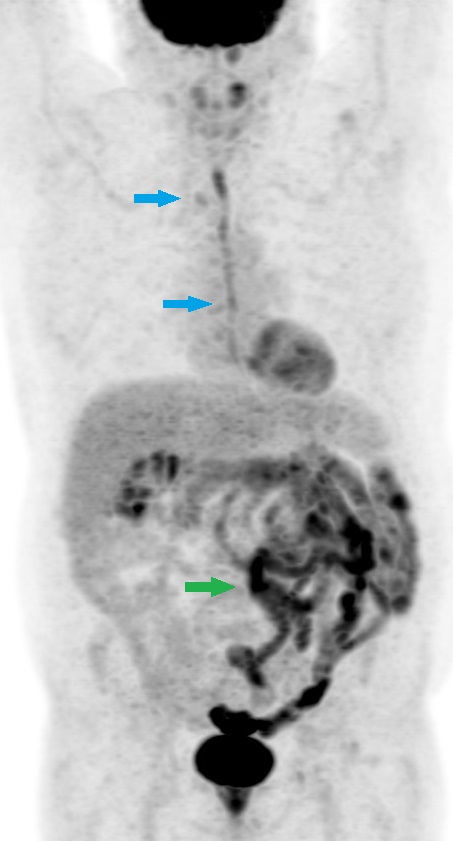
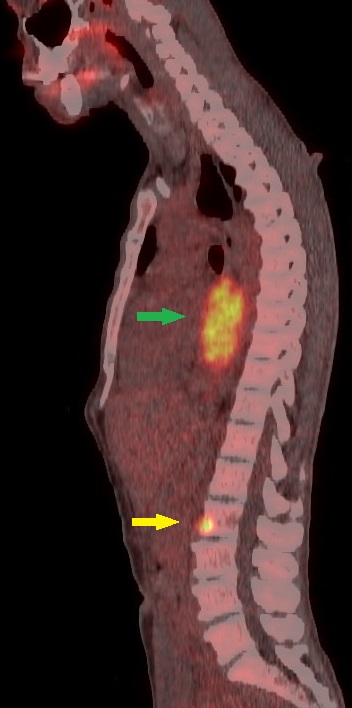
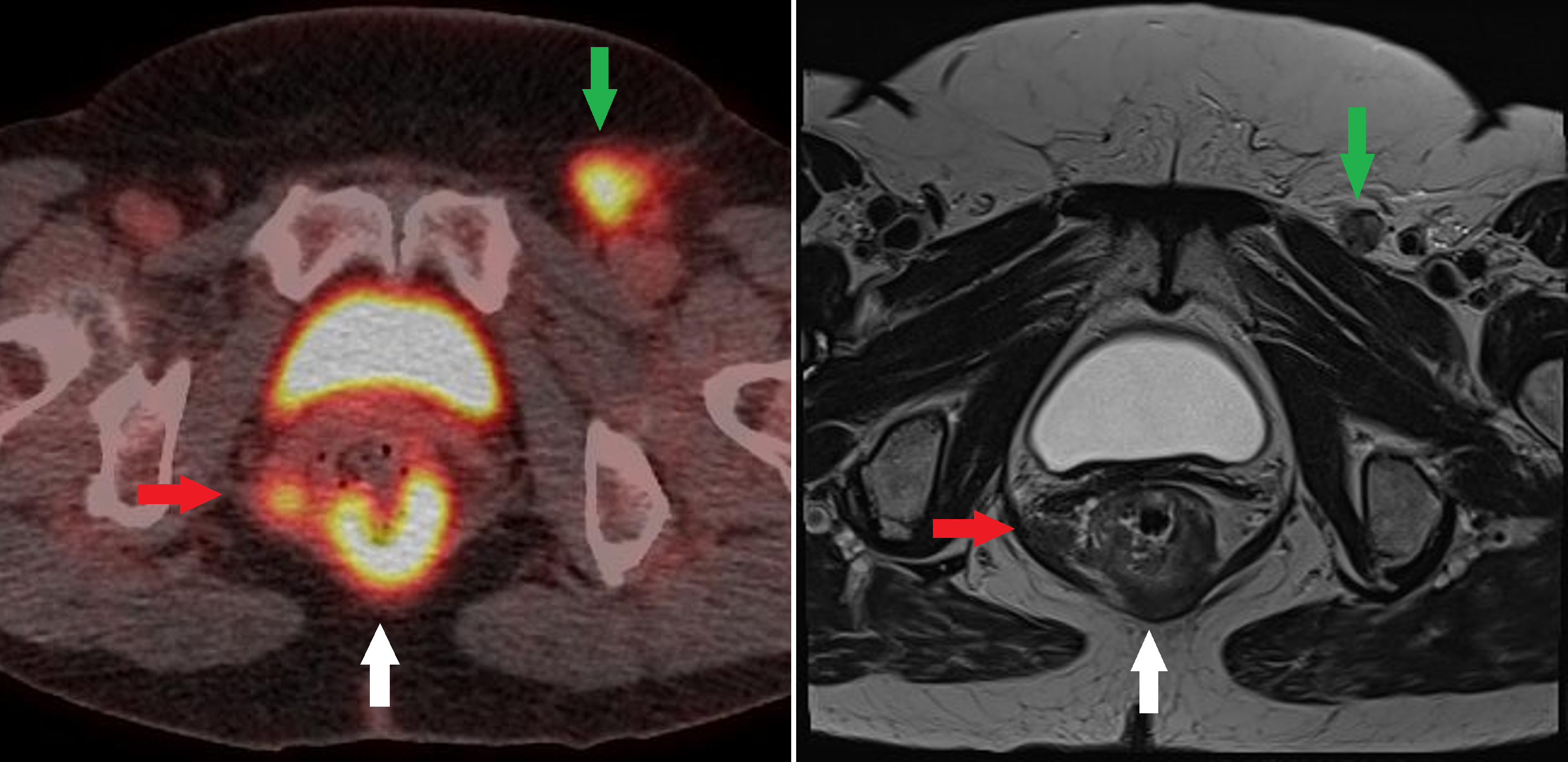
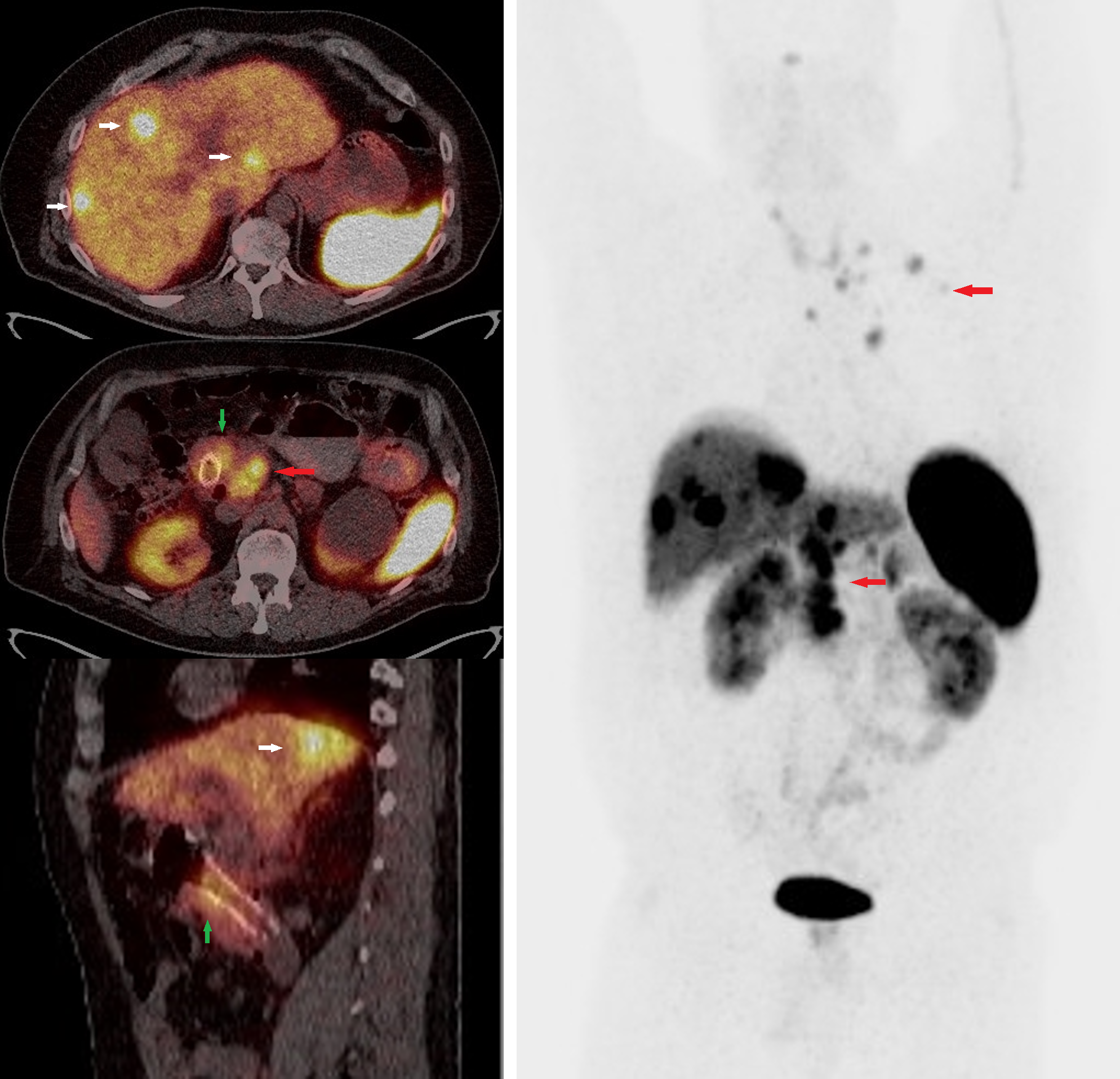
[1]
Bentley-Hibbert S, Schwartz L. Use of Imaging for GI Cancers. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 2015 Jun 1:33(16):1729-36. doi: 10.1200/JCO.2014.60.2847. Epub 2015 Apr 27
[PubMed PMID: 25918300]
[2]
Vaupel P, Schmidberger H, Mayer A. The Warburg effect: essential part of metabolic reprogramming and central contributor to cancer progression. International journal of radiation biology. 2019 Jul:95(7):912-919. doi: 10.1080/09553002.2019.1589653. Epub 2019 Mar 22
[PubMed PMID: 30822194]
[3]
Özgüven S, Filizoğlu N, Kesim S, Öksüzoğlu K, Şen F, Öneş T, İnanır S, Turoğlu HT, Erdil TY. Physiological Biodistribution of (68)Ga-DOTA-TATE in Normal Subjects. Molecular imaging and radionuclide therapy. 2021 Feb 9:30(1):39-46. doi: 10.4274/mirt.galenos.2021.37268. Epub
[PubMed PMID: 33586406]
[4]
Münch S, Marr L, Feuerecker B, Dapper H, Braren R, Combs SE, Duma MN. Impact of (18)F-FDG-PET/CT on the identification of regional lymph node metastases and delineation of the primary tumor in esophageal squamous cell carcinoma patients. Strahlentherapie und Onkologie : Organ der Deutschen Rontgengesellschaft ... [et al]. 2020 Sep:196(9):787-794. doi: 10.1007/s00066-020-01630-y. Epub 2020 May 19
[PubMed PMID: 32430661]
[5]
Shepherd NA, Baxter KJ, Love SB. The prognostic importance of peritoneal involvement in colonic cancer: a prospective evaluation. Gastroenterology. 1997 Apr:112(4):1096-102
[PubMed PMID: 9097991]
[6]
Salati SA, Al Kadi A. Anal cancer - a review. International journal of health sciences. 2012 Jun:6(2):206-30
[PubMed PMID: 23580899]
[7]
Klimstra DS, Modlin IR, Coppola D, Lloyd RV, Suster S. The pathologic classification of neuroendocrine tumors: a review of nomenclature, grading, and staging systems. Pancreas. 2010 Aug:39(6):707-12. doi: 10.1097/MPA.0b013e3181ec124e. Epub
[PubMed PMID: 20664470]
[8]
Cives M, Strosberg JR. Gastroenteropancreatic Neuroendocrine Tumors. CA: a cancer journal for clinicians. 2018 Nov:68(6):471-487. doi: 10.3322/caac.21493. Epub 2018 Oct 8
[PubMed PMID: 30295930]
[9]
Uenomachi M, Takahashi M, Shimazoe K, Takahashi H, Kamada K, Orita T, Ogane K, Tsuji AB. Simultaneous in vivo imaging with PET and SPECT tracers using a Compton-PET hybrid camera. Scientific reports. 2021 Sep 9:11(1):17933. doi: 10.1038/s41598-021-97302-7. Epub 2021 Sep 9
[PubMed PMID: 34504184]
[10]
Karfis I, Marin G, Levillain H, Drisis S, Muteganya R, Critchi G, Taraji-Schiltz L, Guix CA, Shaza L, Elbachiri M, Mans L, Machiels G, Hendlisz A, Flamen P. Prognostic value of a three-scale grading system based on combining molecular imaging with (68)Ga-DOTATATE and (18)F-FDG PET/CT in patients with metastatic gastroenteropancreatic neuroendocrine neoplasias. Oncotarget. 2020 Feb 11:11(6):589-599. doi: 10.18632/oncotarget.27460. Epub 2020 Feb 11
[PubMed PMID: 32110279]
[11]
Long NM, Smith CS. Causes and imaging features of false positives and false negatives on F-PET/CT in oncologic imaging. Insights into imaging. 2011 Dec:2(6):679-698
[PubMed PMID: 22347986]
[12]
Hicks RJ, Binns D, Stabin MG. Pattern of uptake and excretion of (18)F-FDG in the lactating breast. Journal of nuclear medicine : official publication, Society of Nuclear Medicine. 2001 Aug:42(8):1238-42
[PubMed PMID: 11483686]
[13]
Boellaard R, Delgado-Bolton R, Oyen WJ, Giammarile F, Tatsch K, Eschner W, Verzijlbergen FJ, Barrington SF, Pike LC, Weber WA, Stroobants S, Delbeke D, Donohoe KJ, Holbrook S, Graham MM, Testanera G, Hoekstra OS, Zijlstra J, Visser E, Hoekstra CJ, Pruim J, Willemsen A, Arends B, Kotzerke J, Bockisch A, Beyer T, Chiti A, Krause BJ, European Association of Nuclear Medicine (EANM). FDG PET/CT: EANM procedure guidelines for tumour imaging: version 2.0. European journal of nuclear medicine and molecular imaging. 2015 Feb:42(2):328-54. doi: 10.1007/s00259-014-2961-x. Epub 2014 Dec 2
[PubMed PMID: 25452219]
[14]
Benamor M, Ollivier L, Brisse H, Moulin-Romsee G, Servois V, Neuenschwander S. PET/CT imaging: what radiologists need to know. Cancer imaging : the official publication of the International Cancer Imaging Society. 2007 Oct 1:7 Spec No A(Special issue A):S95-9
[PubMed PMID: 17921089]
[15]
Nadig V, Herrmann K, Mottaghy FM, Schulz V. Hybrid total-body pet scanners-current status and future perspectives. European journal of nuclear medicine and molecular imaging. 2022 Jan:49(2):445-459. doi: 10.1007/s00259-021-05536-4. Epub 2021 Oct 14
[PubMed PMID: 34647154]
Level 3 (low-level) evidence
[16]
Vaquero JJ, Kinahan P. Positron Emission Tomography: Current Challenges and Opportunities for Technological Advances in Clinical and Preclinical Imaging Systems. Annual review of biomedical engineering. 2015:17():385-414. doi: 10.1146/annurev-bioeng-071114-040723. Epub
[PubMed PMID: 26643024]
Level 3 (low-level) evidence
[17]
Kapoor V, McCook BM, Torok FS. An introduction to PET-CT imaging. Radiographics : a review publication of the Radiological Society of North America, Inc. 2004 Mar-Apr:24(2):523-43
[PubMed PMID: 15026598]
[18]
Boellaard R, O'Doherty MJ, Weber WA, Mottaghy FM, Lonsdale MN, Stroobants SG, Oyen WJ, Kotzerke J, Hoekstra OS, Pruim J, Marsden PK, Tatsch K, Hoekstra CJ, Visser EP, Arends B, Verzijlbergen FJ, Zijlstra JM, Comans EF, Lammertsma AA, Paans AM, Willemsen AT, Beyer T, Bockisch A, Schaefer-Prokop C, Delbeke D, Baum RP, Chiti A, Krause BJ. FDG PET and PET/CT: EANM procedure guidelines for tumour PET imaging: version 1.0. European journal of nuclear medicine and molecular imaging. 2010 Jan:37(1):181-200. doi: 10.1007/s00259-009-1297-4. Epub
[PubMed PMID: 19915839]
[19]
Ozaslan E, Kiziltepe M, Addulrezzak U, Kula M, Bozkurt O, Kut E, Duran AO, Ucar M, Sakalar T, Dogan E, Topaloglu US, Inanc M, Ozkan M. Is SUVmax of (18)F-FDG PET/CT Predictive Factor for Malignancy in Gastrointestinal Tract? Nigerian journal of clinical practice. 2021 Aug:24(8):1217-1224. doi: 10.4103/njcp.njcp_637_18. Epub
[PubMed PMID: 34397034]
[20]
Shirone N, Shinkai T, Yamane T, Uto F, Yoshimura H, Tamai H, Imai T, Inoue M, Kitano S, Kichikawa K, Hasegawa M. Axillary lymph node accumulation on FDG-PET/CT after influenza vaccination. Annals of nuclear medicine. 2012 Apr:26(3):248-52. doi: 10.1007/s12149-011-0568-x. Epub 2012 Jan 21
[PubMed PMID: 22271546]
[21]
Delbeke D, Coleman RE, Guiberteau MJ, Brown ML, Royal HD, Siegel BA, Townsend DW, Berland LL, Parker JA, Hubner K, Stabin MG, Zubal G, Kachelriess M, Cronin V, Holbrook S. Procedure guideline for tumor imaging with 18F-FDG PET/CT 1.0. Journal of nuclear medicine : official publication, Society of Nuclear Medicine. 2006 May:47(5):885-95
[PubMed PMID: 16644760]
[22]
Kinahan PE, Fletcher JW. Positron emission tomography-computed tomography standardized uptake values in clinical practice and assessing response to therapy. Seminars in ultrasound, CT, and MR. 2010 Dec:31(6):496-505. doi: 10.1053/j.sult.2010.10.001. Epub
[PubMed PMID: 21147377]
[23]
Leide-Svegborn S. Radiation exposure of patients and personnel from a PET/CT procedure with 18F-FDG. Radiation protection dosimetry. 2010 Apr-May:139(1-3):208-13. doi: 10.1093/rpd/ncq026. Epub 2010 Feb 18
[PubMed PMID: 20167792]
[24]
Griffeth LK. Use of PET/CT scanning in cancer patients: technical and practical considerations. Proceedings (Baylor University. Medical Center). 2005 Oct:18(4):321-30
[PubMed PMID: 16252023]
[25]
Berger KL, Nicholson SA, Dehdashti F, Siegel BA. FDG PET evaluation of mucinous neoplasms: correlation of FDG uptake with histopathologic features. AJR. American journal of roentgenology. 2000 Apr:174(4):1005-8
[PubMed PMID: 10749239]
[26]
Steenkamp DW, McDonnell ME, Meibom S. Metformin may be associated with false-negative cancer detection in the gastrointestinal tract on PET/CT. Endocrine practice : official journal of the American College of Endocrinology and the American Association of Clinical Endocrinologists. 2014 Oct:20(10):1079-83. doi: 10.4158/EP14127.RA. Epub
[PubMed PMID: 25100379]
[27]
Lordick F, Mariette C, Haustermans K, Obermannová R, Arnold D, ESMO Guidelines Committee. Oesophageal cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Annals of oncology : official journal of the European Society for Medical Oncology. 2016 Sep:27(suppl 5):v50-v57
[PubMed PMID: 27664261]
Level 1 (high-level) evidence
[28]
Hollis AC, Quinn LM, Hodson J, Evans E, Plowright J, Begum R, Mitchell H, Hallissey MT, Whiting JL, Griffiths EA. Prognostic significance of tumor length in patients receiving esophagectomy for esophageal cancer. Journal of surgical oncology. 2017 Dec:116(8):1114-1122. doi: 10.1002/jso.24789. Epub 2017 Aug 2
[PubMed PMID: 28767142]
[29]
Goense L, van Rossum PS, Reitsma JB, Lam MG, Meijer GJ, van Vulpen M, Ruurda JP, van Hillegersberg R. Diagnostic Performance of ¹⁸F-FDG PET and PET/CT for the Detection of Recurrent Esophageal Cancer After Treatment with Curative Intent: A Systematic Review and Meta-Analysis. Journal of nuclear medicine : official publication, Society of Nuclear Medicine. 2015 Jul:56(7):995-1002. doi: 10.2967/jnumed.115.155580. Epub 2015 May 7
[PubMed PMID: 25952733]
Level 1 (high-level) evidence
[30]
Kitajima K, Nakajo M, Kaida H, Minamimoto R, Hirata K, Tsurusaki M, Doi H, Ueno Y, Sofue K, Tamaki Y, Yamakado K. Present and future roles of FDG-PET/CT imaging in the management of gastrointestinal cancer: an update. Nagoya journal of medical science. 2017 Nov:79(4):527-543. doi: 10.18999/nagjms.79.4.527. Epub
[PubMed PMID: 29238109]
[31]
Jayaprakasam VS, Paroder V, Schöder H. Variants and Pitfalls in PET/CT Imaging of Gastrointestinal Cancers. Seminars in nuclear medicine. 2021 Sep:51(5):485-501. doi: 10.1053/j.semnuclmed.2021.04.001. Epub 2021 May 6
[PubMed PMID: 33965198]
[32]
Moses WW. Fundamental Limits of Spatial Resolution in PET. Nuclear instruments & methods in physics research. Section A, Accelerators, spectrometers, detectors and associated equipment. 2011 Aug 21:648 Supplement 1():S236-S240
[PubMed PMID: 21804677]
[33]
Dahmarde H, Parooie F, Salarzaei M. Is (18)F-FDG PET/CT an Accurate Way to Detect Lymph Node Metastasis in Colorectal Cancer: A Systematic Review and Meta-Analysis. Contrast media & molecular imaging. 2020:2020():5439378. doi: 10.1155/2020/5439378. Epub 2020 Jul 18
[PubMed PMID: 32733174]
Level 1 (high-level) evidence
[34]
Riihimäki M, Hemminki A, Sundquist J, Hemminki K. Patterns of metastasis in colon and rectal cancer. Scientific reports. 2016 Jul 15:6():29765. doi: 10.1038/srep29765. Epub 2016 Jul 15
[PubMed PMID: 27416752]
[35]
Choti MA, Sitzmann JV, Tiburi MF, Sumetchotimetha W, Rangsin R, Schulick RD, Lillemoe KD, Yeo CJ, Cameron JL. Trends in long-term survival following liver resection for hepatic colorectal metastases. Annals of surgery. 2002 Jun:235(6):759-66
[PubMed PMID: 12035031]
[36]
Fernandez FG, Drebin JA, Linehan DC, Dehdashti F, Siegel BA, Strasberg SM. Five-year survival after resection of hepatic metastases from colorectal cancer in patients screened by positron emission tomography with F-18 fluorodeoxyglucose (FDG-PET). Annals of surgery. 2004 Sep:240(3):438-47; discussion 447-50
[PubMed PMID: 15319715]
[37]
Saito Y, Omiya H, Kohno K, Kobayashi T, Itoi K, Teramachi M, Sasaki M, Suzuki H, Takao H, Nakade M. Pulmonary metastasectomy for 165 patients with colorectal carcinoma: A prognostic assessment. The Journal of thoracic and cardiovascular surgery. 2002 Nov:124(5):1007-13
[PubMed PMID: 12407386]
[38]
Even-Sapir E, Parag Y, Lerman H, Gutman M, Levine C, Rabau M, Figer A, Metser U. Detection of recurrence in patients with rectal cancer: PET/CT after abdominoperineal or anterior resection. Radiology. 2004 Sep:232(3):815-22
[PubMed PMID: 15273334]
[39]
Szyszko T, Al-Nahhas A, Canelo R, Habib N, Jiao L, Wasan H, Pagou M, Tait P. Assessment of response to treatment of unresectable liver tumours with 90Y microspheres: value of FDG PET versus computed tomography. Nuclear medicine communications. 2007 Jan:28(1):15-20
[PubMed PMID: 17159544]
[40]
Grulich AE, Poynten IM, Machalek DA, Jin F, Templeton DJ, Hillman RJ. The epidemiology of anal cancer. Sexual health. 2012 Dec:9(6):504-8. doi: 10.1071/SH12070. Epub
[PubMed PMID: 22958581]
[41]
Shiels MS, Kreimer AR, Coghill AE, Darragh TM, Devesa SS. Anal Cancer Incidence in the United States, 1977-2011: Distinct Patterns by Histology and Behavior. Cancer epidemiology, biomarkers & prevention : a publication of the American Association for Cancer Research, cosponsored by the American Society of Preventive Oncology. 2015 Oct:24(10):1548-56. doi: 10.1158/1055-9965.EPI-15-0044. Epub 2015 Jul 29
[PubMed PMID: 26224796]
[42]
Di Carlo C, di Benedetto M, Vicenzi L, Costantini S, Cucciarelli F, Fenu F, Arena E, Mariucci C, Montisci M, Panni V, Patani F, Valenti M, Palucci A, Burroni L, Mantello G. FDG-PET/CT in the Radiotherapy Treatment Planning of Locally Advanced Anal Cancer: A Monoinstitutional Experience. Frontiers in oncology. 2021:11():655322. doi: 10.3389/fonc.2021.655322. Epub 2021 Jul 1
[PubMed PMID: 34277406]
[43]
Sharma P, Singh H, Bal C, Kumar R. PET/CT imaging of neuroendocrine tumors with (68)Gallium-labeled somatostatin analogues: An overview and single institutional experience from India. Indian journal of nuclear medicine : IJNM : the official journal of the Society of Nuclear Medicine, India. 2014 Jan:29(1):2-12. doi: 10.4103/0972-3919.125760. Epub
[PubMed PMID: 24591775]
Level 3 (low-level) evidence
[44]
Tang LH, Untch BR, Reidy DL, O'Reilly E, Dhall D, Jih L, Basturk O, Allen PJ, Klimstra DS. Well-Differentiated Neuroendocrine Tumors with a Morphologically Apparent High-Grade Component: A Pathway Distinct from Poorly Differentiated Neuroendocrine Carcinomas. Clinical cancer research : an official journal of the American Association for Cancer Research. 2016 Feb 15:22(4):1011-7. doi: 10.1158/1078-0432.CCR-15-0548. Epub 2015 Oct 19
[PubMed PMID: 26482044]
[45]
Pobłocki J, Jasińska A, Syrenicz A, Andrysiak-Mamos E, Szczuko M. The Neuroendocrine Neoplasms of the Digestive Tract: Diagnosis, Treatment and Nutrition. Nutrients. 2020 May 15:12(5):. doi: 10.3390/nu12051437. Epub 2020 May 15
[PubMed PMID: 32429294]