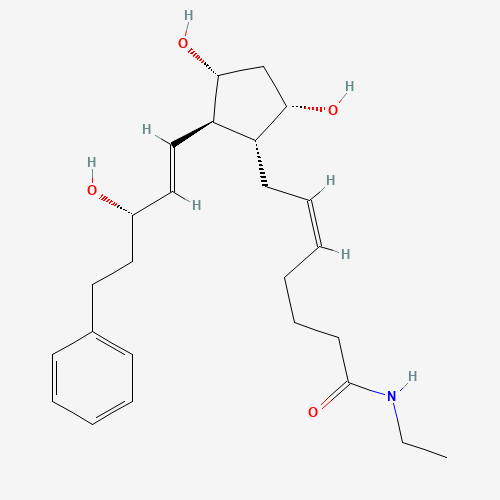
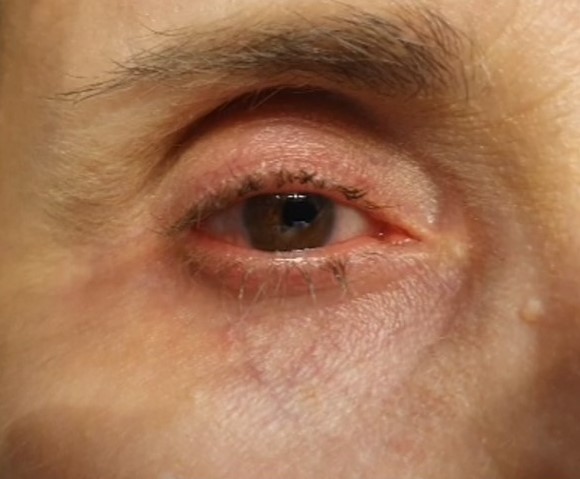
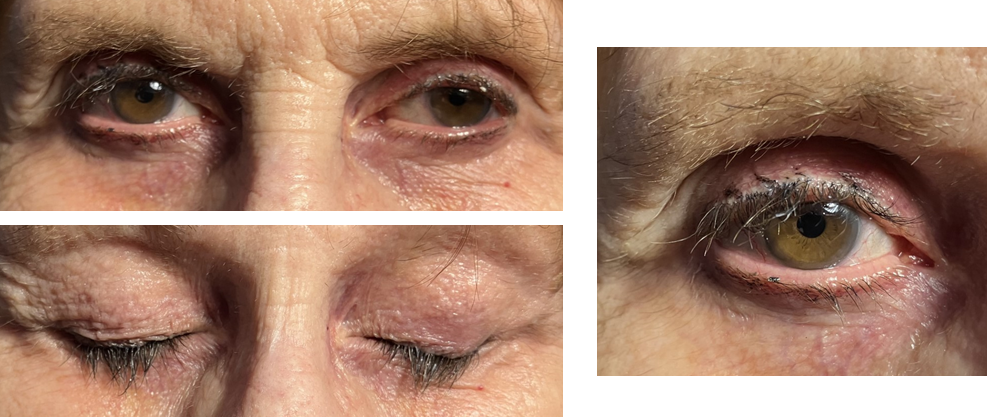
[1]
Li T, Lindsley K, Rouse B, Hong H, Shi Q, Friedman DS, Wormald R, Dickersin K. Comparative Effectiveness of First-Line Medications for Primary Open-Angle Glaucoma: A Systematic Review and Network Meta-analysis. Ophthalmology. 2016 Jan:123(1):129-40. doi: 10.1016/j.ophtha.2015.09.005. Epub 2015 Oct 31
[PubMed PMID: 26526633]
Level 2 (mid-level) evidence
[2]
Cohen JL. Enhancing the growth of natural eyelashes: the mechanism of bimatoprost-induced eyelash growth. Dermatologic surgery : official publication for American Society for Dermatologic Surgery [et al.]. 2010 Sep:36(9):1361-71. doi: 10.1111/j.1524-4725.2010.01522.x. Epub
[PubMed PMID: 20384750]
[3]
Barrón-Hernández YL, Tosti A. Bimatoprost for the treatment of eyelash, eyebrow and scalp alopecia. Expert opinion on investigational drugs. 2017 Apr:26(4):515-522. doi: 10.1080/13543784.2017.1303480. Epub 2017 Mar 16
[PubMed PMID: 28264599]
Level 3 (low-level) evidence
[4]
Wirta D, Baumann L, Bruce S, Ahluwalia G, Weng E, Daniels S. Safety and Efficacy of Bimatoprost for Eyelash Growth in Postchemotherapy Subjects. The Journal of clinical and aesthetic dermatology. 2015 Apr:8(4):11-20
[PubMed PMID: 26060513]
[5]
Jones D. Enhanced eyelashes: prescription and over-the-counter options. Aesthetic plastic surgery. 2011 Feb:35(1):116-21. doi: 10.1007/s00266-010-9561-3. Epub 2010 Aug 21
[PubMed PMID: 20730536]
[6]
Moon SB, Han SB. Efficacy and safety of fixed-combination bimatoprost/timolol ophthalmic solution. Patient preference and adherence. 2017:11():1069-1070. doi: 10.2147/PPA.S141868. Epub 2017 Jun 26
[PubMed PMID: 29200831]
[7]
Lee D, Mantravadi AV, Myers JS. Patient considerations in ocular hypertension: role of bimatoprost ophthalmic solution. Clinical ophthalmology (Auckland, N.Z.). 2017:11():1273-1280. doi: 10.2147/OPTH.S118689. Epub 2017 Jul 10
[PubMed PMID: 28744094]
[8]
Wirta D, Pariser DM, Yoelin SG, Arase S, McMichael A, Weng E, Mao C, Demos G, Vandenburgh A. Bimatoprost 0.03% for the Treatment of Eyelash Hypotrichosis: A Pooled Safety Analysis of Six Randomized, Double-masked Clinical Trials. The Journal of clinical and aesthetic dermatology. 2015 Jul:8(7):17-29
[PubMed PMID: 26203317]
Level 1 (high-level) evidence
[9]
Wan Z, Woodward DF, Cornell CL, Fliri HG, Martos JL, Pettit SN, Wang JW, Kharlamb AB, Wheeler LA, Garst ME, Landsverk KJ, Struble CS, Stamer WD. Bimatoprost, prostamide activity, and conventional drainage. Investigative ophthalmology & visual science. 2007 Sep:48(9):4107-15
[PubMed PMID: 17724194]
[10]
Patil AJ, Vajaranant TS, Edward DP. Bimatoprost - a review. Expert opinion on pharmacotherapy. 2009 Nov:10(16):2759-68. doi: 10.1517/14656560903292649. Epub
[PubMed PMID: 19874254]
Level 3 (low-level) evidence
[11]
Curran MP, Orman JS. Bimatoprost/timolol: a review of its use in glaucoma and ocular hypertension. Drugs & aging. 2009:26(2):169-84. doi: 10.2165/0002512-200926020-00008. Epub
[PubMed PMID: 19220073]
[12]
Doucette LP, Walter MA. Prostaglandins in the eye: Function, expression, and roles in glaucoma. Ophthalmic genetics. 2017 Mar-Apr:38(2):108-116. doi: 10.3109/13816810.2016.1164193. Epub 2016 Apr 12
[PubMed PMID: 27070211]
[13]
Giuffrè G. The effects of prostaglandin F2 alpha in the human eye. Graefe's archive for clinical and experimental ophthalmology = Albrecht von Graefes Archiv fur klinische und experimentelle Ophthalmologie. 1985:222(3):139-41
[PubMed PMID: 3856545]
[14]
Angeli A, Supuran CT. Prostaglandin receptor agonists as antiglaucoma agents (a patent review 2013 - 2018). Expert opinion on therapeutic patents. 2019 Oct:29(10):793-803. doi: 10.1080/13543776.2019.1661992. Epub 2019 Sep 4
[PubMed PMID: 31462124]
Level 3 (low-level) evidence
[15]
Sharif NA, Kelly CR, Crider JY, Williams GW, Xu SX. Ocular hypotensive FP prostaglandin (PG) analogs: PG receptor subtype binding affinities and selectivities, and agonist potencies at FP and other PG receptors in cultured cells. Journal of ocular pharmacology and therapeutics : the official journal of the Association for Ocular Pharmacology and Therapeutics. 2003 Dec:19(6):501-15
[PubMed PMID: 14733708]
[16]
Woodward DF, Liang Y, Krauss AH. Prostamides (prostaglandin-ethanolamides) and their pharmacology. British journal of pharmacology. 2008 Feb:153(3):410-9
[PubMed PMID: 17721551]
[17]
Crowston JG, Lindsey JD, Morris CA, Wheeler L, Medeiros FA, Weinreb RN. Effect of bimatoprost on intraocular pressure in prostaglandin FP receptor knockout mice. Investigative ophthalmology & visual science. 2005 Dec:46(12):4571-7
[PubMed PMID: 16303950]
[18]
Agange N, Mosaed S. Prostaglandin-induced cystoid macular edema following routine cataract extraction. Journal of ophthalmology. 2010:2010():690707. doi: 10.1155/2010/690707. Epub 2010 Nov 7
[PubMed PMID: 21076526]
[19]
Camras CB, Sharif NA, Wax MB, Stjernschantz J. Bimatoprost, the prodrug of a prostaglandin analogue. The British journal of ophthalmology. 2008 Jun:92(6):862-3; author reply 863-4
[PubMed PMID: 18523093]
[20]
Woodward DF, Krauss AH, Chen J, Liang Y, Li C, Protzman CE, Bogardus A, Chen R, Kedzie KM, Krauss HA, Gil DW, Kharlamb A, Wheeler LA, Babusis D, Welty D, Tang-Liu DD, Cherukury M, Andrews SW, Burk RM, Garst ME. Pharmacological characterization of a novel antiglaucoma agent, Bimatoprost (AGN 192024). The Journal of pharmacology and experimental therapeutics. 2003 May:305(2):772-85
[PubMed PMID: 12606640]
[21]
Hellberg MR, Ke TL, Haggard K, Klimko PG, Dean TR, Graff G. The hydrolysis of the prostaglandin analog prodrug bimatoprost to 17-phenyl-trinor PGF2alpha by human and rabbit ocular tissue. Journal of ocular pharmacology and therapeutics : the official journal of the Association for Ocular Pharmacology and Therapeutics. 2003 Apr:19(2):97-103
[PubMed PMID: 12804054]
[22]
Cantor LB, Hoop J, Wudunn D, Yung CW, Catoira Y, Valluri S, Cortes A, Acheampong A, Woodward DF, Wheeler LA. Levels of bimatoprost acid in the aqueous humour after bimatoprost treatment of patients with cataract. The British journal of ophthalmology. 2007 May:91(5):629-32
[PubMed PMID: 17135335]
[23]
Prum BE Jr, Lim MC, Mansberger SL, Stein JD, Moroi SE, Gedde SJ, Herndon LW Jr, Rosenberg LF, Williams RD. Primary Open-Angle Glaucoma Suspect Preferred Practice Pattern(®) Guidelines. Ophthalmology. 2016 Jan:123(1):P112-51. doi: 10.1016/j.ophtha.2015.10.055. Epub 2015 Nov 12
[PubMed PMID: 26581560]
[24]
Christiansen GA, Nau CB, McLaren JW, Johnson DH. Mechanism of ocular hypotensive action of bimatoprost (Lumigan) in patients with ocular hypertension or glaucoma. Ophthalmology. 2004 Sep:111(9):1658-62
[PubMed PMID: 15350319]
[25]
Subbulakshmi S, Kavitha S, Venkatesh R. Prostaglandin analogs in ophthalmology. Indian journal of ophthalmology. 2023 May:71(5):1768-1776. doi: 10.4103/IJO.IJO_2706_22. Epub
[PubMed PMID: 37203029]
[26]
Park KS, Kim KN, Kim KM, Lee HM, Lee SB, Lee NH, Kim CS. Effects of Topical Prostaglandin Analog on Macular Thickness Following Cataract Surgery with Postoperative Topical Bromfenac Treatment. Journal of clinical medicine. 2020 Sep 7:9(9):. doi: 10.3390/jcm9092883. Epub 2020 Sep 7
[PubMed PMID: 32906606]
[27]
Romano MR, Lograno MD. Evidence for the involvement of cannabinoid CB1 receptors in the bimatoprost-induced contractions on the human isolated ciliary muscle. Investigative ophthalmology & visual science. 2007 Aug:48(8):3677-82
[PubMed PMID: 17652738]
[28]
Toris CB, Gabelt BT, Kaufman PL. Update on the mechanism of action of topical prostaglandins for intraocular pressure reduction. Survey of ophthalmology. 2008 Nov:53 Suppl1(SUPPL1):S107-20. doi: 10.1016/j.survophthal.2008.08.010. Epub
[PubMed PMID: 19038618]
Level 3 (low-level) evidence
[29]
Wang JW, Woodward DF, Stamer WD. Differential effects of prostaglandin E2-sensitive receptors on contractility of human ocular cells that regulate conventional outflow. Investigative ophthalmology & visual science. 2013 Jul 16:54(7):4782-90. doi: 10.1167/iovs.13-12363. Epub 2013 Jul 16
[PubMed PMID: 23766471]
[30]
Lim KS, Nau CB, O'Byrne MM, Hodge DO, Toris CB, McLaren JW, Johnson DH. Mechanism of action of bimatoprost, latanoprost, and travoprost in healthy subjects. A crossover study. Ophthalmology. 2008 May:115(5):790-795.e4. doi: 10.1016/j.ophtha.2007.07.002. Epub
[PubMed PMID: 18452763]
[31]
Stein JD, Khawaja AP, Weizer JS. Glaucoma in Adults-Screening, Diagnosis, and Management: A Review. JAMA. 2021 Jan 12:325(2):164-174. doi: 10.1001/jama.2020.21899. Epub
[PubMed PMID: 33433580]
[32]
Tang W, Zhang F, Liu K, Duan X. Efficacy and safety of prostaglandin analogues in primary open-angle glaucoma or ocular hypertension patients: A meta-analysis. Medicine. 2019 Jul:98(30):e16597. doi: 10.1097/MD.0000000000016597. Epub
[PubMed PMID: 31348303]
Level 1 (high-level) evidence
[33]
Yildirim N, Sahin A, Gultekin S. The effect of latanoprost, bimatoprost, and travoprost on circadian variation of intraocular pressure in patients with open-angle glaucoma. Journal of glaucoma. 2008 Jan-Feb:17(1):36-9. doi: 10.1097/IJG.0b013e318133fb70. Epub
[PubMed PMID: 18303382]
[34]
Cai Z, Cao M, Liu K, Duan X. Analysis of the Responsiveness of Latanoprost, Travoprost, Bimatoprost, and Tafluprost in the Treatment of OAG/OHT Patients. Journal of ophthalmology. 2021:2021():5586719. doi: 10.1155/2021/5586719. Epub 2021 May 25
[PubMed PMID: 34123413]
[35]
Law SK, Song BJ, Fang E, Caprioli J. Feasibility and efficacy of a mass switch from latanoprost to bimatoprost in glaucoma patients in a prepaid Health Maintenance Organization. Ophthalmology. 2005 Dec:112(12):2123-30
[PubMed PMID: 16225924]
Level 2 (mid-level) evidence
[36]
Curran MP. Bimatoprost: a review of its use in open-angle glaucoma and ocular hypertension. Drugs & aging. 2009:26(12):1049-71. doi: 10.2165/11203210-000000000-00000. Epub
[PubMed PMID: 19929032]
[37]
Deshpande SS, Sonty S, Ahmad A. Evaluating intraocular pressure-lowering solutions for the treatment of open-angle glaucoma: comparison between bimatoprost 0.03% and bimatoprost 0.01% - an observational switch study. Clinical ophthalmology (Auckland, N.Z.). 2017:11():1371-1376. doi: 10.2147/OPTH.S137537. Epub 2017 Jul 27
[PubMed PMID: 28794615]
[38]
Petricca S, Celenza G, Costagliola C, Tranfa F, Iorio R. Cytotoxicity, Mitochondrial Functionality, and Redox Status of Human Conjunctival Cells after Short and Chronic Exposure to Preservative-Free Bimatoprost 0.03% and 0.01%: An In Vitro Comparative Study. International journal of molecular sciences. 2022 Nov 15:23(22):. doi: 10.3390/ijms232214113. Epub 2022 Nov 15
[PubMed PMID: 36430590]
Level 2 (mid-level) evidence
[39]
Figus M, Nardi M, Piaggi P, Sartini M, Guidi G, Martini L, Lazzeri S. Bimatoprost 0.01% vs bimatoprost 0.03%: a 12-month prospective trial of clinical and in vivo confocal microscopy in glaucoma patients. Eye (London, England). 2014 Apr:28(4):422-9. doi: 10.1038/eye.2013.304. Epub 2014 Jan 17
[PubMed PMID: 24434659]
[40]
Pillunat LE, Eschstruth P, Häsemeyer S, Thelen U, Foja C, Leaback R, Pfennigsdorf S. Preservative-free bimatoprost 0.03% in patients with primary open-angle glaucoma or ocular hypertension in clinical practice. Clinical ophthalmology (Auckland, N.Z.). 2016:10():1759-65. doi: 10.2147/OPTH.S103084. Epub 2016 Sep 12
[PubMed PMID: 27672307]
[41]
Mrukwa-Kominek E, Misiuk-Hojlo M, Csutak A, Stalmans I, Garhofer G. A randomized clinical trial comparing three fixed combinations of bimatoprost with timolol in patients with open-angle glaucoma or ocular hypertension. Current medical research and opinion. 2023 May:39(5):775-783. doi: 10.1080/03007995.2023.2197496. Epub 2023 Apr 17
[PubMed PMID: 37013445]
Level 1 (high-level) evidence
[42]
Xu KM, Cho R, Chan TYB. Retrospective Analysis of Switching Bimatoprost 0.01% to Bimatoprost 0.03% in Patients with Various Types of Glaucoma and Ocular Hypertension. Clinical ophthalmology (Auckland, N.Z.). 2022:16():2385-2390. doi: 10.2147/OPTH.S368214. Epub 2022 Jul 29
[PubMed PMID: 35936971]
Level 2 (mid-level) evidence
[43]
Filippelli M, Campagna G, Ciampa N, Fioretto G, Giannini R, Marino PF, dell'Omo R, Costagliola C. Ocular Tolerability of Bimatoprost 0.1 mg/mL Preservative-Free versus Bimatoprost 0.1 mg/mL with Benzalkonium Chloride or Bimatoprost 0.3 mg/mL Preservative-Free in Patients with Primary Open-Angle Glaucoma. Journal of clinical medicine. 2022 Jun 19:11(12):. doi: 10.3390/jcm11123518. Epub 2022 Jun 19
[PubMed PMID: 35743588]
[44]
Gedde SJ, Vinod K, Wright MM, Muir KW, Lind JT, Chen PP, Li T, Mansberger SL, American Academy of Ophthalmology Preferred Practice Pattern Glaucoma Panel. Primary Open-Angle Glaucoma Preferred Practice Pattern®. Ophthalmology. 2021 Jan:128(1):P71-P150. doi: 10.1016/j.ophtha.2020.10.022. Epub 2020 Nov 12
[PubMed PMID: 34933745]
[45]
Bacharach J, Tatham A, Ferguson G, Belalcázar S, Thieme H, Goodkin ML, Chen MY, Guo Q, Liu J, Robinson MR, Bejanian M, Wirta DL, ARTEMIS 2 Study Group. Phase 3, Randomized, 20-Month Study of the Efficacy and Safety of Bimatoprost Implant in Patients with Open-Angle Glaucoma and Ocular Hypertension (ARTEMIS 2). Drugs. 2021 Nov:81(17):2017-2033. doi: 10.1007/s40265-021-01624-9. Epub 2021 Nov 1
[PubMed PMID: 34724172]
Level 1 (high-level) evidence
[46]
Birt CM, Buys YM, Ahmed II, Trope GE, Toronto Area Glaucoma Society. Prostaglandin efficacy and safety study undertaken by race (the PRESSURE study). Journal of glaucoma. 2010 Sep:19(7):460-7. doi: 10.1097/IJG.0b013e3181c4aeac. Epub
[PubMed PMID: 20051890]
[47]
Menon MG, Goodkin ML. Triple Fixed-Combination Bimatoprost/Brimonidine/Timolol in Glaucoma and Ocular Hypertension in India: A Multicenter, Open-Label, Phase 3 Study. Clinical ophthalmology (Auckland, N.Z.). 2022:16():3559-3569. doi: 10.2147/OPTH.S369626. Epub 2022 Oct 20
[PubMed PMID: 36274674]
[48]
Lemmens S, Rossetti L, Oddone F, Sunaric-Mégevand G, Hommer A, Vandewalle E, Francesca Cordeiro M, McNaught A, Montesano G, Stalmans I. Comparison of preserved bimatoprost 0.01% with preservative-free tafluprost: A randomised, investigator-masked, 3-month crossover, multicentre trial, SPORT II. European journal of ophthalmology. 2021 Apr 5:():11206721211006573. doi: 10.1177/11206721211006573. Epub 2021 Apr 5
[PubMed PMID: 33818170]
Level 1 (high-level) evidence
[49]
Bourne RRA, Kaarniranta K, Lorenz K, Traverso CE, Vuorinen J, Ropo A. Changes in ocular signs and symptoms in patients switching from bimatoprost-timolol to tafluprost-timolol eye drops: an open-label phase IV study. BMJ open. 2019 Apr 2:9(4):e024129. doi: 10.1136/bmjopen-2018-024129. Epub 2019 Apr 2
[PubMed PMID: 30944129]
[50]
Kim JH, Kim EJ, Kim YH, Kim YI, Lee SH, Jung JC, Lee KW, Park YJ. In Vivo Effects of Preservative-free and Preserved Prostaglandin Analogs: Mouse Ocular Surface Study. Korean journal of ophthalmology : KJO. 2015 Aug:29(4):270-9. doi: 10.3341/kjo.2015.29.4.270. Epub 2015 Jul 21
[PubMed PMID: 26240512]
[51]
Tham YC, Li X, Wong TY, Quigley HA, Aung T, Cheng CY. Global prevalence of glaucoma and projections of glaucoma burden through 2040: a systematic review and meta-analysis. Ophthalmology. 2014 Nov:121(11):2081-90. doi: 10.1016/j.ophtha.2014.05.013. Epub 2014 Jun 26
[PubMed PMID: 24974815]
Level 1 (high-level) evidence
[52]
Plosker GL, Keam SJ. Bimatoprost: a pharmacoeconomic review of its use in open-angle glaucoma and ocular hypertension. PharmacoEconomics. 2006:24(3):297-314
[PubMed PMID: 16519552]
[53]
Mohan N, Chakrabarti A, Nazm N, Mehta R, Edward DP. Newer advances in medical management of glaucoma. Indian journal of ophthalmology. 2022 Jun:70(6):1920-1930. doi: 10.4103/ijo.IJO_2239_21. Epub
[PubMed PMID: 35647957]
Level 3 (low-level) evidence
[54]
Alany RG. Adherence, persistence and cost-consequence comparison of bimatoprost topical ocular formulations. Current medical research and opinion. 2013 Sep:29(9):1187-9. doi: 10.1185/03007995.2013.818968. Epub 2013 Jul 5
[PubMed PMID: 23829677]
Level 3 (low-level) evidence
[55]
Holmstrom S, Buchholz P, Walt J, Wickstrøm J, Aagren M. The cost-effectiveness of bimatoprost, latanoprost and timolol in treatment of primary open angle glaucoma in five European countries. Current medical research and opinion. 2006 May:22(5):897-905
[PubMed PMID: 16709311]
Level 3 (low-level) evidence
[56]
Woodward DF, Carling RW, Cornell CL, Fliri HG, Martos JL, Pettit SN, Liang Y, Wang JW. The pharmacology and therapeutic relevance of endocannabinoid derived cyclo-oxygenase (COX)-2 products. Pharmacology & therapeutics. 2008 Oct:120(1):71-80. doi: 10.1016/j.pharmthera.2008.08.001. Epub 2008 Aug 6
[PubMed PMID: 18700152]
[57]
Tauchi M, Fuchs TA, Kellenberger AJ, Woodward DF, Paus R, Lütjen-Drecoll E. Characterization of an in vivo model for the study of eyelash biology and trichomegaly: mouse eyelash morphology, development, growth cycle, and anagen prolongation by bimatoprost. The British journal of dermatology. 2010 Jun:162(6):1186-97. doi: 10.1111/j.1365-2133.2010.09685.x. Epub 2010 Mar 12
[PubMed PMID: 20346040]
[58]
Jiang S, Hao Z, Qi W, Wang Z, Zhou M, Guo N. The efficacy of topical prostaglandin analogs for hair loss: A systematic review and meta-analysis. Frontiers in medicine. 2023:10():1130623. doi: 10.3389/fmed.2023.1130623. Epub 2023 Mar 14
[PubMed PMID: 36999072]
Level 1 (high-level) evidence
[59]
Smith S, Fagien S, Whitcup SM, Ledon F, Somogyi C, Weng E, Beddingfield FC 3rd. Eyelash growth in subjects treated with bimatoprost: a multicenter, randomized, double-masked, vehicle-controlled, parallel-group study. Journal of the American Academy of Dermatology. 2012 May:66(5):801-6. doi: 10.1016/j.jaad.2011.06.005. Epub 2011 Sep 6
[PubMed PMID: 21899919]
Level 1 (high-level) evidence
[60]
Law SK. Bimatoprost in the treatment of eyelash hypotrichosis. Clinical ophthalmology (Auckland, N.Z.). 2010 Apr 26:4():349-58
[PubMed PMID: 20463804]
[61]
Carruthers J, Beer K, Carruthers A, Coleman WP 3rd, Draelos ZD, Jones D, Goldman MP, Pucci ML, VanDenburgh A, Weng E, Whitcup SM. Bimatoprost 0.03% for the Treatment of Eyebrow Hypotrichosis. Dermatologic surgery : official publication for American Society for Dermatologic Surgery [et al.]. 2016 May:42(5):608-17. doi: 10.1097/DSS.0000000000000755. Epub
[PubMed PMID: 27124878]
[62]
Yazdanian N, Mozafarpoor S, Goodarzi A. Phosphodiesterase inhibitors and prostaglandin analogues in dermatology: A comprehensive review. Dermatologic therapy. 2021 Jan:34(1):e14669. doi: 10.1111/dth.14669. Epub 2020 Dec 21
[PubMed PMID: 33314552]
[63]
Sarsik SM, El-Amawy HS. Uses of eye drops in dermatology, literature review. The Journal of dermatological treatment. 2022 Sep:33(6):2758-2770. doi: 10.1080/09546634.2022.2079598. Epub 2022 Jun 2
[PubMed PMID: 35652324]
[64]
Zaher H, Gawdat HI, Hegazy RA, Hassan M. Bimatoprost versus Mometasone Furoate in the Treatment of Scalp Alopecia Areata: A Pilot Study. Dermatology (Basel, Switzerland). 2015:230(4):308-13. doi: 10.1159/000371416. Epub 2015 Mar 4
[PubMed PMID: 25765294]
Level 3 (low-level) evidence
[65]
Fukumoto T, Fukumoto R, Magno E, Oka M, Nishigori C, Horita N. Treatments for alopecia areata: A systematic review and network meta-analysis. Dermatologic therapy. 2021 May:34(3):e14916. doi: 10.1111/dth.14916. Epub 2021 Mar 4
[PubMed PMID: 33631058]
Level 1 (high-level) evidence
[66]
Gupta AK, Bamimore MA, Foley KA. Efficacy of non-surgical treatments for androgenetic alopecia in men and women: a systematic review with network meta-analyses, and an assessment of evidence quality. The Journal of dermatological treatment. 2022 Feb:33(1):62-72. doi: 10.1080/09546634.2020.1749547. Epub 2020 Apr 13
[PubMed PMID: 32250713]
Level 1 (high-level) evidence
[67]
Easthope SE, Perry CM. Topical bimatoprost: a review of its use in open-angle glaucoma and ocular hypertension. Drugs & aging. 2002:19(3):231-48
[PubMed PMID: 12027782]
[68]
Higginbotham EJ, Schuman JS, Goldberg I, Gross RL, VanDenburgh AM, Chen K, Whitcup SM, Bimatoprost Study Groups 1 and 2. One-year, randomized study comparing bimatoprost and timolol in glaucoma and ocular hypertension. Archives of ophthalmology (Chicago, Ill. : 1960). 2002 Oct:120(10):1286-93
[PubMed PMID: 12365906]
Level 1 (high-level) evidence
[69]
Ali MS, Hafiz HSA, Ahmed NA, Galal SA. Combined microneedling with topical vitamin D3 or bimatoprost versus microneedling alone in the treatment of alopecia areata: A comparative randomized trial. Journal of cosmetic dermatology. 2023 Apr:22(4):1286-1296. doi: 10.1111/jocd.15569. Epub 2023 Feb 10
[PubMed PMID: 36762396]
Level 1 (high-level) evidence
[70]
Newman-Casey PA, Robin AL, Blachley T, Farris K, Heisler M, Resnicow K, Lee PP. The Most Common Barriers to Glaucoma Medication Adherence: A Cross-Sectional Survey. Ophthalmology. 2015 Jul:122(7):1308-16. doi: 10.1016/j.ophtha.2015.03.026. Epub 2015 Apr 24
[PubMed PMID: 25912144]
Level 2 (mid-level) evidence
[71]
Gautam M, Gupta R, Singh P, Verma V, Verma S, Mittal P, Karkhur S, Sampath A, Mohan RR, Sharma B. Intracameral Drug Delivery: A Review of Agents, Indications, and Outcomes. Journal of ocular pharmacology and therapeutics : the official journal of the Association for Ocular Pharmacology and Therapeutics. 2023 Mar:39(2):102-116. doi: 10.1089/jop.2022.0144. Epub 2023 Feb 9
[PubMed PMID: 36757304]
[72]
Shirley M. Bimatoprost Implant: First Approval. Drugs & aging. 2020 Jun:37(6):457-462. doi: 10.1007/s40266-020-00769-8. Epub
[PubMed PMID: 32447639]
[73]
Seal JR, Robinson MR, Burke J, Bejanian M, Coote M, Attar M. Intracameral Sustained-Release Bimatoprost Implant Delivers Bimatoprost to Target Tissues with Reduced Drug Exposure to Off-Target Tissues. Journal of ocular pharmacology and therapeutics : the official journal of the Association for Ocular Pharmacology and Therapeutics. 2019 Jan/Feb:35(1):50-57. doi: 10.1089/jop.2018.0067. Epub 2018 Oct 18
[PubMed PMID: 30335560]
[74]
Medeiros FA, Walters TR, Kolko M, Coote M, Bejanian M, Goodkin ML, Guo Q, Zhang J, Robinson MR, Weinreb RN, ARTEMIS 1 Study Group. Phase 3, Randomized, 20-Month Study of Bimatoprost Implant in Open-Angle Glaucoma and Ocular Hypertension (ARTEMIS 1). Ophthalmology. 2020 Dec:127(12):1627-1641. doi: 10.1016/j.ophtha.2020.06.018. Epub 2020 Jun 13
[PubMed PMID: 32544560]
Level 1 (high-level) evidence
[75]
Miller PE, Eaton JS. Medical anti-glaucoma therapy: Beyond the drop. Veterinary ophthalmology. 2021 Mar:24 Suppl 1():2-15. doi: 10.1111/vop.12843. Epub 2020 Nov 9
[PubMed PMID: 33164328]
[76]
Sirinek PE, Lin MM. Intracameral sustained release bimatoprost implants (Durysta). Seminars in ophthalmology. 2022 Apr 3:37(3):385-390. doi: 10.1080/08820538.2021.1985145. Epub 2021 Sep 29
[PubMed PMID: 34586961]
[77]
Brandt JD, DuBiner HB, Benza R, Sall KN, Walker GA, Semba CP, Collaborators. Long-term Safety and Efficacy of a Sustained-Release Bimatoprost Ocular Ring. Ophthalmology. 2017 Oct:124(10):1565-1566. doi: 10.1016/j.ophtha.2017.04.022. Epub 2017 May 17
[PubMed PMID: 28528010]
[78]
Xu J, Ge Y, Bu R, Zhang A, Feng S, Wang J, Gou J, Yin T, He H, Zhang Y, Tang X. Co-delivery of latanoprost and timolol from micelles-laden contact lenses for the treatment of glaucoma. Journal of controlled release : official journal of the Controlled Release Society. 2019 Jul 10:305():18-28. doi: 10.1016/j.jconrel.2019.05.025. Epub 2019 May 16
[PubMed PMID: 31103677]
[79]
Aref AA. Sustained drug delivery for glaucoma: current data and future trends. Current opinion in ophthalmology. 2017 Mar:28(2):169-174. doi: 10.1097/ICU.0000000000000334. Epub
[PubMed PMID: 27764023]
Level 3 (low-level) evidence
[80]
Williams RD, Cohen JS, Gross RL, Liu CC, Safyan E, Batoosingh AL, Bimatoprost Study Group. Long-term efficacy and safety of bimatoprost for intraocular pressure lowering in glaucoma and ocular hypertension: year 4. The British journal of ophthalmology. 2008 Oct:92(10):1387-92. doi: 10.1136/bjo.2007.128454. Epub 2008 Jul 11
[PubMed PMID: 18621791]
[81]
Jbara D, Eiger-Moscovich M, Didkovsky E, Keshet Y, Avisar I. In Vivo Effects of Prostaglandin Analogues Application by Topical Drops or Retrobulbar Injections on the Orbital Fat of a Rat Model. Ocular immunology and inflammation. 2023 Feb:31(2):298-303. doi: 10.1080/09273948.2022.2026977. Epub 2022 Jan 26
[PubMed PMID: 35081015]
[82]
Kucukevcilioglu M, Bayer A, Uysal Y, Altinsoy HI. Prostaglandin associated periorbitopathy in patients using bimatoprost, latanoprost and travoprost. Clinical & experimental ophthalmology. 2014 Mar:42(2):126-31. doi: 10.1111/ceo.12163. Epub 2013 Aug 4
[PubMed PMID: 23844550]
[83]
Shrirao N, Khurana M, Mukherjee B. Prostaglandin-associated periorbitopathy. Indian journal of ophthalmology. 2016 Jun:64(6):459. doi: 10.4103/0301-4738.187676. Epub
[PubMed PMID: 27488155]
[84]
Steinsapir KD, Steinsapir SMG. Revisiting the Safety of Prostaglandin Analog Eyelash Growth Products. Dermatologic surgery : official publication for American Society for Dermatologic Surgery [et al.]. 2021 May 1:47(5):658-665. doi: 10.1097/DSS.0000000000002928. Epub
[PubMed PMID: 33625141]
[85]
Eiger-Moscovich M, Stiebel-Kalish H, Yassur I, Barash D, Gaton D, Avisar I. Prostaglandin analogue drops for the treatment of soft tissue expansion and exophthalmos in patients with inactive thyroid eye disease. Canadian journal of ophthalmology. Journal canadien d'ophtalmologie. 2019 Aug:54(4):426-430. doi: 10.1016/j.jcjo.2018.09.003. Epub 2018 Nov 23
[PubMed PMID: 31358139]
[86]
Kroll DM, Schuman JS. Reactivation of herpes simplex virus keratitis after initiating bimatoprost treatment for glaucoma. American journal of ophthalmology. 2002 Mar:133(3):401-3
[PubMed PMID: 11860979]
[87]
Kothari MT, Mehta BK, Asher NS, Kothari KJ. Recurrence of bilateral herpes simplex virus keratitis following bimatoprost use. Indian journal of ophthalmology. 2006 Mar:54(1):47-8
[PubMed PMID: 16531673]
[88]
Woodward JA, Haggerty CJ, Stinnett SS, Williams ZY. Bimatoprost 0.03% gel for cosmetic eyelash growth and enhancement. Journal of cosmetic dermatology. 2010 Jun:9(2):96-102. doi: 10.1111/j.1473-2165.2010.00487.x. Epub
[PubMed PMID: 20618554]
[89]
Packer M, Fine IH, Hoffman RS. Bilateral nongranulomatous anterior uveitis associated with bimatoprost. Journal of cataract and refractive surgery. 2003 Nov:29(11):2242-3
[PubMed PMID: 14670442]
[90]
Cano Parra J, Díaz-Llopis M. [Drug induced uveitis]. Archivos de la Sociedad Espanola de Oftalmologia. 2005 Mar:80(3):137-49
[PubMed PMID: 15803424]
[91]
Kapur R, Osmanovic S, Toyran S, Edward DP. Bimatoprost-induced periocular skin hyperpigmentation: histopathological study. Archives of ophthalmology (Chicago, Ill. : 1960). 2005 Nov:123(11):1541-6
[PubMed PMID: 16286616]
[92]
Holló G, Aung T, Cantor LB, Aihara M. Cystoid macular edema related to cataract surgery and topical prostaglandin analogs: Mechanism, diagnosis, and management. Survey of ophthalmology. 2020 Sep-Oct:65(5):496-512. doi: 10.1016/j.survophthal.2020.02.004. Epub 2020 Feb 22
[PubMed PMID: 32092363]
Level 3 (low-level) evidence
[93]
Yeh PC, Ramanathan S. Latanoprost and clinically significant cystoid macular edema after uneventful phacoemulsification with intraocular lens implantation. Journal of cataract and refractive surgery. 2002 Oct:28(10):1814-8
[PubMed PMID: 12388034]
[94]
Pepple KL, Nguyen MH, Pakzad-Vaezi K, Williamson K, Odell N, Lee C, Leveque TK, Van Gelder RN. RESPONSE OF INFLAMMATORY CYSTOID MACULAR EDEMA TO TREATMENT USING ORAL ACETAZOLAMIDE. Retina (Philadelphia, Pa.). 2019 May:39(5):948-955. doi: 10.1097/IAE.0000000000002044. Epub
[PubMed PMID: 29346240]
[95]
van der Valk R, Webers CA, Schouten JS, Zeegers MP, Hendrikse F, Prins MH. Intraocular pressure-lowering effects of all commonly used glaucoma drugs: a meta-analysis of randomized clinical trials. Ophthalmology. 2005 Jul:112(7):1177-85
[PubMed PMID: 15921747]
Level 1 (high-level) evidence
[96]
Jamison A, Okafor L, Ullrich K, Schiedler V, Malhotra R. Do Prostaglandin Analogue Lash Lengtheners Cause Eyelid Fat and Volume Loss? Aesthetic surgery journal. 2022 Oct 13:42(11):1241-1249. doi: 10.1093/asj/sjac156. Epub
[PubMed PMID: 35700523]
[97]
Belkin A, Chen T, DeOliveria AR, Johnson SM, Ramulu PY, Buys YM, American Glaucoma Society and the Canadian Glaucoma Society. A Practical Guide to the Pregnant and Breastfeeding Patient with Glaucoma. Ophthalmology. Glaucoma. 2020 Mar-Apr:3(2):79-89. doi: 10.1016/j.ogla.2019.12.004. Epub
[PubMed PMID: 32672600]
[98]
Blumen-Ohana E, Sellem E. [Pregnancy & glaucoma: SFO-SFG recommendations]. Journal francais d'ophtalmologie. 2020 Jan:43(1):63-66. doi: 10.1016/j.jfo.2019.11.001. Epub 2019 Dec 5
[PubMed PMID: 31813552]
[99]
Fagien S, Walt JG, Carruthers J, Cox SE, Wirta D, Weng E, Beddingfield FC 3rd. Patient-reported outcomes of bimatoprost for eyelash growth: results from a randomized, double-masked, vehicle-controlled, parallel-group study. Aesthetic surgery journal. 2013 Aug 1:33(6):789-98. doi: 10.1177/1090820X13495887. Epub 2013 Jul 19
[PubMed PMID: 23873891]
Level 1 (high-level) evidence
[100]
Trattler W, Noecker RJ, Earl ML. A multicentre evaluation of the effect of patient education on acceptance of hyperaemia associated with bimatoprost therapy for glaucoma or ocular hypertension. Advances in therapy. 2008 Mar:25(3):179-89. doi: 10.1007/s12325-008-0029-7. Epub
[PubMed PMID: 18351298]
Level 3 (low-level) evidence