Pathophysiology
A broad range of injury severity exists in facial nerve trauma due to the various mechanisms by which injuries may be produced. Temporal bone fractures tend to cause crush injuries that evolve over the course of hours to days, and sometimes even weeks, although they certainly can, in rare cases, result in transection of the nerve and immediate hemifacial paralysis.[7] Similarly, iatrogenic injuries tend to result from traction or thermal damage from nearby electrocautery but may also involve partial or complete nerve transection, as may be the case when drilling in the mastoid cavity. Likewise, penetrating facial injuries may either stretch or transect, either partially or completely, the affected nerve branch. Frustratingly, the extent of the trauma is often unclear at the time of injury. When conceptualizing nerve injury, two classification schemes are commonly used to describe severity: the Seddon and Sunderland scales (see table).[10][11]
The Seddon scale is simpler, and focused primarily on implications for management, while the Sunderland scale characterizes the extent of injury with respect to the microanatomy of the nerve involved. At its most basic, the functional unit of a motor nerve is the axon, which is the long filament that connects the cell body, located in the pons, to the motor end plate at the neuromuscular junction. The motor axons of the facial nerve, of which there are approximately 6,000, are insulated in myelin produced by Schwann cells.[12] Surrounding the myelin sheath is the endoneurium, a thin connective tissue membrane. These individually-wrapped axons are bundled into fascicles, which are, in turn, surrounded by perineurial membranes. Lastly, the nerve itself is made up of fascicles and surrounded by epineurium.
Seddon Classification
Neurapraxia: a localized conduction block with focal demyelination and no Wallerian degeneration - complete recovery is expected.
Axonotmesis: a variable amount of axonal injury with Wallerian degeneration and an intact epineurium - synkinesis may occur in more severe cases.
Neurotmesis: complete nerve transection with Wallerian degeneration - flaccidity will persist unless the nerve is repaired.
Sunderland Classification
- Class I: equivalent to Seddon's neurapraxia - complete recovery is expected,
- Class II: mild axonotmesis in which the endoneurium remains intact, but Wallerian degeneration occurs - complete recovery is expected.
- Class III: moderate axonotmesis in which the endoneurium is disrupted and Wallerian degeneration occurs, but the perineurium remains intact - mild to moderate synkinesis is expected.
- Class IV: severe axonotmesis in which the endoneurium and perineurium are disrupted, and Wallerian degeneration occurs, but the epineurium remains intact - severe synkinesis is expected.
- Class V: equivalent to Seddon neurotmesis - flaccidity will persist unless the nerve is repaired.
To understand either of these classification systems, it is critical also to be familiar with the concept of Wallerian degeneration. Wallerian degeneration is the process by which axons and Schwann cells disintegrate and are removed in order to make room for regeneration of a new axon.[13] This process begins at the node of Ranvier just proximal to the site of injury and proceeds distally out to the neuromuscular junction, thus clearing the way for the injured axon to grow back to the neuromuscular junction at a rate of approximately 1 mm per day.[14] In cases of total nerve transection, Wallerian degeneration takes roughly 72 hours, but the process may take days or weeks to finish in cases of crush injuries.[15] Once Wallerian degeneration is complete, at least in the case of nerve transection injuries, the distal stump of the nerve is no longer stimulable, which may make exploration and repair more challenging; for this reason, most surgeons prefer to explore the facial nerve early if there is a high index of suspicion for transection.
In cases of nerve transection, the gold standard treatment is primary repair of the epineurium, which effectively converts a Sunderland class V injury into a class IV injury. Regardless, the injury remains severe, and any facial nerve injury of Sunderland class III or higher will likely result in synkinesis due to the disruption of internal neural architecture. In Sunderland class I and II injuries, the epineurium - the membrane that surrounds the myelin covering the axon - is preserved; with this membrane intact, the affected axons will regrow back to their original motor endplates, and complete recovery should occur. Sunderland class III-V injuries involve progressively greater degrees of disruption of the internal neural architecture, with correspondingly greater potential for misdirection of regenerating axons and resulting aberrant reinnervation. This aberrant reinnervation may take the form of involuntary movements accompanying voluntary movements and/or greater resting tension in the affected muscles; these symptoms are termed "synkinesis." Unfortunately, there is no way to correlate a clinical or laboratory examination with the Sunderland classification, making precise prognostication of functional recovery difficult. Electroneuronography, which is a type of evoked electromyography, can be used to tell the difference between neurapraxia and anything more severe, but cannot provide more anatomical detail than that.
Beyond the issue of the severity of the nerve injury itself, there remains the question of what risk factors and mechanisms of injury are likely to produce facial nerve trauma. For temporal bone fractures, the amount of force and the vector with which it is applied are the primary determinants of the type of injury sustained and whether or not facial paralysis will result. The Ulrich classification of temporal bone fractures was suggested in 1926 and describes the orientation of the fracture relative to the petrous ridge of the temporal bone.[8] Longitudinal fractures (approximately 80%) run parallel to the long axis of the petrous ridge. They are typically produced by blows directed laterally towards the skull, as may be obtained during a physical altercation. These fractures rarely involve the otic capsule, as the fracture line follows the path of least resistance towards the petrous apex of the temporal bone; however, ossicular chain disruption, hemotympanum, tympanic membrane perforation, and otorrhea, as well as conductive hearing loss, are often associated with these injuries. Fortunately, facial nerve palsy occurs in only about 20% of longitudinal temporal bone fractures.[8]
Transverse fractures (approximately 20%) run perpendicular to the long axis of the petrous ridge and often straight through the labyrinthine capsule. They are often caused by force-directed anteroposteriorly, such as a motor vehicle accident. These fractures tend to originate near the jugular foramen or foramen magnum and extend into the middle cranial fossa. Consequently, these fractures commonly result in sensorineural hearing loss, which may result from transection of the cochlear nerve, damage to the labyrinthine structures, or stapes footplate injury. They are associated with perilymphatic fistula (with nystagmus away from the fractured side) and facial palsy in 50% of cases.[8] Many fractures, however, do not fit well into this classification scheme, which led to the development of the otic capsule-sparing vs disrupting system described by Brodie and Thompson in 1997.[16]
In this scheme, temporal bone fractures are categorized based on whether or not they spare or disrupt the bone of the otic capsule. Otic capsule-sparing fractures (approximately 95%) tend to result from laterally-directed blows to the skull and are only associated with facial paralysis in about 6% of cases. Otic capsule-disrupting fractures only account for 5% of temporal bone fractures; however, up to half of these injuries will result in facial paralysis.[16] The facial paralysis that accompanies temporal bone fractures is somewhat unique when compared to other types of facial nerve trauma because the bony Fallopian canal, which encases the facial nerve during its intratemporal course, makes it difficult for the injured nerve to swell without compromising its own perfusion.[17]
In the case of extratemporal facial nerve trauma, the edematous nerve is unlikely to become compressed and can therefore maintain its perfusion. With extratemporal injuries, even delayed paralysis will likely present within a matter of several hours to a few days. Within the temporal bone, however, even comparatively minor facial nerve insults can result in edema that progresses very slowly over the course of days to weeks before reaching a point at which perfusion is compromised, and nerve function is impaired.
From the cerebellopontine angle, where the facial nerve exits the pons, to the stylomastoid foramen, where it exits the skull, the facial nerve takes a wandering course through the temporal bone. It enters the bone through the internal auditory meatus, along with the cochlear and vestibular nerves, occupying the anterosuperior position within the internal auditory canal, where it travels for 8 to 10 mm before separating from the other nerves and arriving at the labyrinthine segment. Here, it enters the Fallopian canal and makes its first turn, or "genu," at the geniculate ganglion, where the greater superficial petrosal nerve is given off to provide parasympathetic innervation to the lacrimal gland and the mucous glands of the nose and palate. In the ~4 mm long labyrinthine segment, the overall diameter of the bony canal narrows to approximately 0.7 mm, its narrowest portion, and therefore the area most likely to be affected in temporal bone fractures.[18][19]
The tympanic or "horizontal" segment follows, roughly 9 to 10 mm in length, where the facial nerve courses superior to the stapes footplate within the middle ear and may occasionally be incompletely covered by bone, rendering it vulnerable to otological pathologies, such as infection or barotrauma. Finally, the nerve turns again at its second genu to travel inferiorly through the mastoid cavity, where it gives off the chorda tympani nerve to provide taste sensation to the anterior two-thirds of the tongue; this portion is known as the mastoid or "vertical" segment, and it is 11 to 12 mm long, ending where the nerve passes through the stylomastoid foramen.[20]
After exiting the skull, the main trunk of the facial nerve turns anteriorly to enter the parotid gland, where it divides into the main extratemporal branches at the "pes anserinus," so named because the separation of branches resembles the foot of a goose. These branches serve as the division between the superficial and deep lobes of the parotid gland. From superior to inferior, the branches are the frontal (which controls the muscles of the forehead and upper eyelid), the zygomatic (which controls the muscles of the eyelids and midface), the buccal (which controls the muscles of the midface and mouth), the marginal mandibular (which controls the depressors of the lower lip), and the cervical (which controls the platysma and some depressors of the lower lip as well). Branching patterns and control of specific muscles vary somewhat between individuals, but the facial nerve branches run deep to the superficial musculoaponeurotic system of the face and the temporoparietal fascia of the scalp as well as deep to all the mimetic muscles other than the mentalis, buccinator, and levator anguli oris. Additionally, the facial nerve innervates the auricular muscles, the stapedius muscle, the stylohyoid muscle, and the posterior belly of the digastric muscle.
As mentioned above, in some patients, the facial nerve is anatomically susceptible to barotrauma, which is a comparatively uncommon occurrence. Case reports have been published detailing the stories of a handful of patients who have suffered acute facial paralysis due to SCUBA diving or traveling at high altitudes in an airplane or a car driving through a mountain pass.[21][22][23][24] The pathogenesis of the palsy has not been clearly elucidated, but it may be a result of dehiscence of the facial nerve within the middle ear, making it susceptible to pressure changes, particularly in the setting of Eustachian tube dysfunction. In some cases, hearing loss and subdural air on neuroimaging have also been reported with facial paralysis.[22]
Treatment / Management
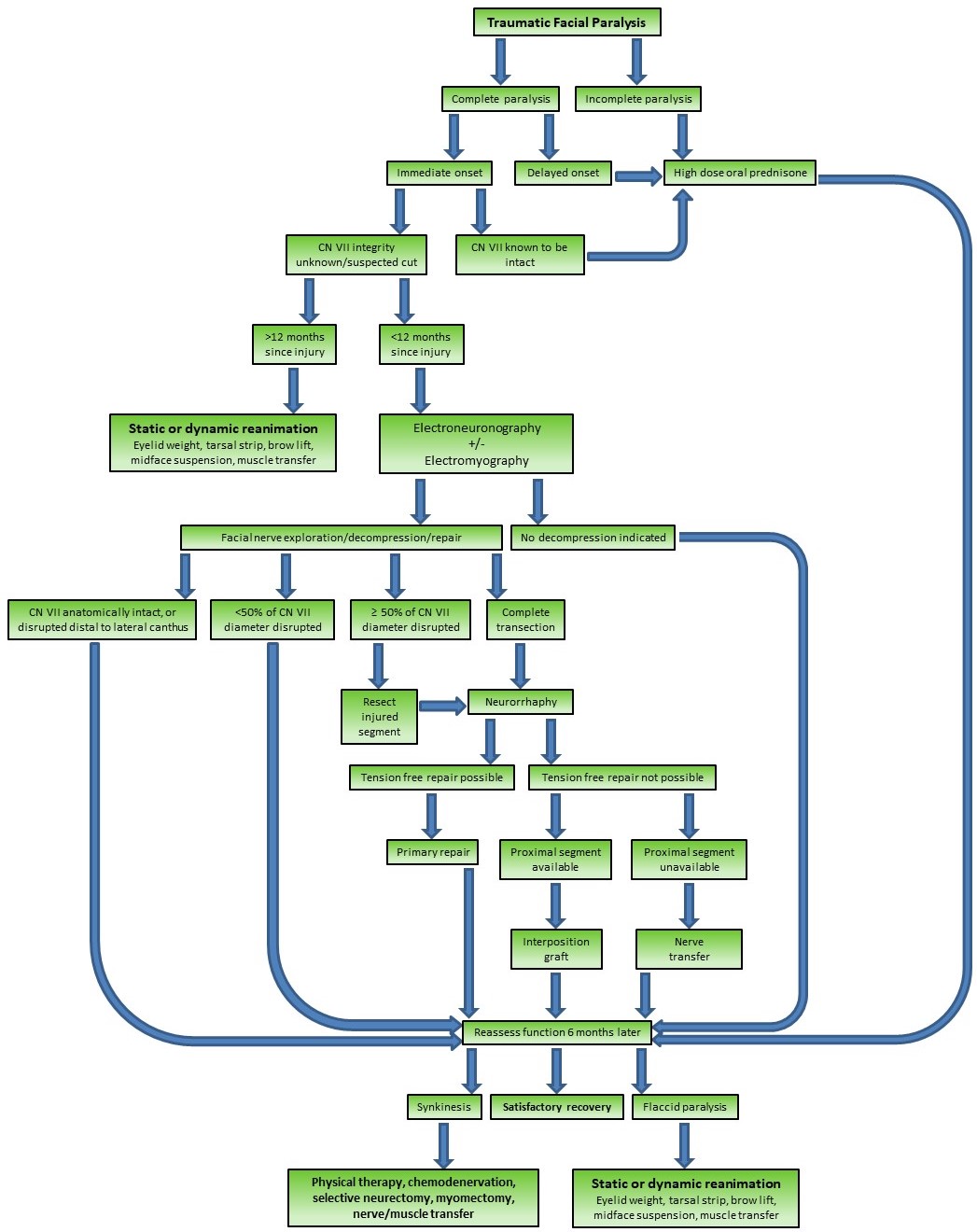
In essence, the goal of treating traumatic facial paralysis must be to preserve or restore as much facial mimetic function as possible. This can be accomplished in myriad ways, depending upon the severity of the injury and the interval between when the injury occurred and when the patient seeks care.
Acute injury: <12 Months Since Injury
Medical Management
For the majority of patients with acute traumatic facial paralysis, similar to patients suffering from viral reactivation facial paralyses, a course of high-dose oral steroids (prednisone 60 mg PO daily x 5 days, with an additional 5-day taper) can improve outcomes by reducing inflammation and edema of the facial nerve.[2][32][33] Care should be taken when prescribing corticosteroids to patients with diabetes, patients with gastric/peptic ulcers, patients already taking steroids, patients taking fluoroquinolones, and others, due to the potential adverse effects of steroids. In addition to systemic corticosteroids, there is some evidence that dihydropyridine calcium channel blockers, specifically nimodipine, can improve outcomes after facial nerve trauma by reducing apoptosis and improving axonal sprouting.[34] Nifedipine has been investigated as well, but the evidence is less clear that this medication improves facial nerve outcomes clinically, although animal studies in which nifedipine was applied topically to the injured nerve do demonstrate better histological recovery with nifedipine than control subjects had.
Eye Care
Patients with acute facial paralysis of any etiology require close follow-up to ensure the health of the corneal surface, particularly patients with intracranial injuries to the facial nerve that may have also affected the trigeminal nerve and caused corneal hypesthesia. Most patients with paralytic lagophthalmos are acutely aware of any ocular issues that occur, such as corneal abrasions or ulcerations, because of the pain that accompanies them, but patients lacking the ability to feel corneal injuries are at a high risk of developing ocular surface complications.[1]
Likewise, patients with poor Bell's phenomena should be considered high-risk and followed more carefully or treated more aggressively. Standard treatment for paralytic lagophthalmos should include artificial tear drops as needed during the day, along with eyelid stretching exercises and ocular lubricant at night, with eyelid taping if necessary.[35] For patients at high risk for ocular surface injury and who continue to have ocular discomfort despite the conservative management previously mentioned, fitting for a scleral contact lens or placement of an upper eyelid weight should be considered.[36][37] In some cases, additional periocular procedures may also be required, such as lateral tarsal strip canthopexy, tarsorrhaphy, or tarsoconjunctival flap transfer.
Nerve Transection
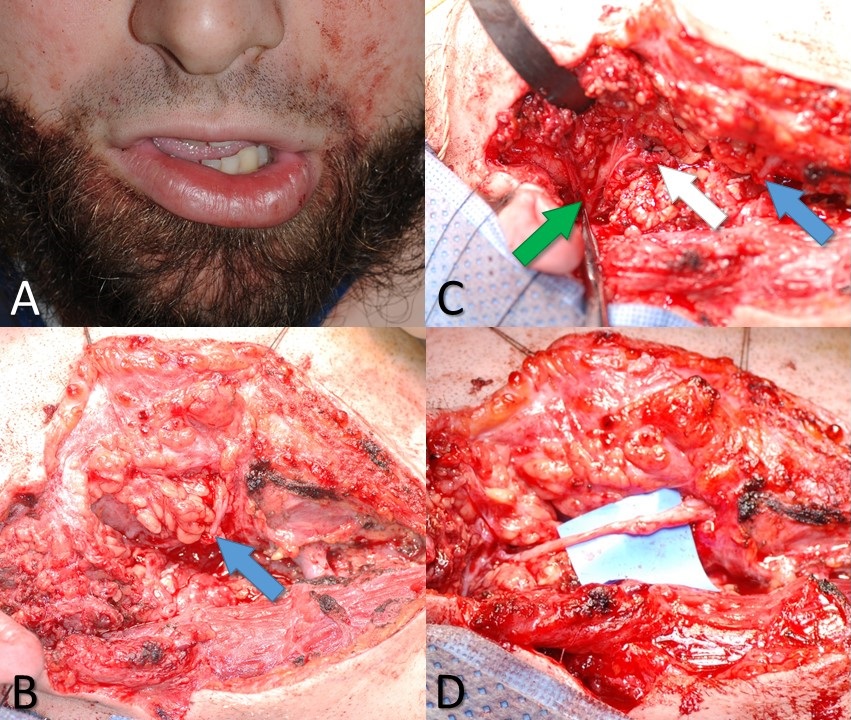
When the facial nerve is known to have been transected, or there is a high suspicion that this has occurred, an effort should be made to locate the injury site and repair the nerve. Ideally, the exploration will occur within the first 72 hours after injury so that the process of Wallerian degeneration will not have been completed yet; this will keep the distal stump stimulable and, therefore, easier to find in the wound bed. The proximal stump can always be located via antegrade dissection from the main trunk of the facial nerve or the pes anserinus. For cases of very proximal injury, the nerve may be located within the mastoid and followed distally through the stylomastoid foramen, where it exits the skull base. Other methods of identifying the main trunk of the facial nerve include following the anterior surface of the tragal cartilage until reaching the tip (tragal pointer) and then continuing the dissection deeper for 1 cm and 1 cm more inferiorly. The tympanomastoid suture line is also palpable, and this can be followed to within about 3 mm of the stylomastoid foramen.[38]
The approach to the facial nerve exploration will vary depending on the suspected location of the injury, but in the face itself, the nerve is typically visualized via the preauricular portion of a Blair incision, which provides access to the extratemporal main truck of the facial nerve as well as the pes anserinus and the main branches. If the injury is located medial to the lateral canthus, as may occur with a facial laceration, the nerve branch does not usually need to be repaired because of the degree of redundancy among distal midfacial branches and the high likelihood of spontaneous functional recovery. When preparing for surgery, it is important to avoid injecting local anesthetics into the area (in some patients, lidocaine may diffuse farther than expected and last longer than two hours; injection of plain 1:100,000 epinephrine may be preferable) and to request that the anesthesia provider not administer a long-acting paralytic medication, which will interfere with the use of a nerve stimulator to find the distal stump.
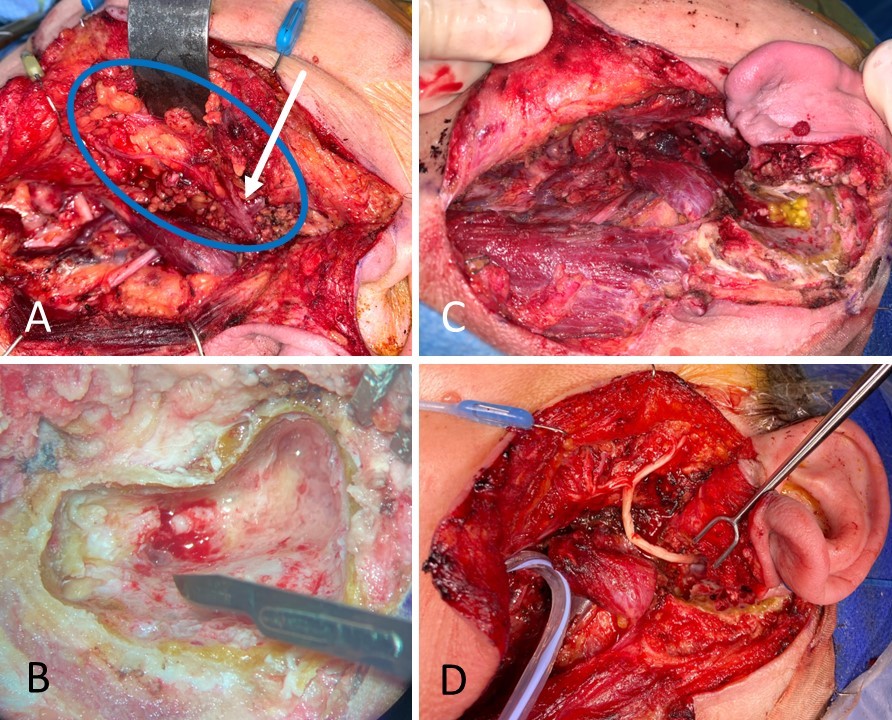
In some cases, rather than encountering a transection injury, the surgeon may find the nerve intact but non-stimulable and surrounded by scar tissue. External neurolysis - removing the scar tissue from around the epineurium - may reduce inflammation and improve function. If the scar tissue appears to penetrate the epineurium and involve the interior of the nerve, the nerve should be divided and trimmed back to normal neural tissue, which should demonstrate distinct fascicles and pinpoint bleeding. Significant scarring within the nerve itself may indicate a severe crush or shearing injury or even a transection injury that failed to distract the nerve stumps far enough away from each other to prevent coaptation during healing. Segmental resection of the nerve is also recommended when a portion of the nerve has had >50% of its diameter disrupted, which would likely result in extensive scarring; a common clinical scenario resulting in this type of injury is mastoidectomy, in which a drill is inadvertently raked along the facial nerve. Identifying scarring within the nerve is critical because scarring will prevent proper axonal regeneration, resulting in suboptimal recovery. Internal neurolysis, and removal of scar tissue from within the nerve, must be performed to optimize outcomes in these cases. Occasionally, after debriding the nerve stumps back to normal tissue, the ends may not be sewn back together easily without tension. Tension-free coaptation of the nerve ends is critical because stretching the nerves compromises perfusion, which in turn compromises healing and subsequent functional recovery. A gap of up to 6 mm between nerve ends is acceptable to relieve tension, provided some form of the sheath is applied to contain the regenerating axons.[39][40]
If the gap between nerve ends is going to be >6 mm, placement of an interposition graft ("cable graft") is preferable to applying excessive tension to the nerve ends or excessively dissecting around the nerve stumps to mobilize them, both of which will decrease perfusion. The most common source of cable graft material is the greater auricular nerve, which is a good size match for the main trunk of the facial nerve and is conveniently located such that it can be harvested via the same incision. The greater auricular nerve travels on the superficial surface of the sternocleidomastoid muscle from Erb's point superiorly toward the lobule of the auricle, coursing roughly halfway between the angle of the mandible and mastoid tip, 1 to 2 cm posterior to the external jugular vein.[41]
Other commonly harvested sensory nerve grafts include the sural nerve, which is particularly useful for providing long grafts for cross-face applications, and the medial antebrachial cutaneous nerve, which has a branching pattern similar to that of the facial nerve. Of note, when using a non-branching graft, it is important to rotate the graft 180 degrees before suturing it in place to maximize the number of axons that successfully traverse the length of the graft. Because even apparently non-branching grafts, such as the proximal greater auricular nerve, still have microscopic branches, coapting the distal end of the graft to the proximal facial nerve stump and the proximal end of the graft to the distal facial nerve stump helps to prevent axonal loss via unseen branches. Doing everything possible to minimize axonal loss during regeneration is critical because each neurorrhaphy represents the potential to lose up to half of the regenerating axons, which means that up to 75% of axons could be lost traversing the two neurorrhaphies required for the use of a cable graft.[42] Some surgeons will use cadaveric nerve grafts rather than harvesting sensory nerves from the patient, but there is evidence that these grafts do not perform reliably.[43][44]
From a technical standpoint, the neurorrhaphy should be performed under high magnification, typically using an operating microscope, with a 9-0 or 10-0 nylon suture on a cutting or spatula-tip needle because it can be difficult to pass a tapered needle through epineurium. Ideally, the nerve ends will be brought together so that they are just touching but not pulled firmly together. Sutures are typically placed in an interrupted fashion into the epineurium, and it is rare that more than 3 or 4 are required. If fascicles herniate out through the suture line, either the suture line should be replaced with less tension, or the fascicles should be amputated to allow them to fit back through the neurorrhaphy. While it may be tempting to place sutures into the perineurium of the fascicles before repairing the endoneurium, it is nearly impossible to determine which fascicles should be coapted to each other with any degree of precision. Therefore outcomes of perineurial and epineurial repair are equivalent, despite the greater time and effort required to repair the nerve at a fascicular level.[45][46] Pathophysiologically speaking, the goal of a neurorrhaphy is to take a Sunderland class V injury and convert it to a class IV injury, which in the main trunk of the facial nerve improves the prognosis from long-term flaccidity to synkinetic movement. Patients undergoing repair or grafting of the facial nerve main trunk should be counseled to expect synkinesis and informed that they would require rehabilitation as function returns.
While primary repair of the facial nerve or cable grafting is preferred, there are situations in which these options are unavailable. The classic example is the case of a skull base tumor in which the proximal facial nerve has been extirpated intracranially, or the facial nucleus itself has been rendered nonfunctional or absent due to central nervous system pathology. For these patients, a new axonal source is required to populate the distal facial nerve and reinnervate the mimetic muscles. Historically, the most commonly employed options were the placement of a cross-face nerve graft or using a hypoglossal-facial nerve transfer. Cross-face nerve grafting typically uses a length of sural nerve to connect the facial nerve on the unaffected side to the paralyzed side, most often sacrificing a single buccal branch on the uninjured side to send axons to a buccal branch on the paralyzed side for the purposes of smile rehabilitation. The sural nerve contains approximately 1,100 axons, which provides enough room for the ~900 axons of the usual donor buccal branch to regenerate across the face into the recipient nerve.[47][48]
Cross-face nerve grafting is often used to power free muscle flaps for smile rehabilitation (see below). The decision to use a cross-face nerve graft should only be made after considering the following points: 1) results of cross-face grafting are generally better in younger patients, who presumably have greater neural regeneration potential, and are unreliable in older patients; 2) placement of a cross-face graft requires dissecting the uninjured facial nerve, which places it at risk for injury, even though sacrificing a buccal branch on the uninjured side does not typically result in noticeable weakness, and 3) because the cross-face graft requires normal function on the donor side, patients with bilateral injuries are not candidates, nor are patients at risk for contralateral facial paralysis, such as those with type 2 neurofibromatosis.[49]
However, the primary advantage of a cross-face nerve graft is that it is the only nerve transfer option that reliably provides a spontaneous smile because the signal source for the mimetic muscles after cross-face nerve grafting is still a facial nerve. Smiling after hypoglossal-facial transfer, on the other hand, requires the patient to move the tongue to activate the facial muscles. Hypoglossal-facial transfer also comes with an important caveat: the outcome of the facial reanimation is proportional to the number of axons transferred, but the functional deficit in the tongue is also proportional to the number of axons transferred. The main trunk of the facial nerve contains about 6,000 axons, while the hypoglossal nerve contains approximately 9,000 axons, making a transfer of the entire hypoglossal nerve a potentially good match for the facial nerve, accounting for a 33% loss of axons due to neurorrhaphy.[12][50][51]
This procedure will likely provide good resting facial tone and facial movement with tongue motion, but tongue function will be severely compromised, and facial movement will be severely synkinetic. Transferring approximately 1/3 of the hypoglossal nerve's diameter end-to-end into the facial nerve will provide less tone and movement but will also limit the lingual function deficit. An interposition "jump" graft, often using the greater auricular nerve, can also be placed end-to-side between the main trunk of the facial nerve and the hypoglossal nerve if some facial function remains and the surgeon is reluctant to divide the facial nerve. Alternatively, if the main trunk of the facial nerve is already divided, but the surgeon is reluctant to divide the hypoglossal nerve, the facial nerve trunk may be sutured end-to-side into the hypoglossal nerve. Regardless of the chosen transfer technique, the use of end-to-side neurorrhaphies will result in fewer transferred axons than an end-to-end coaptation but also cause less functional loss in the tongue. To perform an end-to-side neurorrhaphy, a window must be cut into the epineurium of the nerve whose side is being used, and dividing some of the fascicles within that nerve will also promote axonal growth. One way to minimize facial synkinesis is to perform the hypoglossal-facial transfer from the hypoglossal nerve to a single branch of the facial nerve, such as the branch to the zygomaticus major muscle, which will have the effect of focusing the signal from the hypoglossal nerve on a single facial movement but will also prevent the restoration of resting tone. Another solution is to transfer the masseteric nerve to a branch of the facial nerve, which will provide control over a single facial movement, such as smiling.[52]
The masseteric nerve, which is found 3 cm anterior to the tragus, 1 cm inferior to the zygomatic arch, and 1.5 cm deep to the masseteric fascia, is a reliable reinnervation option for this purpose and contains around 1,500 axons, which should be sufficient to replace the ~900 axons found in the average buccal branch of the facial nerve.[53][12][54]
Unlike the hypoglossal nerve, the masseteric nerve has a low basal firing rate, which makes it a poor choice for restoring resting facial tone.[55] For this reason, a hypoglossal-facial transfer may be employed to restore tone, and a masseteric nerve-facial nerve transfer may be used for smile rehabilitation. As with hypoglossal-facial transfer, masseteric nerve transfer requires a deliberate movement from the patient - biting down, in this case - to activate the facial muscles. Over time, many patients learn to activate their smiles without having to clench their jaws, but it is comparatively uncommon for adults to develop true spontaneity of the smile after a nerve transfer, with women being more likely to do so than men.[56] Unlike with the hypoglossal-facial nerve transfer, using the masseteric nerve results in comparatively little long-term morbidity; the ipsilateral masseter will usually atrophy to some extent, which may cause facial hollowing, but the muscle will often reinnervate over time and regain some of the lost bulk. Ultimately, the goal of surgical management for facial paralysis is to restore mimetic function with as natural an appearance as possible, and that is best accomplished by reinnervating the native musculature.
Temporal Bone Fracture
In cases of temporal bone fracture, the facial nerve generally suffers a blunt injury rather than a transection due to edema occurring within the tight confines of the bony Fallopian canal.[7] The most common area affected is the labyrinthine segment, most often the geniculate ganglion.[18] It can be challenging to differentiate a transection injury, which requires operative repair, from a blunt injury, such as intraneural hematoma, on CT scan; fortunately, blunt injuries account for 86% of facial paralyses in temporal bone fractures, and transection injuries only 14%.[19] As with penetrating or iatrogenic injuries, the severity of the paralysis and the timeline will determine the level of suspicion for a transection injury in patients with temporal bone fractures, but a unique aspect of temporal bone fractures is that operative intervention may also be helpful in certain patients who have not suffered nerve transection. Patients with House-Brackmann grade VI paralysis should undergo electroneuronography, waiting at least three days from the time of injury for Wallerian degeneration to occur, even if the paralysis is complete. If there is >90% amplitude degeneration of the paralyzed side compared to the unaffected side, facial nerve decompression should be performed.[18]
The intratemporal facial nerve can be accessed via transmastoid, and middle fossa craniotomy approaches if the hearing is intact or via a translabyrinthine craniotomy if the labyrinth was damaged by the fracture and profound hearing loss has occurred.[57] Should a transection injury be encountered during exposure of the facial nerve, an appropriate repair should be performed, either primarily or with an interposition graft, if necessary (see above). Because facial paralysis from temporal bone fractures may evolve slowly, patients should be followed closely until they meet decompression criteria (House-Brackmann grade VI paralysis and >90% amplitude loss on ENoG), clinical recovery becomes apparent, or two months from the time of injury elapse, after which decompression is unlikely to provide a benefit to the recovery process.[18] As with patients suffering facial paralysis from other traumatic etiologies, facial paralysis patients with temporal bone fractures should also receive high-dose oral steroids and be monitored for corneal complications, using eyedrops, ocular lubricant, and eyelid taping as necessary. In some cases, periocular surgical procedures may be required to protect the cornea adequately (see above).
Barotrauma
While barotrauma is an uncommon cause of facial paralysis, it is worth noting that adjunctive treatment along with high-dose oral steroids appears to be helpful. Adding decongestants and antihistamines, as might be recommended for symptomatic relief of an upper respiratory infection, can expedite the return of facial function, as can myringotomy with or without placement of a pressure equalization tube in the ipsilateral ear.[22] Essentially, the goal is to alleviate any acute Eustachian tube dysfunction and permit inflammation in the facial nerve to resolve.[21][23][24][23][21]
Chronic injury: >12 Months Since Injury
Beyond 12 months from paralysis onset, the likelihood of successfully reinnervating the mimetic muscles decreases substantially because they have most likely begun to atrophy and fibrose as a result of chronic denervation. Attempting to reinnervate them at this late stage runs a higher than usual risk of failure, particularly because after a nerve transfer or repair, axons would still have to grow to the neuromuscular junctions, which occurs at a rate of approximately 1 mm/day; thus, several additional weeks to months typically still pass after the reinnervation procedure has been performed but before the muscles can receive any neural input. If the patient and surgeon feel that reinnervation is the preferred option despite prolonged denervation of the mimetic muscles, for example, if the patient is a poor candidate for a muscle transfer, electromyography may be used to assess the status of the facial muscles. If fibrillation potentials are present, the muscles should be capable of receiving neural input, but electrical silence indicates non-viable muscles. Of course, demonstration of viable muscles on EMG does not guarantee that they will remain viable by the time axons from a reinnervation procedure are able to reach them, although some surgeons anecdotally report successful facial reinnervation performed up to 24 months after the onset of paralysis.
Facial reanimation may be offered for patients not deemed suitable candidates for reinnervation procedures. Reanimation techniques are classically categorized as either "static" or "dynamic," with dynamic reanimation procedures further divided into regional and free muscle transfer options. Most often, static reanimation techniques are applied in the middle and upper thirds of the face, while dynamic techniques are utilized to rehabilitate the smile.
Upper Face
Reanimation of the forehead and periocular region is often performed using "static" techniques that do not in and of themselves provide movement to the face but still improve symmetry and function. Restoring symmetric brow elevation by transferring functional muscle would be challenging because the muscle bulk required would be very noticeable lying between the skin and soft tissue of the forehead and the frontal bone. For this reason, conventional brow lifting is often employed, commonly using direct, endoscopic, or suture suspension techniques.[58] Endoscopic lifting minimizes the visible scarring but provides a less effective elevation of the brow (approximately 2 mm), particularly when performed unilaterally or asymmetrically, and is therefore only applicable when there is minimal brow ptosis, or the patient is reluctant to undergo a procedure that will produce a noticeable scar.[59]
Direct brow lifting, on the other hand, provides more elevation and more control over brow shape and position, but at the cost of a scar along the superior edge of the eyebrow. For patients who need significant brow elevation but will not tolerate a scar, suture suspension brow lifting is a compromise between the endoscopic and direct techniques, but it tends to peak the eyebrow and also causes dimpling where the sutures are passed through the dermis of the brow. Regardless of the technique chosen, the goal for the brow lift should be for the paralyzed brow to sit at a height midway between the non-paralyzed brow's resting position and its elevated position, which are typically approximately 10 to 15 mm apart. The average lay bystander will notice a difference in brow height of >4 mm; therefore, placing the paralyzed brow at the halfway position between resting and elevation minimizes apparent asymmetry.[60]
Similar to reanimation of the brow, reanimation of the eyelid is generally undertaken using static techniques. Common options include scleral contact lens placement, eyelid weight placement, tarsorrhaphy, tarsoconjunctival flap transfer, and lateral tarsal strip canthopexy. The primary goal of periocular reanimation should be to protect the cornea, and the secondary goal is to produce a synchronous blink. Contact lenses can be very effective for maintaining a healthy corneal environment in patients who tolerate long-term wear, although these lenses do nothing to facilitate eyelid closure per se.[36]
Weight placement onto or into the upper eyelid via either adhesive or a surgical procedure both protects the cornea and improves actual eyelid closure, although the closure produced is slower than that on the unaffected side and often less complete. The other drawback to eyelid weight placement is the necessary compromise between the completeness of the closure produced and the degree of blepharoptosis present at rest. Patients who complain that wind, rain, shampoo, etc., still get into their eyes after weight placement may request larger weights be implanted, but this will usually result in noticeable depression of the upper eyelid margin and potential visual field obstruction. Various sizes of weights may be applied with adhesive to determine which is the ideal one for a given patient, and this may be used to inform implantable weight selection if the patient and surgeon feel that an operation is appropriate to improve patient convenience or because a prolonged recovery is anticipated. Most patients will do well with a 1.2 gram weight. The weight may be surgically placed in a pretarsal position, roughly 2 mm above and parallel to the lash line, or in a post-septal position, located more superiorly in the upper eyelid. In either case, the weight is centered over the point of maximum lagophthalmos, which is typically between the mid-pupil and the medial limbus. A post-septal location for the weight permits it to be covered by preaponeurotic orbital fat, thereby concealing it and improving cosmesis; the disadvantage to weight placement in this position is that the weight has less of a mechanical advantage than a weight in the pretarsal position and therefore a heavier weight is typically required. The post-septal position also means that the weight is likely to pull the eyelid open when the patient is supine, which may necessitate taping the eye closed at night - this is not a problem encountered with pretarsal weight placement.[61]
Pretarsal weight placement, however, does come with its own disadvantages: the skin in the pretarsal eyelid is the thinnest in the human body, and the contour of the weight is often visible underneath it. Placement of weight in this location is also liable to cause astigmatism due to pressure on the globe, but this can be mitigated with the use of an articulated chain instead of a solid plate.[62] Again, however, a compromise is necessary because using a chain increases the risk of extrusion over solid weight. Lastly, the choice of weight material should be considered: most eyelid weights are made of gold, which has a low tissue reactivity rate (around 5%), but platinum has an even lower rate of tissue reactions and allergies (about 1%) as well as a slightly higher density, making the size of the implant for a given mass even smaller and less visible.[37] Regardless of material or articulation, eyelid weights are easily removed in the event the patient recovers spontaneous eye closure.
When lower eyelid laxity contributes substantially to a patient's paralytic lagophthalmos, tightening the lower eyelid should be considered. The most common way of accomplishing this is to perform a lateral tarsal strip canthopexy, which removes a portion of the lateral lower eyelid margin and fixates the lateral aspect of the inferior tarsal plate to the lateral orbital wall periosteum posterior and superior to Whitnall's tubercle, thus tightening and elevating the lower eyelid. In some cases, elevation of the medial canthus is also required, particularly in patients with heavy soft tissues and a prior failed lateral canthopexy. Medial canthopexy can be achieved with the placement of a small screw into the maxilla, which is then used to anchor a suture suspension of the medial aspect of the inferior tarsal plate or even a suspension of the entire margin of the lower eyelid using fascia lata or a ribbon of expanded polytetrafluoroethylene (ePTFE).[62]
When operating in the medial canthal area, avoiding injuring the lacrimal system is critical. Lateral tarsal strip canthopexy is often accompanied by a tarsorrhaphy to decrease the resting height of the palpebral fissure and thereby improve the corneal environment, but many patients, particularly younger ones concerned with cosmesis, will not tolerate tarsorrhaphy, whether temporary or permanent. Despite its effectiveness, the end result is often considered unsightly, regardless of whether the tarsorrhaphy is surgical or simply a horizontal mattress suture passed over bolsters. An alternative to tarsorrhaphy is the tarsoconjunctival flap, in which a 3 to 6 mm wide and roughly 4 to 5 mm high inferiorly-based flap of composite tarsal plate and conjunctiva are transferred from the upper eyelid to the lower, thereby coupling the two eyelids without suturing the lid margins together in an aesthetically displeasing manner.[63]
These techniques are all capable of providing corneal protection if applied appropriately, but restoration of a synchronous blink is extremely challenging due to the speed of normal eyelid closure because the orbicularis oculi are the fastest muscles in the human body. When the muscle is transferred from a remote site to assist with eyelid closure, the result is not ordinarily worth the effort due to the unnatural appearance of the transferred muscle's bulk and the persistent dyssynchrony of eye closure.[64] Some authors, however, have reported tunneling a portion of the orbicularis oculi of the non-paralyzed side across the nasal root as a pedicled flap, demonstrating improved blinking and corneal health as a result.[65][66]
Mid and Lower Face
The main goals of reanimation in the mid and lower faces are to reduce nasal obstruction, improve nasolabial fold symmetry, and rehabilitate the smile. Static reanimation techniques can be applied to any of these objectives, although smile rehabilitation is most often undertaken with muscle transfer when possible. Alleviation of nasal obstruction in facial paralysis centers on restoring nasal valve patency, which is compromised due to weakness of the nasalis muscle. While support of the nasal valve may be accomplished using rhinoplasty techniques, such as spreader and alar batten grafting, valve suspension is more often performed using a technique in which a screw is placed into the maxilla. A suspension suture is anchored to it, or a ribbon of fascia lata or ePTFE is used to suspend the lateral aspect of the nasal ala to the temporalis fascia.[67][35]
Placement of fascia lata or ePFTE is performed via a preauricular incision, which can also be used to place material to suspend the nasolabial fold and/or the oral commissure. Static oral commissure suspension may be the best smile rehabilitation option for patients who are not healthy enough to tolerate lengthy dynamic reanimation procedures or who do not wish to undergo them for other reasons. Suspension of the nasolabial fold, on the other hand, is often performed in conjunction with dynamic smile reanimation to improve the overall symmetry of the face; an oral commissure that moves normally when smiling but that has an effaced, or otherwise asymmetric nasolabial fold above it does not provide the overall natural appearance that patients and surgeons are trying to achieve. Nasolabial fold suspension may be performed using an additional incision through the nasolabial fold itself or, more commonly, using a percutaneous approach in which needles are passed through stab incisions just inferior to the fold in order to anchor a sheet of fascia lata.[68] Similarly, a strip of fascia or ePTFE may be fixated to the modiolus of the oral commissure via an incision in the corner of the mouth or internally under direct visualization through the preauricular incision. However, fascia grafting or alloplast implantation are not always required for oral commissure repositioning and nasal valve opening. The modiolar rotational cheiloplasty is a simple procedure in which an incision along the nasolabial fold and around the inferior aspect of the nasal ala permits superior advancement of the oral commissure along with the lateral expansion of the nasal vestibule.[69]
While elevating the oral commissure to correct a droop in the corner of the mouth can be readily accomplished with static reanimation techniques, providing movement to the smile on the paralyzed side of the face is substantially more complicated and requires precise control over the vector and amount of oral commissure movement as well as its timing. Providing dynamic reanimation of the smile can be performed using either regional or free muscle transfer, with the most common muscles used being the temporalis (as a regional flap) and the gracilis (as a free muscle flap). Regional muscle transfer is less time-consuming than free muscle transfer as well as technically easier to perform. The primary drawback to regional muscle transfer is that the amount of oral commissure movement produced is not as great as that achieved with free muscle transfer, and there are limitations to the vectors of movement that can be obtained as well. As an example, masseteric muscle transfer is a technique that involves releasing a portion of the inferior aspect of the masseter and suturing it to the oral commissure so that a smile would occur when the patient clenches the jaw; because the origin of the masseter muscle is the zygomatic arch, which lies too far posteriorly to the oral commissure, the vector of movement during the smile produced by masseter muscle transfer is much more lateral than that of a natural smile and the technique has been largely abandoned, except some surgeons who reposition the origin of the muscle more anteriorly to achieve the correct vector.[70]
Moving the whole masseter muscle, however, redistributes facial volume, leading to additional noticeable asymmetries. For this reason, the temporalis muscle is employed far more frequently for facial reanimation than the masseter muscle, given that its origin is located more superiorly and it provides a more natural vector for oral commissure movement than the masseter does. The temporalis may be transferred in two different ways: detaching the central portion of the muscle from the skull and reflecting it downward over the zygomatic arch to insert into the oral commissure (temporalis flap transfer) and detaching the insertion of the muscle from the mandible and advancing it to the oral commissure (temporalis tendon transfer). The former method tends to leave a large depression in the side of the head, which can be mitigated somewhat by filling the defect with temporoparietal fascia, but the volume of the muscle lying superficial to the zygomatic arch looks noticeably unnatural regardless.[71] Temporalis tendon transfer has largely supplanted temporalis flap transfer because it can be performed intraorally, thereby avoiding external scars, and it does not transfer facial volume unnaturally.[72]
The drawback of temporalis transfer remains the amount of oral commissure excursion, however, which is only around 5 mm as compared to the 7 to 9 mm obtained with gracilis-free muscle transfer.[73] Other regional muscles transfers that are occasionally performed for smile rehabilitation are the anterior digastric and platysmal transfers, which are used to improve lower lip depression in the case of marginal mandibular nerve injury; the platysmal transfer, of course, is not an available option in the case of a hemifacial paralysis.[74][75]
The gold standard for smile rehabilitation in patients who can tolerate lengthy surgical procedures is free muscle transfer because it has the advantages of precise control of movement vectors, or vectors, as well as greater excursion, albeit at the expense of a more challenging operation. Multiple muscles are available to transfer, including the pectoralis minor, latissimus dorsi, gracilis, serratus anterior, and strap muscles, none of which seem to produce any long-term donor site morbidity. The pectoralis minor and latissimus dorsi muscles are often second-line choices because of unreliable venous outflow and excessive bulk, respectively. Gracilis muscle transfer is widely considered to be the option of choice because of the consistent anatomy of its vascular pedicle and the reliability of its outcomes.[76][49]
The gracilis is a large postural muscle of the medial thigh, located between the adductor magnus and the adductor longus, extending from the medial aspect of the pubic and ischial rami to the pes anserinus at the medial tibial epicondyle. It is perfused by the adductor artery, which branches from the deep femoral artery and is drained by the adductor artery's two venae comitantes. The motor control to the gracilis comes from the obturator nerve, which arrives at the muscle at the same point and courses in the same plane as the vascular pedicle (between the adductors longus and magnus) but takes a different course because it originates more superiorly, from the lumbar plexus. Typically, a 20-50 gram section of gracilis muscle is harvested, with its neurovascular pedicle, and then inset into the face between the temporalis fascia and the modiolus of the oral commissure, with the adductor artery anastomosed to the facial artery and the dominant vena comitans anastomosed to the facial vein.[77]
In some situations, with awkward flap geometry, the superficial temporal vessels may be used instead of the facial artery and vein. The obturator nerve is then coapted to a donor nerve via primary neurorrhaphy, and the choice of nerve will determine the characteristics of the flap's movement. The original description of the microneurovascular gracilis transfer by Harii in 1976 reported the use of the deep temporal nerves, but these are challenging to locate and use; in some cases, the residual facial nerve trunk may be employed, but the basal firing rate in this nerve is so high that it tends to cause excessive contracture of the muscle over time.[76] Currently, the most commonly employed donor nerves are the masseteric nerve and the cross-face nerve graft. Paralleling the foregoing discussion regarding nerve transfer, the use of the masseteric nerve to control a free gracilis muscle transfer provides more reliability (94%) and excursion (8.7 mm) than the use of a cross-face nerve graft (84% and 6.5 mm), although the cross face nerve graft provides a spontaneous smile without the need to clench the jaw as well as better smile symmetry.[49] The other drawback to cross-face grafting is that it requires a two-stage procedure, with the cross-face graft placement (including sural nerve harvest and facial nerve exploration on the non-paralyzed side) preceding the gracilis transfer by 6 to 9 months.
Gracilis free muscle transfer, therefore, is a reliable means of restoring a meaningful smile to a paralyzed hemiface, but the technique remains imperfect due to a number of compromises: 1) the large bulk of gracilis muscle, when transferred to the face, appears unnatural; 2) the single movement vector of a gracilis inset between the temporalis fascia and the oral commissure produces a tight-lipped Mona Lisa smile rather than a genuine Duchenne smile with visible dentition; 3) the surgeon must choose between a reliable donor nerve (the masseteric nerve) and a nerve that can provide spontaneity (a cross-face nerve graft); 4) the gracilis is a postural muscle, which makes it incapable of contracting as rapidly as a native zygomaticus major muscle, thereby introducing dyssynchrony or requiring the patient to smile more slowly to maintain symmetry during motion. The problem of bulk can be solved by thinning the flap, but this introduces a greater risk of devascularization and denervation of the muscle; thinning the muscle also removes the fascia, which in turn increases the risk of the flap tethering to the overlying dermis and creating unnatural dimples during smiling. If the flap is not debulked, it can potentially be split into two or more slips of muscle that share the same neurovascular supply in order to replace not just the zygomaticus major but also the levator labii superioris and other muscles that provide a more natural smile, but doing so then introduces a large amount of excess bulk, and the smile remains somewhat slow.[78]
Some authors report coapting both a cross-face nerve graft and the masseteric nerve into the obturator nerve to combine the advantages of both donor nerves, reliability, and spontaneity, but results remain somewhat variable, perhaps because axons from both nerves must compete for space within the single obturator nerve.[79][49]
One approach to mitigating these problems is using the serratus anterior muscle, which is harvested as several anatomically separate muscle slips, all perfused by the thoracodorsal artery and controlled by the long thoracic nerve, that permits more precise control of the vector of smile movement.[80] The serratus anterior may provide more rapid contraction than the gracilis, but the volume of the muscle slips remains substantial, and the problem of donor nerve selection persists. A more promising option may be the use of cervical strap muscles, the first description of which was Alam's report of sternohyoid muscle transfer in 2016 that emphasized not only the strap muscle's minimal bulk but also its histological similarity to mimetic muscles, giving it the ability to contract rapidly and maintain a temporally synchronous smile.[81]
One step further is the dual-vector, combined sternohyoid and omohyoid flap described by Vincent et al. in 2020, which uses both muscles to provide rapid superolateral excursion to the oral commissure as well as elevation of the upper lip to produce a natural dental show. The strap muscles combined weigh only 10 to 15 grams and introduce minimal additional bulk. Both muscles are perfused by the superior thyroid artery, which is easily coapted to the facial or superficial temporal artery, although the flap's venous outflow, like that of the pectoralis minor, is somewhat variable. The greatest advantage to strap muscle transfer, however, may be that the flap's motor control comes via the ansa cervicalis, which has two ends, thereby facilitating dual innervation by the masseteric nerve and a cross-face nerve graft. While the technique remains in its infancy, it appears very promising with respect to ameliorating the compromises inherent to gracilis muscle transfer.[82] Regardless of the muscle transfer procedure ultimately employed, whether regional or free, physical therapy will be required to help the patient maximize the use of the new muscle or muscles.
Synkinesis/Non-Flaccid Facial Paralysis
The foregoing discussion has focused on the management of complete transection injuries and rehabilitation of the flaccidly paralyzed face; however, many, if not the majority, of facial nerve trauma patients ultimately recover function that is dyscoordinated and spastic. While this synkinesis is not as noticeable at rest as complete, flaccid hemifacial paralysis is, it can be near as disfiguring with movement and just as disruptive to patients' quality of life. Management of post-paralytic facial dysfunction varies greatly from flaccid paralysis, although some techniques may be applied to both conditions. Patients with synkinesis often complain of involuntary movements that accompany voluntary ones and tightness and soreness that tend to progress throughout the day. Common involuntary movements include eye closure, corner-of-mouth lateralization, chin dimpling, and platysmal contraction. These same areas are often sites of chronic pain, particularly the midface and neck. In addition to these involuntary movements, patients will frequently note the inability to control their muscles appropriately, persistently complaining of incomplete eye closure and an asymmetric smile, for example. In most cases, the first line treatment for synkinesis should be physical therapy, which consists of exercises that patients perform before a mirror to improve coordination and symmetry, as well as stretches and massages to reduce discomfort.[83] Once physical therapy has been initiated, additional relaxation of the hyperactive facial muscles can be achieved with chemodenervation, typically with botulinum toxin.[84]
Regular injections of comparatively high doses may be required, which risks developing antibodies and reducing the injections' effectiveness. In order to mitigate this problem, patients may need to switch isoforms over time, such as shifting from onabotulinumtoxin to abobotulinumtoxin, or similar. Most patients will be satisfied with improvements made between physical therapy and chemodenervation, but for some, operative intervention may be indicated, typically in patients who do not wish to continue with botulinum toxin injections or for whom regular visits for healthcare are impractical. Surgical options involve division of nerve branches that cause synkinetic movements (selective neurectomy, particularly around the eye, lower face, and neck), removal of muscles causing synkinetic movements (myomectomy, typically the platysma and lower lip depressors), and occasionally reinnervation (such as masseteric nerve transfer) or reanimation of the smile (with free muscle transfer, such as gracilis or sterno-omohyoid), any of which may be combined in the same procedure as necessary.[85][86][87][88][55][89][90] Ultimately, however, non-flaccid facial paralysis is a very challenging problem to solve, as even surgical interventions may provide only temporary relief, given that facial nerve axons tend to regenerate and synkinesis will often recur with time.