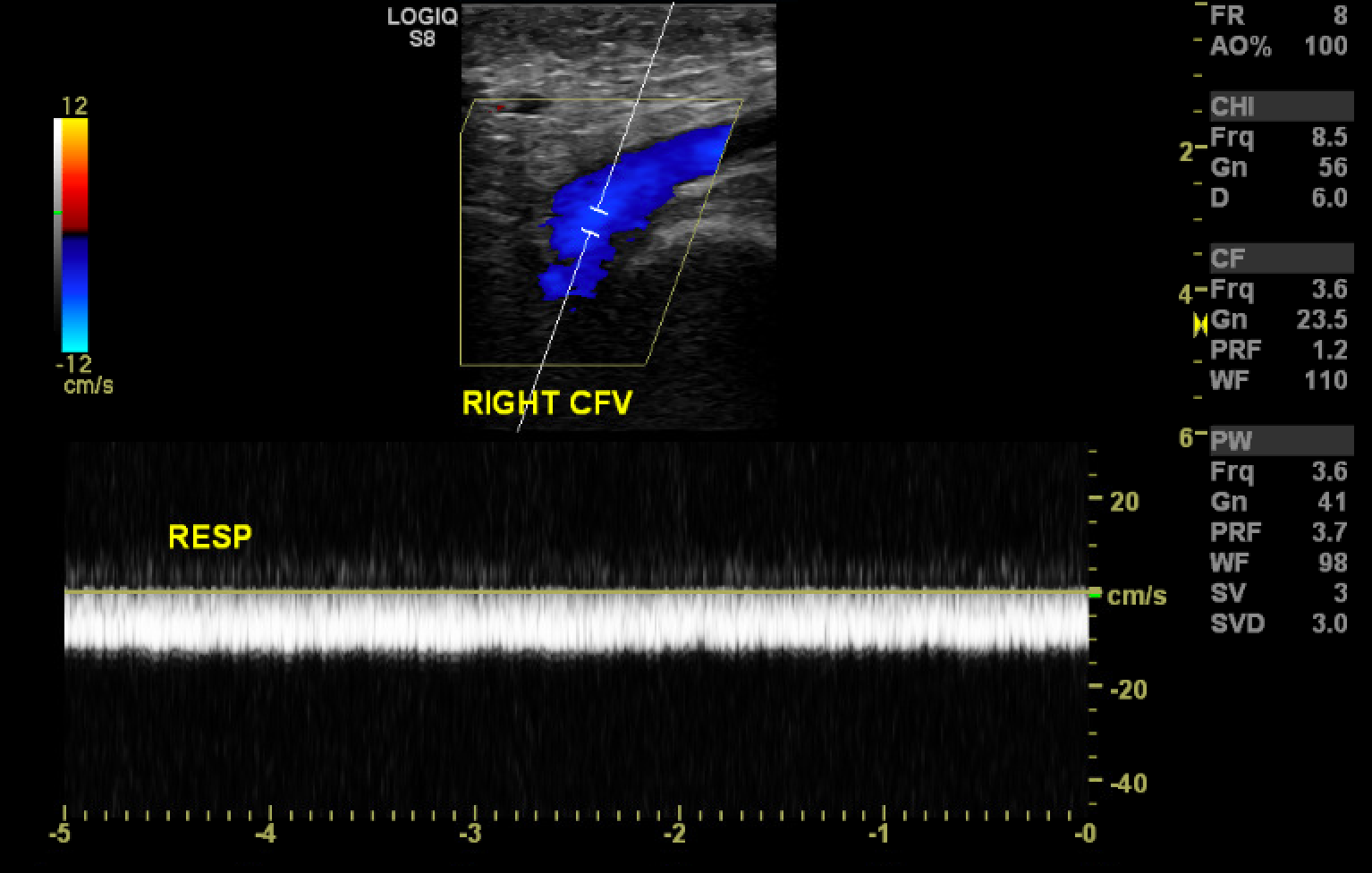
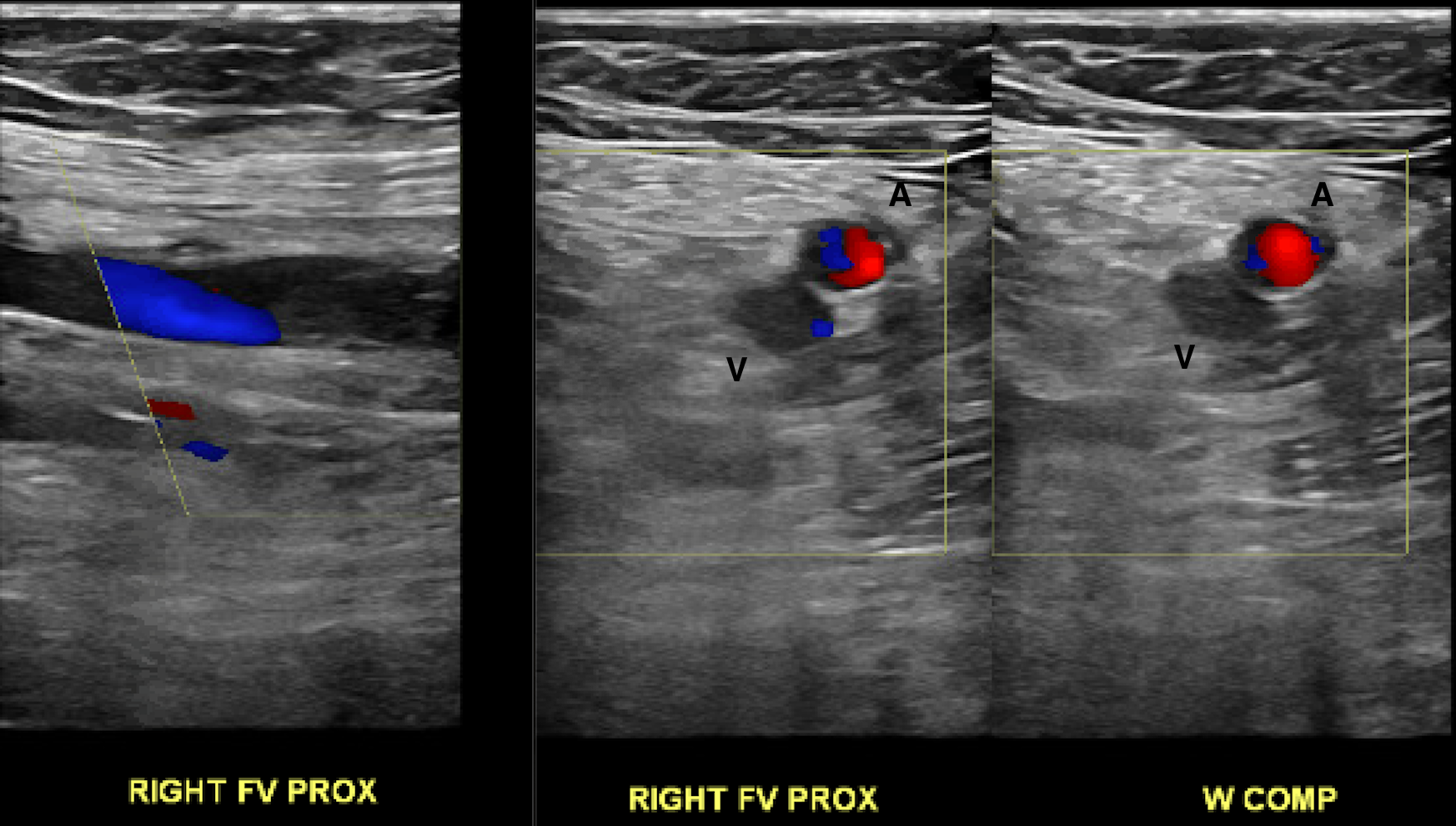
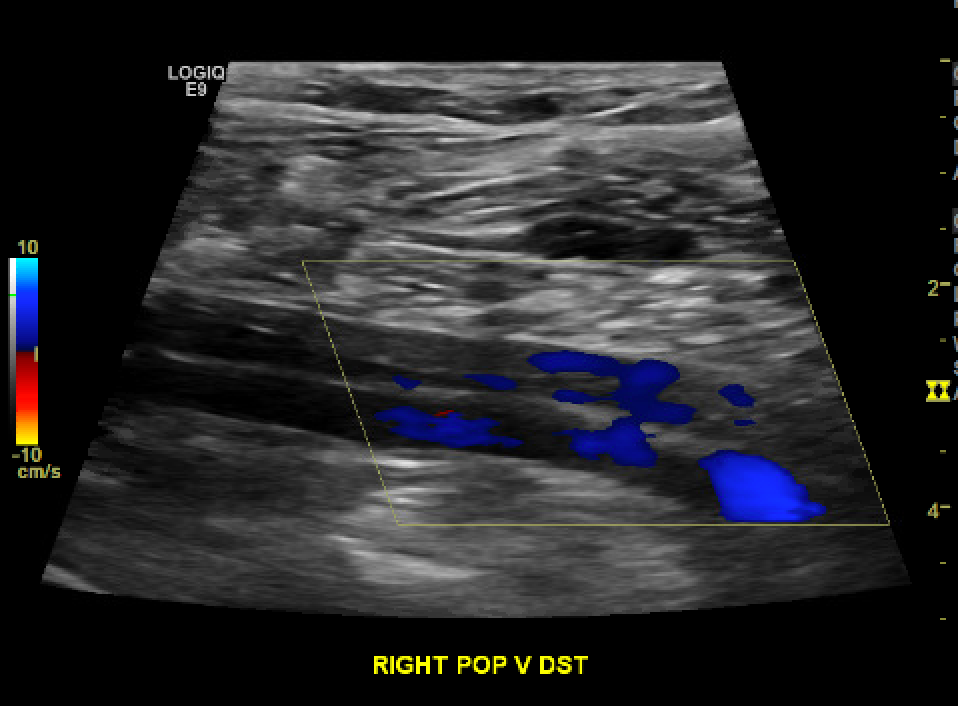
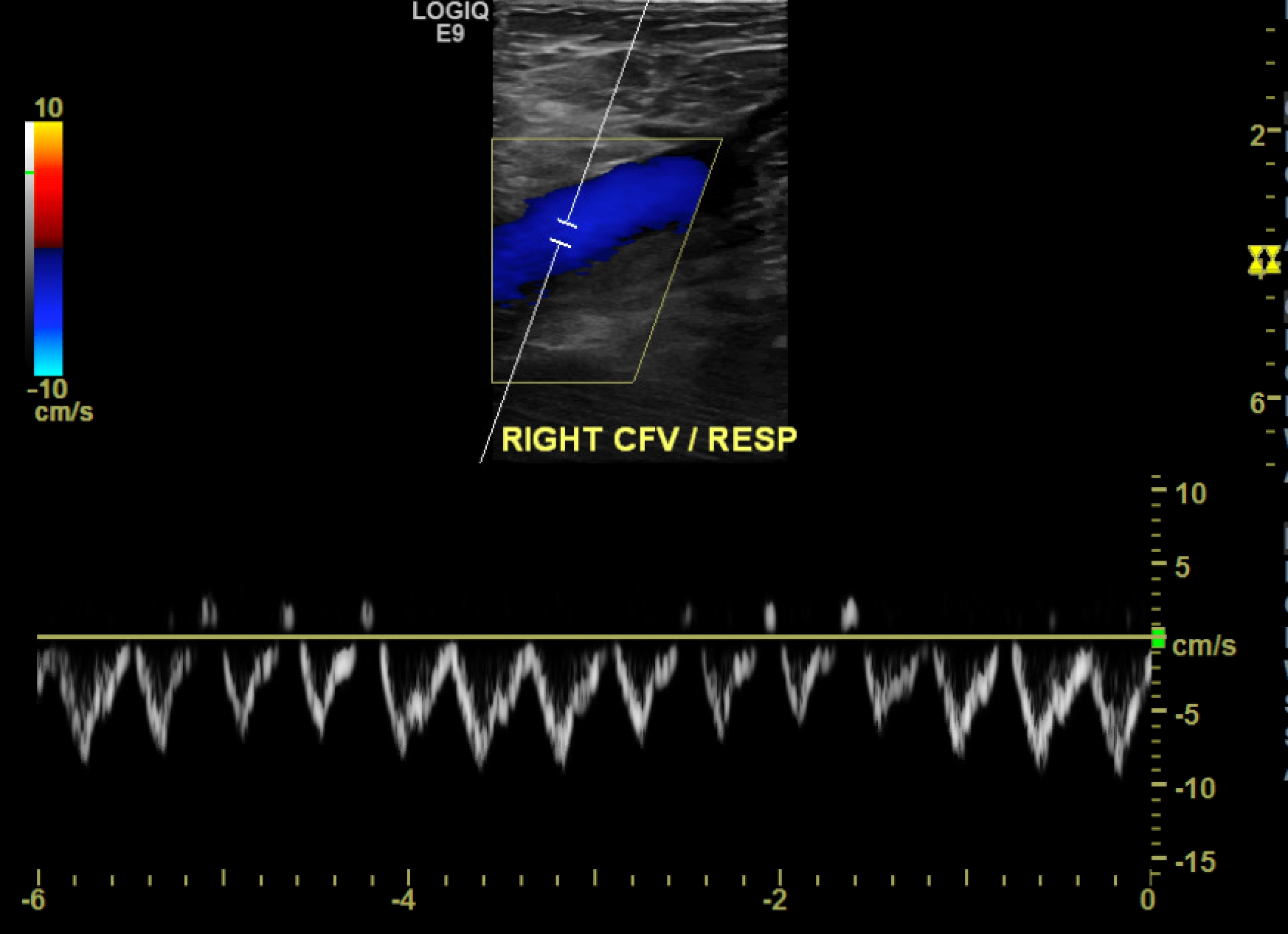
[1]
Needleman L,Cronan JJ,Lilly MP,Merli GJ,Adhikari S,Hertzberg BS,DeJong MR,Streiff MB,Meissner MH, Ultrasound for Lower Extremity Deep Venous Thrombosis: Multidisciplinary Recommendations From the Society of Radiologists in Ultrasound Consensus Conference. Circulation. 2018 Apr 3;
[PubMed PMID: 29610129]
Level 3 (low-level) evidence
[3]
Meissner MH, Duplex follow-up of patients with DVT: does it have clinical significance? Seminars in vascular surgery. 2001 Sep;
[PubMed PMID: 11561283]
[4]
Sharafuddin MJ,Sun S,Hoballah JJ,Youness FM,Sharp WJ,Roh BS, Endovascular management of venous thrombotic and occlusive diseases of the lower extremities. Journal of vascular and interventional radiology : JVIR. 2003 Apr;
[PubMed PMID: 12682198]
[5]
Hirsh J,Hoak J, Management of deep vein thrombosis and pulmonary embolism. A statement for healthcare professionals. Council on Thrombosis (in consultation with the Council on Cardiovascular Radiology), American Heart Association. Circulation. 1996 Jun 15;
[PubMed PMID: 8925592]
[6]
Liu D,Peterson E,Dooner J,Baerlocher M,Zypchen L,Gagnon J,Delorme M,Sing CK,Wong J,Guzman R,Greenfield G,Moodley O,Yenson P,Interdisciplinary Expert Panel on Iliofemoral Deep Vein Thrombosis (InterEPID)., Diagnosis and management of iliofemoral deep vein thrombosis: clinical practice guideline. CMAJ : Canadian Medical Association journal = journal de l'Association medicale canadienne. 2015 Nov 17;
[PubMed PMID: 26416989]
Level 1 (high-level) evidence
[7]
Kahn SR,Ginsberg JS, Relationship between deep venous thrombosis and the postthrombotic syndrome. Archives of internal medicine. 2004 Jan 12;
[PubMed PMID: 14718318]
[8]
Spiridon M,Corduneanu D, Chronic Venous Insufficiency: a Frequently Underdiagnosed and Undertreated Pathology. Maedica. 2017 Jan
[PubMed PMID: 28878840]
[9]
Jawien A, The influence of environmental factors in chronic venous insufficiency. Angiology. 2003 Jul-Aug
[PubMed PMID: 12934754]
[10]
Nicolaides AN,Cardiovascular Disease Educational and Research Trust.,European Society of Vascular Surgery.,,The International Angiology Scientific Activity Congress Organization.,International Union of Angiology.,Union Internationale de Phlebologie at the Abbaye des Vaux de Cernay., Investigation of chronic venous insufficiency: A consensus statement (France, March 5-9, 1997). Circulation. 2000 Nov 14
[PubMed PMID: 11076834]
Level 3 (low-level) evidence
[11]
Oklu R,Habito R,Mayr M,Deipolyi AR,Albadawi H,Hesketh R,Walker TG,Linskey KR,Long CA,Wicky S,Stoughton J,Watkins MT, Pathogenesis of varicose veins. Journal of vascular and interventional radiology : JVIR. 2012 Jan;
[PubMed PMID: 22030459]
[12]
Galanaud JP,Holcroft CA,Rodger MA,Kovacs MJ,Betancourt MT,Wells PS,Anderson DR,Chagnon I,Le Gal G,Solymoss S,Crowther MA,Perrier A,White RH,Vickars LM,Ramsay T,Kahn SR, Comparison of the Villalta post-thrombotic syndrome score in the ipsilateral vs. contralateral leg after a first unprovoked deep vein thrombosis. Journal of thrombosis and haemostasis : JTH. 2012 Jun
[PubMed PMID: 22646832]
[14]
Soosainathan A,Moore HM,Gohel MS,Davies AH, Scoring systems for the post-thrombotic syndrome. Journal of vascular surgery. 2013 Jan;
[PubMed PMID: 23182156]
[15]
Meissner MH. Lower extremity venous anatomy. Seminars in interventional radiology. 2005 Sep:22(3):147-56. doi: 10.1055/s-2005-921948. Epub
[PubMed PMID: 21326687]
[17]
Raffetto JD, Pathophysiology of Chronic Venous Disease and Venous Ulcers. The Surgical clinics of North America. 2018 Apr;
[PubMed PMID: 29502775]
[18]
Shah DM, Chang BB, Leopold PW, Corson JD, Leather RP, Karmody AM. The anatomy of the greater saphenous venous system. Journal of vascular surgery. 1986 Feb:3(2):273-83
[PubMed PMID: 3944931]
[19]
Schweighofer G,Mühlberger D,Brenner E, The anatomy of the small saphenous vein: fascial and neural relations, saphenofemoral junction, and valves. Journal of vascular surgery. 2010 Apr
[PubMed PMID: 20022210]
[20]
Baliyan V, Tajmir S, Hedgire SS, Ganguli S, Prabhakar AM. Lower extremity venous reflux. Cardiovascular diagnosis and therapy. 2016 Dec:6(6):533-543. doi: 10.21037/cdt.2016.11.14. Epub
[PubMed PMID: 28123974]
[21]
Hochauf S,Sternitzky R,Schellong SM, [Structure and function of the peripheral venous system]. Herz. 2007 Feb
[PubMed PMID: 17323029]
[22]
Migirov A, Arbor TC, Vilella RC. Anatomy, Abdomen and Pelvis: Adductor Canal (Subsartorial Canal, Hunter Canal). StatPearls. 2024 Jan:():
[PubMed PMID: 32310506]
[24]
Tolu I, Durmaz MS. Frequency and Significance of Perforating Venous Insufficiency in Patients with Chronic Venous Insufficiency of Lower Extremity. The Eurasian journal of medicine. 2018 Jun:50(2):99-104. doi: 10.5152/eurasianjmed.2018.18338. Epub 2018 Apr 30
[PubMed PMID: 30002576]
[25]
Anaya-Ayala JE, Younes HK, Kaiser CL, Syed O, Ismail N, Naoum JJ, Davies MG, Peden EK. Prevalence of variant brachial-basilic vein anatomy and implications for vascular access planning. Journal of vascular surgery. 2011 Mar:53(3):720-4. doi: 10.1016/j.jvs.2010.09.072. Epub 2010 Dec 8
[PubMed PMID: 21144691]
[26]
Yang HJ,Gil YC,Jin JD,Cho H,Kim H,Lee HY, Novel findings of the anatomy and variations of the axillary vein and its tributaries. Clinical anatomy (New York, N.Y.). 2012 Oct
[PubMed PMID: 22623347]
[28]
. AIUM Practice Parameter for the Performance of a Peripheral Venous Ultrasound Examination. Journal of ultrasound in medicine : official journal of the American Institute of Ultrasound in Medicine. 2020 May:39(5):E49-E56. doi: 10.1002/jum.15263. Epub 2020 Mar 12
[PubMed PMID: 32162338]
[29]
Scovell SD,Ergul EA,Conrad MF, Medical management of acute superficial vein thrombosis of the saphenous vein. Journal of vascular surgery. Venous and lymphatic disorders. 2018 Jan;
[PubMed PMID: 29097174]
[30]
Prandoni P,Lensing AW,Bernardi E,Villalta S,Bagatella P,Girolami A,DERECUS Investigators Group., The diagnostic value of compression ultrasonography in patients with suspected recurrent deep vein thrombosis. Thrombosis and haemostasis. 2002 Sep
[PubMed PMID: 12353067]
[31]
Baxter GM,McKechnie S,Duffy P, Colour Doppler ultrasound in deep venous thrombosis: a comparison with venography. Clinical radiology. 1990 Jul;
[PubMed PMID: 2202536]
[32]
Kim ES,Sharma AM,Scissons R,Dawson D,Eberhardt RT,Gerhard-Herman M,Hughes JP,Knight S,Marie Kupinski A,Mahe G,Neumyer M,Poe P,Shugart R,Wennberg P,Williams DM,Zierler RE, Interpretation of peripheral arterial and venous Doppler waveforms: A consensus statement from the Society for Vascular Medicine and Society for Vascular Ultrasound. Vascular medicine (London, England). 2020 Oct
[PubMed PMID: 32667274]
Level 3 (low-level) evidence
[33]
Linkins LA,Stretton R,Probyn L,Kearon C, Interobserver agreement on ultrasound measurements of residual vein diameter, thrombus echogenicity and Doppler venous flow in patients with previous venous thrombosis. Thrombosis research. 2006
[PubMed PMID: 16378830]
[34]
Rubin JM,Xie H,Kim K,Weitzel WF,Emelianov SY,Aglyamov SR,Wakefield TW,Urquhart AG,O'Donnell M, Sonographic elasticity imaging of acute and chronic deep venous thrombosis in humans. Journal of ultrasound in medicine : official journal of the American Institute of Ultrasound in Medicine. 2006 Sep
[PubMed PMID: 16929019]
[35]
Karande GY,Hedgire SS,Sanchez Y,Baliyan V,Mishra V,Ganguli S,Prabhakar AM, Advanced imaging in acute and chronic deep vein thrombosis. Cardiovascular diagnosis and therapy. 2016 Dec;
[PubMed PMID: 28123971]
[36]
Rose SC,Nelson TR, Ultrasonographic modalities to assess vascular anatomy and disease. Journal of vascular and interventional radiology : JVIR. 2004 Jan;
[PubMed PMID: 14709684]
[37]
Souroullas P, Barnes R, Smith G, Nandhra S, Carradice D, Chetter I. The classic saphenofemoral junction and its anatomical variations. Phlebology. 2017 Apr:32(3):172-178. doi: 10.1177/0268355516635960. Epub 2016 Jul 9
[PubMed PMID: 26924361]
[38]
Joh JH,Park HC, The cutoff value of saphenous vein diameter to predict reflux. Journal of the Korean Surgical Society. 2013 Oct
[PubMed PMID: 24106683]
[39]
Min RJ,Khilnani NM,Golia P, Duplex ultrasound evaluation of lower extremity venous insufficiency. Journal of vascular and interventional radiology : JVIR. 2003 Oct;
[PubMed PMID: 14551269]
[40]
Eberhardt RT, Raffetto JD. Chronic venous insufficiency. Circulation. 2014 Jul 22:130(4):333-46. doi: 10.1161/CIRCULATIONAHA.113.006898. Epub
[PubMed PMID: 25047584]
[41]
Zygmunt JA, Duplex ultrasound for chronic venous insufficiency. The Journal of invasive cardiology. 2014 Nov;
[PubMed PMID: 25364006]
[42]
Lurie F,Comerota A,Eklof B,Kistner RL,Labropoulos N,Lohr J,Marston W,Meissner M,Moneta G,Neglén P,Neuhardt D,Padberg F Jr,Welsh HJ, Multicenter assessment of venous reflux by duplex ultrasound. Journal of vascular surgery. 2012 Feb;
[PubMed PMID: 22178437]
[43]
Abenhaim L,Kurz X, The VEINES study (VEnous Insufficiency Epidemiologic and Economic Study): an international cohort study on chronic venous disorders of the leg. VEINES Group. Angiology. 1997 Jan;
[PubMed PMID: 8995345]
[44]
Christenson JT, The impact of the creation of a venous surgical centre within the department of cardiovascular surgery at a university hospital. Phlebology. 2007;
[PubMed PMID: 18268853]