Introduction
Peripheral arterial disease (PAD) affects 3% to 5% of the adult population over the age of 40.[1] PAD is associated with increased mortality, occurring concomitantly with coronary artery disease and cerebrovascular accidents.[2] PAD might present with claudication, rest pain, and or gangrene.[3][4]
Noninvasive methods are the preferred diagnostic and monitoring modalities for PAD. Patients with a history and physical examination compatible with PAD are first evaluated with Duplex ultrasonography (DUS), which does not require contrast exposure and produces portable and reproducible images. However, in patients with obesity, the aortoiliac bifurcation may be obscured by bowel gas, so results must be interpreted cautiously. Arterial DUS provides two-dimensional structural images and Doppler ultrasonic signals over time. These signals can be analyzed with spectrum analysis and color flow velocity mapping for better documentation.
The results of DUS are usually interpreted in conjunction with limb-pressure measurements to optimally categorize arterial hemodynamics and functional impairment. The accurate and precise interpretation of velocity spectra measured by DUS is essential to successfully applying the Duplex technique.[5]
The diagnosis of PAD relies on noninvasive arterial tests providing physiological and morphological information. Physiological tests include segmental pressures, ankle- and toe-brachial indices, pulse volume recordings, exercise tests, segmental volume plethysmography, transcutaneous oxygen assessments, and photoplethysmography. Morphological tests, including computed tomography (CT), magnetic resonance imaging (MRI), and catheter-based angiography, are ideal for evaluating preprocedural anatomic configurations in individuals with already established vascular disease. Peripheral arterial DUS provides a unique spectrum of details, including the anatomical location of the lesion, flow velocity, and volume.[6]
DUS combines conventional B-mode imaging with pulsed Doppler flow detection techniques for spectral analysis.[7] DUS accurately determines the location and degree of arterial stenosis while differentiating stenosis from an occlusion.[8] DUS provides comprehensive anatomic and hemodynamic data crucial to planning revascularization.[9] DUS is also used to monitor patients after angioplasty, stent replacement, and bypass grafting.[10]
Anatomy and Physiology
Anatomy of the Lower Extremity Arterial Supply
The abdominal aorta bifurcates into the common iliac arteries at the L4-L5 level. The common iliac arteries traverse anterior to the common iliac veins. Each common iliac artery bifurcates into an internal and external iliac artery at the level of the sacroiliac joint.
The external iliac artery is the larger of the two terminal branches of the common iliac artery. It courses through the pelvis along the medial border of the psoas muscle. Prior to coursing under the inguinal ligament, at which point the external iliac artery becomes the common femoral artery, the external iliac artery gives off the inferior epigastric artery medially and the deep iliac circumflex artery. Eventually, the common femoral artery divides into the superficial and deep profunda femoris artery.
The superficial femoral artery is the main arterial supply to the thigh. This artery runs distally through the Hunter or adductor canal and becomes the popliteal artery as it emerges. The popliteal artery bifurcates into the anterior tibial artery and the tibioperoneal trunk at the proximal calf.
The anterior tibial artery supplies the anterior compartment of the lower leg and terminates at the ankle as the dorsalis pedis artery. The tibioperoneal trunk bifurcates into the posterior tibial artery and common peroneal artery. The posterior tibial artery supplies the posterior calf, courses posterior to the medial malleolus, and forms the plantar arteries of the foot. The peroneal artery supplies the musculature of the lateral calf and courses posterior to the fibula.[8]
Noninvasive Imaging of the Lower Extremity Arterial Supply
DUS and spectral Doppler waveform evaluation measure blood flow velocity in the appropriate arterial segments. In the lower extremity, a detailed evaluation of the common femoral artery, proximal and mid-superficial femoral artery, distal superficial femoral artery and popliteal artery above the knee, and popliteal artery below the knee is necessary. If deemed clinically necessary, imaging of the iliac vessels, profunda femoris artery, tibioperoneal trunk, anterior tibial artery, posterior tibial artery, and dorsalis pedis artery should also be performed.
B-mode imaging displays a two-dimensional depiction of the arterial lumen and wall, evaluating any plaque characteristics. It is recommended to adjust grayscale settings for optimal visualization of intraluminal lesions. Utilizing color Doppler can further enhance the detection of arterial lesions by detecting visual narrowing and color differences and assist in placing the sample volume for spectral Doppler assessment.
The color Doppler parameters are fine-tuned to distinguish between normal and abnormal flow patterns. The pulse repetition frequency (PRF) plays a crucial role in determining the amount of color displayed and the filling of the arterial lumen. The PRF is adjusted so that normal laminar flow appears as a consistent area of color. When stenosis is present, the color flow pattern changes and is identified by either aliasing or desaturating (whitening) the color display at the site of luminal narrowing. Longitudinal spectral Doppler images measure velocity, and the angle between the blood flow direction and the ultrasound beam direction should be ≤60 degrees. Velocity measurements taken from images using larger angles were found to be less accurate.
Doppler sonography relies on changes in signal frequencies reflected from moving objects, specifically red blood cells, which change in proportion to the target's velocity. Vascular sonography has made great strides with the introduction of duplex scanners, allowing for spectral analysis that identifies a full range of frequencies (blood flow velocities) present in the arterial waveform during a single cardiac cycle. The Doppler waveform morphology is influenced by multiple physiological factors, including metabolic demands of the tissue, changes in resistance and pressure, wave reflection, and propagation, both at the site being examined and proximal and distal to it. These factors all contribute to the unique waveform patterns.
In DUS, some distinct features help distinguish between arterial and venous structures. Arteries can be identified by their rounded shape with visible, discrete, and partially compressible walls. Additionally, arteries are generally smaller in size.[11]
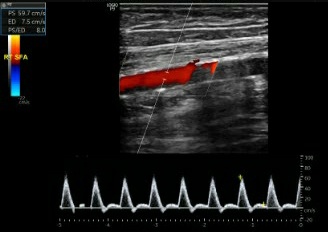
A typical peripheral arterial waveform appears as a narrow, sharply defined tracing. This indicates that all red blood cells move at a similar speed throughout the cardiac cycle. The typical waveform configuration is triphasic, with 3 components corresponding to the different phases of arterial flow (see Image. Normal Triphasic Arterial Waveform). These components include a rapid antegrade flow that peaks during systole, a transient reversal of flow during early diastole, and a slow antegrade flow during late diastole. The high-velocity forward flow during systole causes the rapid antegrade flow component. In contrast, the early diastolic reverse flow component is caused by distal resistance at the level of small caliber vessels and capillaries. The late diastolic component in this phase is due to the small amount of forward flow and the elastic recoil of vessel walls.[10]
Real-time ultrasonography uses sound waves to create images and measure blood velocity. Two major ultrasound modes are used for vascular imaging: the B-mode, which provides a grayscale two-dimensional image, and the Doppler mode, which can be continuous or pulsed. Other less common modes include power Doppler, three-dimensional, and contrast-enhanced ultrasound. In the B-mode, bright dots on the image represent different amplitudes of echo signals, with their brightness indicating their strength. However, the quality of the B-mode image can be affected by the tissue it passes through, which can either attenuate or scatter the sound waves.
To obtain high-quality images, it is essential to consider both the insonation angle and the frequency of the sound waves being used. Using high-frequency transducers at a 90-degree insonation angle is optimal for shallow structures. The Doppler mode can be combined with the B-mode to identify vascular structures. This mode detects the change in sound frequency caused by the movement of red blood cells, which is then reflected in the generated velocity. The color mode enhances this effect, but the colors do not determine whether the flow is arterial or venous. Instead, the flow towards or away from the transducer is displayed in red and blue.
The arterial velocity waveform typically exhibits three phases, known as triphasic. When the waveform is multiphasic or triphasic with a narrow spectral width throughout the pulse cycle, it indicates that the movement of red blood cells is consistent in speed and direction, with a laminar flow pattern. This includes a sharp peak during systole, a downward peak in early diastole, and forward flow in late diastole. However, the late diastolic wave decreases or disappears when peripheral resistance develops.
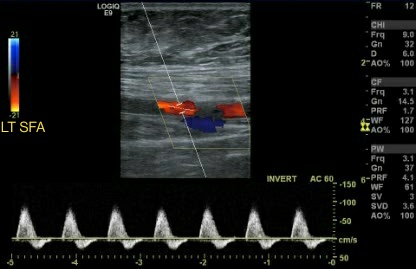
Doppler velocity waveforms can distinguish between single and multilevel arterial occlusive disease. A biphasic arterial Doppler velocity waveform has a quick upstroke followed by a relatively fast downstroke, with flow above the baseline during all phases and brief periods of antegrade flow during diastole separating each wave (see Image. Biphasic Arterial Waveform). Non-sharp, rounded peaks, no flow reversal, and a faint upstroke and downstroke characterize the monophasic Doppler waveform, typically seen in multilevel obstructive arterial disease (see Image. Monophasic Arterial Waveform).
The velocity waveforms produced are linked to the probe's location with respect to the narrowed artery. When the narrowing is closer to the beginning of the artery, the systolic peaks become more rounded, the reversed flow component disappears, and the late diastolic phase is prolonged. However, the absence of a reversed flow component is not solely due to reduced peripheral vascular resistance and should be interpreted cautiously. It could result from compensatory poststenotic vasodilation or be a normal finding in older patients.
The pitch of duplex signals is linked to the change in the speed of blood flow. This means that high-pitched and thumping sounds are typically heard in areas with stenosis or near the stenotic lesion. When peripheral arteries develop any flow disturbance or stenoses, the flow inside the blood vessels becomes turbulent and is no longer laminar. As a result, instead of a clear tracing, the Doppler waveform displays spectral broadening.
As peripheral arterial disease progresses, the normal triphasic flow is replaced by biphasic flow due to arterial fibrosis, which reduces the elastic recoil. If the disease worsens or is at the site of severe stenosis or occlusion, the early diastolic flow reversal disappears, resulting in a monophasic waveform. The Bernoulli principle states that when the diameter of an artery decreases, as in arterial stenosis, the flow velocity through the stenosis should increase. However, when the stenosis severity exceeds 95%, the velocity decreases.
The Doppler waveform is a valuable tool for identifying and locating arterial obstruction. A delayed systolic upstroke indicates a flow-limiting lesion near the recording site, which can result in reduced resistance and pressure drop distal to the stenosis. As a result, there is increased diastolic flow throughout the cardiac cycle. However, high-grade stenosis or occlusion can increase resistance and decrease antegrade diastolic flow or reflected wave. The severity of arterial compromise is reflected in a continual rise of peak systolic velocity (PSV) and end-diastolic velocity (EDV) to a threshold value, suggesting a pre-occlusive lesion.[12]
To obtain accurate velocity measurements, it is essential to have precise and valid Doppler shift records. When measuring velocity, the angle of the probe in relation to the flow direction or angle of insonation, as well as its position relative to the stenotic lesion, should be considered. It is recommended to keep the angle of insonation between 30 and 70 degrees, with the ideal angle being 60 degrees. Additionally, the ultrasound probe should be positioned at or distal to the suspicious lesion, and the characteristics of the color flow imaging can help determine the lesion's location.
To determine the degree of stenosis, the velocity ratio is assessed. The peak systolic velocity ratio between the stenotic lesion and the unaffected section of the vessel is used to calculate this ratio. A ratio of 1.5 to 2 indicates 30% to 49% stenosis, while ratios of 2 to 4 and above 4 indicate 50% to 75% and over 75% stenosis, respectively. The accuracy of limb arterial DUS varies depending on the scanned segment and levels involved. However, DUS is highly accurate in grading peripheral arterial disease, with a relative accuracy of over 80% compared to CT angiography. According to another classification, a peripheral arterial lesion is significant if it causes over 50% stenosis in the presence of an atherosclerotic plaque. Predictable DUS features include a PSV of over 200 cm/s and a PSV ratio of over 2.4 (PSV in the stenotic site to the proximal non-stenotic area). However, stenotic lesions with a PSV of over 300 cm/s and a ratio of over 3.5 are classified as severe.
To interpret the PAD severity, the following indexes are provided by duplex-acquired velocity spectra, including acceleration time, pulsatility index (PI), and maximum spectra velocity measured at PSV.[5]
Equipment
Ultrasonography utilizes various techniques to assess blood flow. These techniques include color Doppler imaging, pulsed Doppler, spectral analysis, power Doppler imaging, B-flow imaging, and high-resolution B-mode imaging for arterial anatomy.
For peripheral arterial ultrasound, using a real-time scanner and a transducer with pulsed and color Doppler capabilities is recommended. A linear array transducer is preferred over a curved array transducer, especially if it allows adequate penetration.
An average-sized adult can be examined using a 5-MHz linear array transducer. Linear 3.5-MHz probes are preferred for iliac arterial intervention, and 5-MHz probes are preferred for common femoral arterial interventions, including percutaneous transluminal angioplasty (PTA). Higher frequency probes of 7.5 to 10 MHz are used for thin or small individuals when identifying superficial vessels. In contrast, lower frequency probes of 3 MHz are used for larger patients to identify deep vessels.
Studies suggest that DUS instrumentation requires the application of both curved and linear array transducers, utilizing 3.5- to 7-MHz imaging frequencies for comprehensive high-resolution images of the abdominal and lower extremity arteries.
Doppler ultrasound displays may be color flow Doppler imaging or spectral Doppler imaging. While the former demonstrates mean flow velocity distribution as a color-encoded map integrated over the gray-scale B-mode tissue image, the latter shows the time-varying flow velocity distribution within a specific sample volume. Spectral Doppler imaging provides further information, including peak velocity during the pulse cycle, spectral content, and range of velocities.
Technique or Treatment
Duplex Ultrasonography of the Lower Extremity
When conducting lower extremity DUS, the patient should be supine with a slight outward rotation of the hip and leg. DUS begins at the inguinal crease, where the transducer is placed horizontally over the common femoral artery. A "Mickey Mouse" shape is a common finding of the superficial and deep femoral arteries and the femoral vein located just below the crease. In contrast, a longitudinal scan reveals an inverted Y-shape in the common femoral, superficial, and deep femoral arteries.
The scan should continue by moving the transducer distally. Examination of the popliteal artery is performed with the knee in flexion. Begin the scan at the popliteal fossa and progress proximally toward the Hunter canal. The left lateral or prone patient positions may facilitate a full evaluation of the arteries of the popliteal fossa, including the popliteal, posterior tibialis, and peroneal arteries.
The popliteal artery can be located in the center of the popliteal fossa, between the medial and lateral heads of the gastrocnemius muscle. Examination of the posterior tibialis artery begins distally, behind the medial malleolus. The peroneal artery is located on the lateral side of the posterior calf along the fibula. The anterior tibialis artery may be evaluated by following the arterial course from the distal end at the neck of the talus and moving proximally or by beginning at the proximal arterial end at the tibiofibular plane and scanning distally.
Occlusive Disease
To evaluate characteristics such as arterial diameter, intima-media thickening, and atherosclerotic plaque composition, it is recommended to obtain angle-corrected spectral Doppler waveforms from longitudinal images proximal, at, and distal to sites of suspected stenosis using a 90-degree imaging angle.[5][14] A Doppler beam angle ≤60 degrees relative to the transducer insonation beam and the arterial wall of interest is recommended for pulsed spectral Doppler recordings.[16]
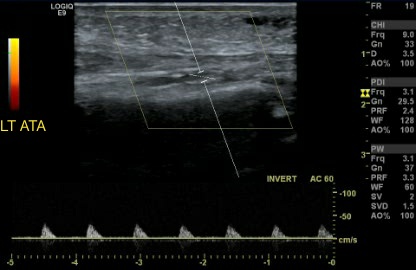
When diagnosing hemodynamically significant stenoses of 50% to 99%, it is necessary to double-check that the PSV at the site of the stenosis is at least twice that of a more proximal segment. This ratio of PSVs provides a more accurate evaluation of peripheral arterial stenosis than absolute PSV measurements. The jet will disperse into eddy currents after a significant stenosis, leading to a substantial decrease in PSV. The waveform will lose its rapid systolic upstroke and demonstrate a delay to peak systole, which is referred to as a tardus parvus pattern (see Image. Monophasic Tardus Parvus Waveform) Identifying this waveform is crucial as it indicates proximal high-grade stenosis or frank occlusion. If occlusion occurs, there will be no flow within the arterial segment. Without collaterals, the segment proximal to the occlusion will display high-resistance waveforms. However, if collaterals are present, a low-resistance, low-velocity tardus parvus flow pattern will be found in the reconstructed segment. It is also essential to document the estimated lengths of the stenosed or occluded segments during the evaluation.
Pseudoaneurysms and Arteriovenous Fistulae
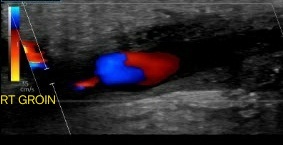
Evaluation of pseudoaneurysms should include a notation of their size, the measurements of the residual lumen, and the length and width of the surrounding channel. This can be achieved by utilizing grayscale and color Doppler imaging. The latter can reveal the unique 'yin-yang' sign caused by the swirling blood within the affected area (see Image. Pseudoaneurysm).
A femoral pseudoaneurysm typically exhibits a unique DUS feature wherein the stalk of the aneurysm displays a "to-and-fro" flow pattern while the sac empties during the diastolic and systolic phases. The flow pattern appears bidirectional when examining the communicating channel using spectral Doppler waveforms. The waveform also displays a rapid systolic upstroke, an exaggerated deceleration, and an elongated reverse flow component.[12]
Arteriovenous fistulae (AVF) are seen less frequently. To assess AVF using DUS, obtaining a spectral Doppler waveform from the artery located proximal, at, and distal to the fistula site is crucial. Additionally, flow within the fistula and the draining vein should be documented. Color Doppler is useful in identifying the level of communication as the flow disturbance in the fistula creates color in the adjacent soft tissues due to pressure changes and transmitted vibrations, termed a color bruit. Blood flow from a high-pressure artery to a low-pressure vein results in spectral broadening, increased systolic and diastolic velocities, and continuous forward flow throughout the cardiac cycle.
When examining the cardiac cycle within an AVF, spectral Doppler may show increased flow within the vein, indicating arterialization. To detect AVF formation, look for a high-velocity flow jet with a peak systolic velocity greater than 300 cm/s. In cases of femoral artery cannulation, the external iliac artery velocity spectra may show increased PSVs, and altered flow signal proximal to the AVF.
While evaluating peripheral aneurysms, their location and widest diameter of the aneurysm should be documented on grayscale images. If present within the aneurysm, their patency and intraluminal thrombus should be documented using color Doppler.[17]
Enhancing Healthcare Team Outcomes
PAD requires an interprofessional team approach due to the complex nature of this disease which involves multiple organs. A team approach has been found to significantly improve treatment outcomes, prevent disease progression, and minimize the risk of significant sequelae.
The interprofessional team caring for patients with PAD involves practitioners skilled in the medical management of PAD, wound care experts, and the revascularization team. Medical management of PAD is provided by primary care practitioners, cardiologists, endocrinologists, infectious disease teams, and dietitians. Wound care is provided by podiatrists, vascular surgeons, nursing staff, orthopedics, and the plastic surgery team. Ultrasound technicians provide the scans themselves, which form the basis for directing clinical decision-making. Nurses can also provide valuable assistance in patient evaluation, counseling, and assisting during revascularization procedures. This interprofessional approach will yield the best patient results.
Interventional radiologists, cardiologists, and the vascular surgery team participate in revascularization. A complete team should include at least one member from each specialty to treat patients diagnosed with PAD or critical limb ischemia. By bringing together the knowledge of different team members and employing effective communication, optimal patient-centered care can be provided, and the limb salvage rate can be increased.[20]