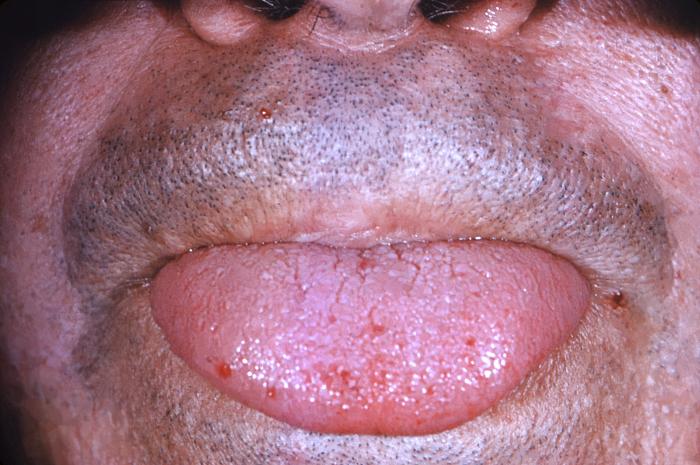
[1]
Kritharis A, Al-Samkari H, Kuter DJ. Hereditary hemorrhagic telangiectasia: diagnosis and management from the hematologist's perspective. Haematologica. 2018 Sep:103(9):1433-1443. doi: 10.3324/haematol.2018.193003. Epub 2018 May 24
[PubMed PMID: 29794143]
Level 3 (low-level) evidence
[2]
Halderman AA, Ryan MW, Clark C, Sindwani R, Reh DD, Poetker DM, Invernizzi R, Marple BF. Medical treatment of epistaxis in hereditary hemorrhagic telangiectasia: an evidence-based review. International forum of allergy & rhinology. 2018 Jun:8(6):713-728. doi: 10.1002/alr.22094. Epub 2018 Feb 2
[PubMed PMID: 29393992]
[3]
Jackson SB, Villano NP, Benhammou JN, Lewis M, Pisegna JR, Padua D. Gastrointestinal Manifestations of Hereditary Hemorrhagic Telangiectasia (HHT): A Systematic Review of the Literature. Digestive diseases and sciences. 2017 Oct:62(10):2623-2630. doi: 10.1007/s10620-017-4719-3. Epub 2017 Aug 23
[PubMed PMID: 28836046]
Level 1 (high-level) evidence
[4]
Morgan T,McDonald J,Anderson C,Ismail M,Miller F,Mao R,Madan A,Barnes P,Hudgins L,Manning M, Intracranial hemorrhage in infants and children with hereditary hemorrhagic telangiectasia (Osler-Weber-Rendu syndrome). Pediatrics. 2002 Jan;
[PubMed PMID: 11773580]
[5]
Shovlin CL, Guttmacher AE, Buscarini E, Faughnan ME, Hyland RH, Westermann CJ, Kjeldsen AD, Plauchu H. Diagnostic criteria for hereditary hemorrhagic telangiectasia (Rendu-Osler-Weber syndrome). American journal of medical genetics. 2000 Mar 6:91(1):66-7
[PubMed PMID: 10751092]
[6]
Govani FS, Shovlin CL. Hereditary haemorrhagic telangiectasia: a clinical and scientific review. European journal of human genetics : EJHG. 2009 Jul:17(7):860-71. doi: 10.1038/ejhg.2009.35. Epub 2009 Apr 1
[PubMed PMID: 19337313]
[7]
Plauchu H, de Chadarévian JP, Bideau A, Robert JM. Age-related clinical profile of hereditary hemorrhagic telangiectasia in an epidemiologically recruited population. American journal of medical genetics. 1989 Mar:32(3):291-7
[PubMed PMID: 2729347]
Level 2 (mid-level) evidence
[8]
McDonald J,Wooderchak-Donahue W,VanSant Webb C,Whitehead K,Stevenson DA,Bayrak-Toydemir P, Hereditary hemorrhagic telangiectasia: genetics and molecular diagnostics in a new era. Frontiers in genetics. 2015;
[PubMed PMID: 25674101]
[9]
Sautter NB, Smith TL. Treatment of Hereditary Hemorrhagic Telangiectasia-Related Epistaxis. Otolaryngologic clinics of North America. 2016 Jun:49(3):639-54. doi: 10.1016/j.otc.2016.02.010. Epub
[PubMed PMID: 27267016]
[10]
Lesca G, Burnichon N, Raux G, Tosi M, Pinson S, Marion MJ, Babin E, Gilbert-Dussardier B, Rivière S, Goizet C, Faivre L, Plauchu H, Frébourg T, Calender A, Giraud S, French Rendu-Osler Network. Distribution of ENG and ACVRL1 (ALK1) mutations in French HHT patients. Human mutation. 2006 Jun:27(6):598
[PubMed PMID: 16705692]
[11]
Lesca G, Genin E, Blachier C, Olivieri C, Coulet F, Brunet G, Dupuis-Girod S, Buscarini E, Soubrier F, Calender A, Danesino C, Giraud S, Plauchu H, French-Italian HHT Network. Hereditary hemorrhagic telangiectasia: evidence for regional founder effects of ACVRL1 mutations in French and Italian patients. European journal of human genetics : EJHG. 2008 Jun:16(6):742-9. doi: 10.1038/ejhg.2008.3. Epub 2008 Feb 20
[PubMed PMID: 18285823]
[12]
McAllister KA, Grogg KM, Johnson DW, Gallione CJ, Baldwin MA, Jackson CE, Helmbold EA, Markel DS, McKinnon WC, Murrell J. Endoglin, a TGF-beta binding protein of endothelial cells, is the gene for hereditary haemorrhagic telangiectasia type 1. Nature genetics. 1994 Dec:8(4):345-51
[PubMed PMID: 7894484]
[13]
Johnson DW, Berg JN, Baldwin MA, Gallione CJ, Marondel I, Yoon SJ, Stenzel TT, Speer M, Pericak-Vance MA, Diamond A, Guttmacher AE, Jackson CE, Attisano L, Kucherlapati R, Porteous ME, Marchuk DA. Mutations in the activin receptor-like kinase 1 gene in hereditary haemorrhagic telangiectasia type 2. Nature genetics. 1996 Jun:13(2):189-95
[PubMed PMID: 8640225]
[14]
Azuma H. Genetic and molecular pathogenesis of hereditary hemorrhagic telangiectasia. The journal of medical investigation : JMI. 2000 Aug:47(3-4):81-90
[PubMed PMID: 11019486]
[15]
Gallione CJ, Repetto GM, Legius E, Rustgi AK, Schelley SL, Tejpar S, Mitchell G, Drouin E, Westermann CJ, Marchuk DA. A combined syndrome of juvenile polyposis and hereditary haemorrhagic telangiectasia associated with mutations in MADH4 (SMAD4). Lancet (London, England). 2004 Mar 13:363(9412):852-9
[PubMed PMID: 15031030]
[16]
Donaldson JW, McKeever TM, Hall IP, Hubbard RB, Fogarty AW. The UK prevalence of hereditary haemorrhagic telangiectasia and its association with sex, socioeconomic status and region of residence: a population-based study. Thorax. 2014 Feb:69(2):161-7. doi: 10.1136/thoraxjnl-2013-203720. Epub 2013 Nov 4
[PubMed PMID: 24188926]
[17]
Donaldson JW, McKeever TM, Hall IP, Hubbard RB, Fogarty AW. Complications and mortality in hereditary hemorrhagic telangiectasia: A population-based study. Neurology. 2015 May 5:84(18):1886-93. doi: 10.1212/WNL.0000000000001538. Epub 2015 Apr 10
[PubMed PMID: 25862798]
[18]
Fernández-L A, Sanz-Rodriguez F, Blanco FJ, Bernabéu C, Botella LM. Hereditary hemorrhagic telangiectasia, a vascular dysplasia affecting the TGF-beta signaling pathway. Clinical medicine & research. 2006 Mar:4(1):66-78
[PubMed PMID: 16595794]
[19]
Grigg C, Anderson D, Earnshaw J. Diagnosis and Treatment of Hereditary Hemorrhagic Telangiectasia. Ochsner journal. 2017 Summer:17(2):157-161
[PubMed PMID: 28638289]
[20]
Dupuis-Girod S, Bailly S, Plauchu H. Hereditary hemorrhagic telangiectasia: from molecular biology to patient care. Journal of thrombosis and haemostasis : JTH. 2010 Jul:8(7):1447-56. doi: 10.1111/j.1538-7836.2010.03860.x. Epub 2010 Mar 19
[PubMed PMID: 20345718]
[21]
Duncan BW, Kneebone JM, Chi EY, Hraska V, Isik FF, Rosenthal GL, Jones TK, Starnes SL, Lupinetti FM. A detailed histologic analysis of pulmonary arteriovenous malformations in children with cyanotic congenital heart disease. The Journal of thoracic and cardiovascular surgery. 1999 May:117(5):931-8
[PubMed PMID: 10220688]
[22]
Faughnan ME, Mager JJ, Hetts SW, Palda VA, Lang-Robertson K, Buscarini E, Deslandres E, Kasthuri RS, Lausman A, Poetker D, Ratjen F, Chesnutt MS, Clancy M, Whitehead KJ, Al-Samkari H, Chakinala M, Conrad M, Cortes D, Crocione C, Darling J, de Gussem E, Derksen C, Dupuis-Girod S, Foy P, Geisthoff U, Gossage JR, Hammill A, Heimdal K, Henderson K, Iyer VN, Kjeldsen AD, Komiyama M, Korenblatt K, McDonald J, McMahon J, McWilliams J, Meek ME, Mei-Zahav M, Olitsky S, Palmer S, Pantalone R, Piccirillo JF, Plahn B, Porteous MEM, Post MC, Radovanovic I, Rochon PJ, Rodriguez-Lopez J, Sabba C, Serra M, Shovlin C, Sprecher D, White AJ, Winship I, Zarrabeitia R. Second International Guidelines for the Diagnosis and Management of Hereditary Hemorrhagic Telangiectasia. Annals of internal medicine. 2020 Dec 15:173(12):989-1001. doi: 10.7326/M20-1443. Epub 2020 Sep 8
[PubMed PMID: 32894695]
[23]
AAssar OS, Friedman CM, White RI Jr. The natural history of epistaxis in hereditary hemorrhagic telangiectasia. The Laryngoscope. 1991 Sep:101(9):977-80
[PubMed PMID: 1886446]
[24]
Shovlin CL, Jackson JE, Bamford KB, Jenkins IH, Benjamin AR, Ramadan H, Kulinskaya E. Primary determinants of ischaemic stroke/brain abscess risks are independent of severity of pulmonary arteriovenous malformations in hereditary haemorrhagic telangiectasia. Thorax. 2008 Mar:63(3):259-66
[PubMed PMID: 17981912]
[25]
Garg N, Khunger M, Gupta A, Kumar N. Optimal management of hereditary hemorrhagic telangiectasia. Journal of blood medicine. 2014:5():191-206. doi: 10.2147/JBM.S45295. Epub 2014 Oct 15
[PubMed PMID: 25342923]
[26]
Shovlin CL, Hughes JM, Tuddenham EG, Temperley I, Perembelon YF, Scott J, Seidman CE, Seidman JG. A gene for hereditary haemorrhagic telangiectasia maps to chromosome 9q3. Nature genetics. 1994 Feb:6(2):205-9
[PubMed PMID: 8162076]
[27]
Pahl KS, Choudhury A, Wusik K, Hammill A, White A, Henderson K, Pollak J, Kasthuri RS. Applicability of the Curaçao Criteria for the Diagnosis of Hereditary Hemorrhagic Telangiectasia in the Pediatric Population. The Journal of pediatrics. 2018 Jun:197():207-213. doi: 10.1016/j.jpeds.2018.01.079. Epub 2018 Apr 11
[PubMed PMID: 29655863]
[28]
Bharatha A, Faughnan ME, Kim H, Pourmohamad T, Krings T, Bayrak-Toydemir P, Pawlikowska L, McCulloch CE, Lawton MT, Dowd CF, Young WL, Terbrugge KG. Brain arteriovenous malformation multiplicity predicts the diagnosis of hereditary hemorrhagic telangiectasia: quantitative assessment. Stroke. 2012 Jan:43(1):72-8. doi: 10.1161/STROKEAHA.111.629865. Epub 2011 Oct 27
[PubMed PMID: 22034007]
[29]
Shovlin CL, Sodhi V, McCarthy A, Lasjaunias P, Jackson JE, Sheppard MN. Estimates of maternal risks of pregnancy for women with hereditary haemorrhagic telangiectasia (Osler-Weber-Rendu syndrome): suggested approach for obstetric services. BJOG : an international journal of obstetrics and gynaecology. 2008 Aug:115(9):1108-15. doi: 10.1111/j.1471-0528.2008.01786.x. Epub 2008 May 30
[PubMed PMID: 18518871]
[30]
Tunkel DE, Anne S, Payne SC, Ishman SL, Rosenfeld RM, Abramson PJ, Alikhaani JD, Benoit MM, Bercovitz RS, Brown MD, Chernobilsky B, Feldstein DA, Hackell JM, Holbrook EH, Holdsworth SM, Lin KW, Lind MM, Poetker DM, Riley CA, Schneider JS, Seidman MD, Vadlamudi V, Valdez TA, Nnacheta LC, Monjur TM. Clinical Practice Guideline: Nosebleed (Epistaxis). Otolaryngology--head and neck surgery : official journal of American Academy of Otolaryngology-Head and Neck Surgery. 2020 Jan:162(1_suppl):S1-S38. doi: 10.1177/0194599819890327. Epub
[PubMed PMID: 31910111]
Level 1 (high-level) evidence
[31]
Yaniv E, Preis M, Shevro J, Nageris B, Hadar T. Anti-estrogen therapy for hereditary hemorrhagic telangiectasia - a long-term clinical trial. Rhinology. 2011 Jun:49(2):214-6. doi: 10.4193/Rhino09.201. Epub
[PubMed PMID: 21743879]
[32]
Al-Samkari H. Hereditary hemorrhagic telangiectasia: systemic therapies, guidelines, and an evolving standard of care. Blood. 2021 Feb 18:137(7):888-895. doi: 10.1182/blood.2020008739. Epub
[PubMed PMID: 33171488]
[33]
Gaillard S, Dupuis-Girod S, Boutitie F, Rivière S, Morinière S, Hatron PY, Manfredi G, Kaminsky P, Capitaine AL, Roy P, Gueyffier F, Plauchu H, ATERO Study Group. Tranexamic acid for epistaxis in hereditary hemorrhagic telangiectasia patients: a European cross-over controlled trial in a rare disease. Journal of thrombosis and haemostasis : JTH. 2014 Sep:12(9):1494-502. doi: 10.1111/jth.12654. Epub 2014 Jul 29
[PubMed PMID: 25040799]
[34]
Geisthoff UW, Seyfert UT, Kübler M, Bieg B, Plinkert PK, König J. Treatment of epistaxis in hereditary hemorrhagic telangiectasia with tranexamic acid - a double-blind placebo-controlled cross-over phase IIIB study. Thrombosis research. 2014 Sep:134(3):565-71. doi: 10.1016/j.thromres.2014.06.012. Epub 2014 Jun 16
[PubMed PMID: 25005464]
Level 1 (high-level) evidence
[35]
Boyer H, Fernandes P, Le C, Yueh B. Prospective randomized trial of sclerotherapy vs standard treatment for epistaxis due to hereditary hemorrhagic telangiectasia. International forum of allergy & rhinology. 2015 May:5(5):435-40. doi: 10.1002/alr.21484. Epub 2015 Feb 2
[PubMed PMID: 25643928]
Level 1 (high-level) evidence
[36]
Kuan EC, Peng KA, Thompson CF, Suh JD, Wang MB. Sinonasal quality of life outcomes following laser treatment of epistaxis related to hereditary hemorrhagic telangiectasia. Lasers in medical science. 2017 Apr:32(3):527-531. doi: 10.1007/s10103-017-2144-7. Epub 2017 Jan 24
[PubMed PMID: 28116537]
Level 2 (mid-level) evidence
[37]
Lund VJ, Darby Y, Rimmer J, Amin M, Husain S. Nasal closure for severe hereditary haemorrhagic telangiectasia in 100 patients. The Lund modification of the Young's procedure: a 22-year experience. Rhinology. 2017 Jun 1:55(2):135-141. doi: 10.4193/Rhin16.315. Epub
[PubMed PMID: 28064338]
[38]
Peyrin-Biroulet L, Williet N, Cacoub P. Guidelines on the diagnosis and treatment of iron deficiency across indications: a systematic review. The American journal of clinical nutrition. 2015 Dec:102(6):1585-94. doi: 10.3945/ajcn.114.103366. Epub 2015 Nov 11
[PubMed PMID: 26561626]
Level 1 (high-level) evidence
[39]
Edwards CP, Shehata N, Faughnan ME. Hereditary hemorrhagic telangiectasia patients can tolerate anticoagulation. Annals of hematology. 2012 Dec:91(12):1959-68. doi: 10.1007/s00277-012-1553-8. Epub 2012 Sep 30
[PubMed PMID: 23053175]
[40]
Shovlin CL, Millar CM, Droege F, Kjeldsen A, Manfredi G, Suppressa P, Ugolini S, Coote N, Fialla AD, Geisthoff U, Lenato GM, Mager HJ, Pagella F, Post MC, Sabbà C, Sure U, Torring PM, Dupuis-Girod S, Buscarini E, VASCERN-HHT. Safety of direct oral anticoagulants in patients with hereditary hemorrhagic telangiectasia. Orphanet journal of rare diseases. 2019 Aug 28:14(1):210. doi: 10.1186/s13023-019-1179-1. Epub 2019 Aug 28
[PubMed PMID: 31462308]
[41]
Faughnan ME, Palda VA, Garcia-Tsao G, Geisthoff UW, McDonald J, Proctor DD, Spears J, Brown DH, Buscarini E, Chesnutt MS, Cottin V, Ganguly A, Gossage JR, Guttmacher AE, Hyland RH, Kennedy SJ, Korzenik J, Mager JJ, Ozanne AP, Piccirillo JF, Picus D, Plauchu H, Porteous ME, Pyeritz RE, Ross DA, Sabba C, Swanson K, Terry P, Wallace MC, Westermann CJ, White RI, Young LH, Zarrabeitia R, HHT Foundation International - Guidelines Working Group. International guidelines for the diagnosis and management of hereditary haemorrhagic telangiectasia. Journal of medical genetics. 2011 Feb:48(2):73-87. doi: 10.1136/jmg.2009.069013. Epub 2009 Jun 23
[PubMed PMID: 19553198]
[42]
Shovlin C, Bamford K, Wray D. Post-NICE 2008: Antibiotic prophylaxis prior to dental procedures for patients with pulmonary arteriovenous malformations (PAVMs) and hereditary haemorrhagic telangiectasia. British dental journal. 2008 Nov 22:205(10):531-3. doi: 10.1038/sj.bdj.2008.978. Epub
[PubMed PMID: 19023305]
[43]
Nam TK, Park YS, Kwon JT. Brain Abscesses Associated with Asymptomatic Pulmonary Arteriovenous Fistulas. Journal of Korean Neurosurgical Society. 2017 Jan 1:60(1):118-124. doi: 10.3340/jkns.2015.0707.023. Epub 2016 Dec 29
[PubMed PMID: 28061502]
[44]
Lerut J, Orlando G, Adam R, Sabbà C, Pfitzmann R, Klempnauer J, Belghiti J, Pirenne J, Thevenot T, Hillert C, Brown CM, Gonze D, Karam V, Boillot O, European Liver Transplant Association. Liver transplantation for hereditary hemorrhagic telangiectasia: Report of the European liver transplant registry. Annals of surgery. 2006 Dec:244(6):854-62; discussion 862-4
[PubMed PMID: 17122610]
[45]
Yang W, Liu A, Hung AL, Braileanu M, Wang JY, Caplan JM, Colby GP, Coon AL, Tamargo RJ, Ahn ES, Huang J. Lower Risk of Intracranial Arteriovenous Malformation Hemorrhage in Patients With Hereditary Hemorrhagic Telangiectasia. Neurosurgery. 2016 May:78(5):684-93. doi: 10.1227/NEU.0000000000001103. Epub
[PubMed PMID: 26540357]
[46]
Mohr JP, Parides MK, Stapf C, Moquete E, Moy CS, Overbey JR, Al-Shahi Salman R, Vicaut E, Young WL, Houdart E, Cordonnier C, Stefani MA, Hartmann A, von Kummer R, Biondi A, Berkefeld J, Klijn CJ, Harkness K, Libman R, Barreau X, Moskowitz AJ, international ARUBA investigators. Medical management with or without interventional therapy for unruptured brain arteriovenous malformations (ARUBA): a multicentre, non-blinded, randomised trial. Lancet (London, England). 2014 Feb 15:383(9917):614-21. doi: 10.1016/S0140-6736(13)62302-8. Epub 2013 Nov 20
[PubMed PMID: 24268105]
Level 1 (high-level) evidence
[47]
Spetzler RF, Martin NA. A proposed grading system for arteriovenous malformations. Journal of neurosurgery. 1986 Oct:65(4):476-83
[PubMed PMID: 3760956]
[48]
Schramm J, Schaller K, Esche J, Boström A. Microsurgery for cerebral arteriovenous malformations: subgroup outcomes in a consecutive series of 288 cases. Journal of neurosurgery. 2017 Apr:126(4):1056-1063. doi: 10.3171/2016.4.JNS153017. Epub 2016 Jun 10
[PubMed PMID: 27285541]
Level 3 (low-level) evidence
[49]
Lunsford LD, Niranjan A, Kondziolka D, Sirin S, Flickinger JC. Arteriovenous malformation radiosurgery: a twenty year perspective. Clinical neurosurgery. 2008:55():108-19
[PubMed PMID: 19248675]
Level 3 (low-level) evidence
[52]
Blume JE. Generalized essential telangiectasia: a case report and review of the literature. Cutis. 2005 Apr:75(4):223-4
[PubMed PMID: 15916219]
Level 3 (low-level) evidence
[53]
Rebellato PR, Martins LE, Stella BT, Tokarski MC. A case of benign hereditary telangiectasia without family history. Anais brasileiros de dermatologia. 2017 Jan-Feb:92(1):162-163. doi: 10.1590/abd1806-4841.20174537. Epub
[PubMed PMID: 28225986]
Level 3 (low-level) evidence
[55]
Costa DL, Moura HH, Rodrigues R, Pineiro-Maceira J, Ramos-E-Silva M. Telangiectasia macularis eruptiva perstans: a rare form of adult mastocytosis. The Journal of clinical and aesthetic dermatology. 2011 Oct:4(10):52-4
[PubMed PMID: 22010057]
[58]
Gamboa NT, Joyce EJ, Eli I, Park MS, Taussky P, Schmidt RH, McDonald J, Whitehead KJ, Kalani MYS. Clinical presentation and treatment paradigms of brain arteriovenous malformations in patients with hereditary hemorrhagic telangiectasia. Journal of clinical neuroscience : official journal of the Neurosurgical Society of Australasia. 2018 May:51():22-28. doi: 10.1016/j.jocn.2018.01.019. Epub 2018 Feb 23
[PubMed PMID: 29483005]
[59]
Woodall MN, Nakaji P, Spetzler RF. Benefits of Treating Arteriovenous Malformations in Hereditary Hemorrhagic Telangiectasia: A Retrospective Analysis of 14 Patients. World neurosurgery: X. 2019 Jul:3():100029. doi: 10.1016/j.wnsx.2019.100029. Epub 2019 Mar 9
[PubMed PMID: 31225521]
Level 2 (mid-level) evidence
[60]
Arizmendez NP, Rudmik L, Poetker DM. Intravenous bevacizumab for complications of hereditary hemorrhagic telangiectasia: a review of the literature. International forum of allergy & rhinology. 2015 Nov:5(11):1042-7. doi: 10.1002/alr.21587. Epub 2015 Jul 22
[PubMed PMID: 26202958]
[61]
Dupuis-Girod S, Ginon I, Saurin JC, Marion D, Guillot E, Decullier E, Roux A, Carette MF, Gilbert-Dussardier B, Hatron PY, Lacombe P, Lorcerie B, Rivière S, Corre R, Giraud S, Bailly S, Paintaud G, Ternant D, Valette PJ, Plauchu H, Faure F. Bevacizumab in patients with hereditary hemorrhagic telangiectasia and severe hepatic vascular malformations and high cardiac output. JAMA. 2012 Mar 7:307(9):948-55. doi: 10.1001/jama.2012.250. Epub
[PubMed PMID: 22396517]
[62]
Dupuis-Girod S, Ambrun A, Decullier E, Samson G, Roux A, Fargeton AE, Rioufol C, Schwiertz V, Disant F, Chapuis F, Donazzolo Y, Paintaud G, Edery P, Faure F. ELLIPSE Study: a Phase 1 study evaluating the tolerance of bevacizumab nasal spray in the treatment of epistaxis in hereditary hemorrhagic telangiectasia. mAbs. 2014 May-Jun:6(3):794-9. doi: 10.4161/mabs.28025. Epub 2014 Jan 30
[PubMed PMID: 24481211]
[63]
Riss D, Burian M, Wolf A, Kranebitter V, Kaider A, Arnoldner C. Intranasal submucosal bevacizumab for epistaxis in hereditary hemorrhagic telangiectasia: a double-blind, randomized, placebo-controlled trial. Head & neck. 2015 Jun:37(6):783-7. doi: 10.1002/hed.23655. Epub 2014 Apr 30
[PubMed PMID: 24595923]
Level 1 (high-level) evidence
[64]
Karnezis TT, Davidson TM. Treatment of hereditary hemorrhagic telangiectasia with submucosal and topical bevacizumab therapy. The Laryngoscope. 2012 Mar:122(3):495-7. doi: 10.1002/lary.22501. Epub 2011 Dec 6
[PubMed PMID: 22147664]
[65]
Stokes P, Rimmer J. Intranasal bevacizumab in the treatment of HHT -related epistaxis: a systematic review. Rhinology. 2018 Mar 1:56(1):3-10. doi: 10.4193/Rhin17.166. Epub
[PubMed PMID: 29166422]
Level 1 (high-level) evidence
[66]
Guldmann R, Dupret A, Nivoix Y, Schultz P, Debry C. Bevacizumab nasal spray: Noninvasive treatment of epistaxis in patients with Rendu-Osler disease. The Laryngoscope. 2012 May:122(5):953-5. doi: 10.1002/lary.23230. Epub 2012 Mar 23
[PubMed PMID: 22447341]
[67]
Dheyauldeen S, Østertun Geirdal A, Osnes T, Vartdal LS, Dollner R. Bevacizumab in hereditary hemorrhagic telangiectasia-associated epistaxis: effectiveness of an injection protocol based on the vascular anatomy of the nose. The Laryngoscope. 2012 Jun:122(6):1210-4. doi: 10.1002/lary.23303. Epub 2012 May 7
[PubMed PMID: 22565282]
[68]
Invernizzi R, Quaglia F, Klersy C, Pagella F, Ornati F, Chu F, Matti E, Spinozzi G, Plumitallo S, Grignani P, Olivieri C, Bastia R, Bellistri F, Danesino C, Benazzo M, Balduini CL. Efficacy and safety of thalidomide for the treatment of severe recurrent epistaxis in hereditary haemorrhagic telangiectasia: results of a non-randomised, single-centre, phase 2 study. The Lancet. Haematology. 2015 Nov:2(11):e465-73. doi: 10.1016/S2352-3026(15)00195-7. Epub 2015 Oct 27
[PubMed PMID: 26686256]
Level 2 (mid-level) evidence
[69]
Lebrin F, Srun S, Raymond K, Martin S, van den Brink S, Freitas C, Bréant C, Mathivet T, Larrivée B, Thomas JL, Arthur HM, Westermann CJ, Disch F, Mager JJ, Snijder RJ, Eichmann A, Mummery CL. Thalidomide stimulates vessel maturation and reduces epistaxis in individuals with hereditary hemorrhagic telangiectasia. Nature medicine. 2010 Apr:16(4):420-8. doi: 10.1038/nm.2131. Epub 2010 Apr 4
[PubMed PMID: 20364125]
[70]
Hosman A, Westermann CJ, Snijder R, Disch F, Mummery CL, Mager JJ. Follow-up of Thalidomide treatment in patients with Hereditary Haemorrhagic Telangiectasia. Rhinology. 2015 Dec:53(4):340-4. doi: 10.4193/Rhino14.289. Epub
[PubMed PMID: 26735132]
[71]
Jameson JJ, Cave DR. Hormonal and antihormonal therapy for epistaxis in hereditary hemorrhagic telangiectasia. The Laryngoscope. 2004 Apr:114(4):705-9
[PubMed PMID: 15064628]
[72]
Sabbà C, Pasculli G, Suppressa P, D'Ovidio F, Lenato GM, Resta F, Assennato G, Guanti G. Life expectancy in patients with hereditary haemorrhagic telangiectasia. QJM : monthly journal of the Association of Physicians. 2006 May:99(5):327-34
[PubMed PMID: 16595564]
[73]
Trerotola SO, Pyeritz RE. PAVM embolization: an update. AJR. American journal of roentgenology. 2010 Oct:195(4):837-45. doi: 10.2214/AJR.10.5230. Epub
[PubMed PMID: 20858807]
[74]
Chamarthy MR, Park H, Sutphin P, Kumar G, Lamus D, Saboo S, Anderson M, Kalva SP. Pulmonary arteriovenous malformations: endovascular therapy. Cardiovascular diagnosis and therapy. 2018 Jun:8(3):338-349. doi: 10.21037/cdt.2017.12.08. Epub
[PubMed PMID: 30057880]