraju VR, Saunderson P. Global practices in regard to implementation of preventive measures for leprosy. PLoS neglected tropical diseases. 2017 May:11(5):e0005399. doi: 10.1371/journal.pntd.0005399. Epub 2017 May 4
[PubMed PMID: 28472183]