Continuing Education Activity
Keratoconus (KC) is one of the most common corneal ectasia, characterized by progressive, non-inflammatory changes in corneal stromal collagen and manifests as the protrusion and alteration of the central and paracentral cornea. The pathology usually progresses in the first three decades of life and, if not managed on time, can result in a drop in visual acuity. The management of keratoconus is governed by many factors such as age, stage of keratoconus, schiempflug imaging characteristics, and visual acuity. The various management options include spectacles, contact lenses, corneal collagen cross-linking (CXL)with riboflavin, Intacs, penetrating keratoplasty, and deep anterior lamellar keratoplasty. CXL is a technique of strengthening the corneal tissue by using riboflavin and ultraviolet-A light to halt the progression of progressive keratoconus. In CXL, riboflavin act as a photosensitizing agent, and UVA improves the formation of intra and interfibrillar covalent bonds by oxidative photosensitization. This activity describes the collagen cross-linking procedure's role in managing keratoconus. It highlights a number of clinical factors that should be considered in patients who undergo collagen cross-linking.
Objectives:
- Describe the common clinical and topographic features of keratoconus.
- Summarize the mechanism of action of collagen crosslinking.
- Outline the indications for the collagen crosslinking procedure.
- Review potential outcomes of the procedure in the available literature.
Introduction
Keratoconus (KC) is one of the most prevalent corneal ectatic disorders characterized by progressive, non-inflammatory changes in stromal collagen structure and usually results in protrusion and alteration of the central and paracentral cornea.[1] The etiology of this condition remains unknown; however, several ocular and systemic associations exist, like Leber congenital amaurosis, atopy, Down syndrome, and the connective tissue disorders of Ehlers-Danlos and Marfan syndromes. Presentation is typically in the second or third decade of life with features of progressive myopia and astigmatism. The initial presentation is unilateral; however, both eyes eventually become involved.[2]
On examination, several eponymous clinical signs may increase the suspicion for KC. Munson’s sign is a V shape bulging of the lower eyelid on downgaze. A slit-lamp examination may reveal Vogt striae: fine, vertical, stromal stress lines, and a Fleischer ring: a ring-like configuration of epithelial iron deposits. Distant direct ophthalmoscopy reveals a characteristic “oil-droplet” reflex, and retinoscopy can demonstrate a characteristic scissoring reflex.
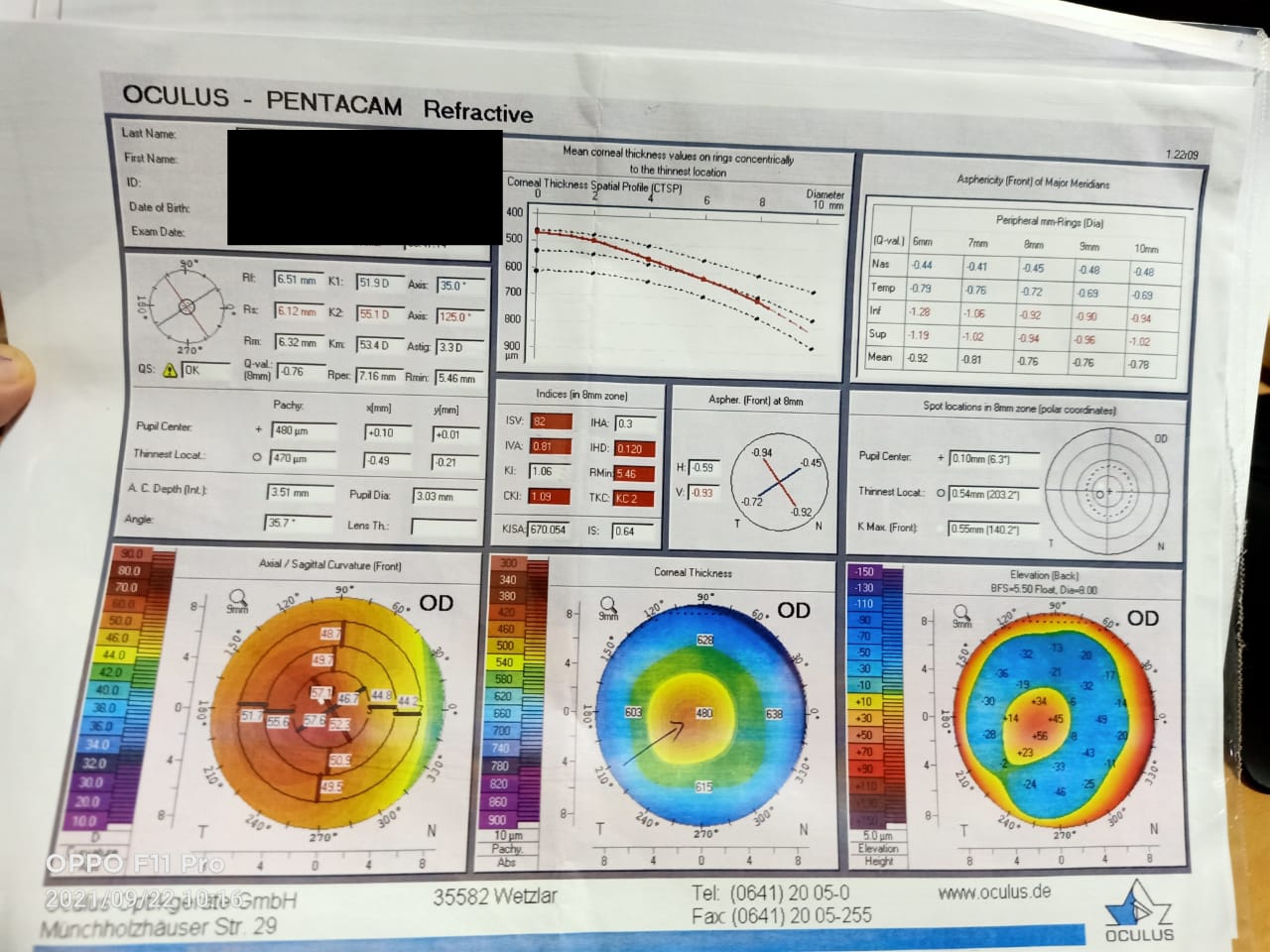
Placido-disc topography, Scheimpflug imaging, and optical coherence tomography allow for detecting subtle changes in corneal topography, tomography, and epithelium changes associated with KC. A well-known classification system is an Amsler-Krumeich system which uses the patient’s refractive error, central keratometry readings, central corneal thickness, and the presence or absence of scarring. Notably, the Amsler-Krumeich system does not utilize corneal topographic values. Various topographic indices have been proposed for diagnosing preclinical KC (forme fruste keratoconus) and clinical KC. Rabinowitz suggests the following topographical characteristics of KC: increased areas of keratometric readings surrounded by areas of reduced corneal power, inferior-superior symmetry, and skewed radial axes.[3] The newer Scheimpflug imaging-based Pentacam system (Oculus GmbH, Wetzlar, Germany) utilizes the Belin/Ambrósio Enhanced Ectasia Display (BAD) to screen for KC using maximal keratometry, anterior and posterior elevation, and tomographic thickness data.[4]
Treatment of early keratoconus involves prescribing spectacles to improve vision, but as the disease progresses, rigid gas-permeable contact lenses are required. In a small but significant proportion of patients, disease progression may require eventual corneal transplantation. Several new therapeutic options have emerged, including refractive, optical, and lamellar surgery, which slow the progression of the disease and/or delay more intensive treatment. Collagen crosslinking (CXL) with ultraviolet A (UV-A) light and riboflavin (vitamin B2) is a relatively new treatment that reportedly slows the advancement of the disease in its early stages.[5]
CXL was introduced to clinical practice in the late 1990s and has since completely modified conservative management of progressive corneal ectasia. CXL utilizes riboflavin as a photosensitizer, which, when exposed to longer wavelength UV-A, induces chemical reactions in the corneal stroma and ultimately results in the formation of covalent bonds between the collagen molecules. This collagen crosslinking increases the tensile strength and rigidity of the cornea, preventing further thinning and ectasia.[5]
Anatomy and Physiology
The cornea comprises distinct, densely placed, multi-scale structures with fibers of distinct patter arranged in various layers. Each layer is composed of distinct components having variations in collagen density, elastin, fibronectin, laminin, and proteoglycan.[6] These components give rise to different mechanical strengths due to various mechanical properties. The cornea is composed of 6 layers which are 550 um thick. A healthy cornea can transmit approximately 80% of light in the visible spectrum.[7] The cornea is densely packed with proteins called collagen. The collagen in the cornea resides for approximately 2-3 years. The corneal transparency is maintained by the regular packing of proteins with a proteoglycan-rich matrix, allowing the light to pass unimpeded.[8] Collagen crosslinking help in the formation of bonds between the polymeric chains of proteins. In biological tissue like cornea, these bonds strengthen the tissue, making them more resistant to mechanical degradation and deformation.[9]
The anterior-most layer over the corneal surface is the tear film composed of lipid and aqueous components, and the protein layer is typically 3 um thick in healthy subjects. The tear film helps provide lubrication and protection through soluble factors and maintains a smooth optical surface.[10] The next layer is the epithelial layer which lacks a continuous protein framework. Below the epithelial layer is an epithelial basement membrane which is 0.3 um thick and made up of collagen and laminin. This is followed by Bowman's layer, an acellular layer measuring 17 um and made up of randomly distributed collagen fibers.[11] Below Bowman's layer is the stroma, which makes up the bulk of corneal tissue. The stroma is 500 um thick, but the thickness can vary between individuals. The stromal is made up of densely packed collagen fibrils which are organized into lamella, and these run approximately parallel to the corneal surface. The lamellar fibers in the central cornea are approximately 200 in number and stacked one over the other. These lamellae are dispersed at varying angles throughout the cornea.[7]
The spacing between the lamella and collagen fibrils is essential for maintaining corneal transparency, and the proteoglycans maintain this spacing. The stroma is acellular, and one keratocyte is found in every 50,000 cubic microns. The Dua's or Pre-Descemet's layer is nearly identical to adjacent stromal tissue. There is a difference in proteoglycan distribution due to the higher density of lamella and more spacing between the collagen fibrils.[12] The next is the acellular fibrous layer formed by the endothelial cells, which is 3 um thick. The final is the monolayer of endothelial cells, which lacks a continuous protein network. It is imperative to understand the structure and constituents of these layers so that some preliminary hypotheses can be drawn.[7] The hypotheses revolve around the common properties of macromolecules. The macromolecules such as collagen, laminin, fibers, and fibronectin are responsible for forming strong fibers and a good network, thus serving as a tissue scaffold. Elastin has nearly similar properties to collagen and can tolerate significant strain without causing permanent deformation. Elastin is majorly found in tissues that are load-bearing or conserve mechanical energy.[13]
The different macromolecules have varied subunits that point to other mechanical functions or different evolutionary developmental origins. Another group of molecules is glycosaminoglycans (GAG) which attract and retain water, absorb the shock, and impact tissue viscosity.[14] Proteoglycans are a group of proteins in which GAG is attached to one or more core proteins, retaining a similar function to other GAGs. Based on this knowledge, hypotheses about each corneal later can be made. The tear film is a liquid interface with no major polymeric components. Hence it is thought to have little direct mechanical impact on the cornea. The epithelial and endothelial cell layers also lack any regular protein framework; therefore, they contribute little mechanical strength to the cornea.[15]
The endothelial pump plays a significant role in maintaining hydration of the cornea, which also plays a crucial role in maintaining corneal mechanics. The stroma also contributes to the major strength of the cornea. The interweaving corneal lamellae also provide resistance to the tensile strength of the cornea. The degree of collagen fibril alignment and the fibril diameter can vary; it will be essential to understand that the mechanical properties will also vary with the depth. Another critical point of consideration is lateral heterogeneity in the cornea. This fibril orientation provides a distinct lateral pattern at varying depths in the cornea. This is important because keratoconus may manifest in varied ways due to tissue structure, and surgical planning will benefit by taking these heterogeneities into account.[16]
Indications
Keratoconus, with evidence of progression, is the most common indication for CXL. Other indications include pellucid marginal degeneration, Terrien marginal degeneration, and post-refractive surgery (such as LASIK, PRK, or radial keratotomy) ectasia. The Global Delphi Panel of Keratoconus and Ectatic Diseases recognized the lack of set criteria for documenting disease progression and suggested that two of the following three parameters should be considered when considering progression: steepening of either the anterior or posterior corneal surfaces or corneal thinning.[17]
Many studies have based their criteria based on an increase in Kmax value, myopia and/or astigmatism, mean central K-readings, and a decrease in mean central corneal thickness.[18]
Not every cornea with keratoconus needs to undergo CXL; simple spectacle correction and rigid contact lenses form the basis of conservative therapy. CXL is not indicated for stable KC, as might be the case in older eyes that naturally have stiffer corneas due to age-related changes.
Contraindications
The contraindications to standard CXL treatment include a corneal thickness of fewer than 400 microns and prior herpetic ocular infection. Traditionally, corneal thickness less than 400 microns is considered a contraindication for CXL, but hypo osmolar CXL can be performed when corneal thickness varies from 370 to 400 microns. Other contraindications are concurrent ocular infection, presence of severe corneal scarring, corneal opacity, neurotrophic keratitis, severe dry eye, history of poor epithelial wound healing, autoimmune disorders, and pregnancy.[18]
Price et al., in their analysis of CXL in microbial keratitis, having negative bacterial or fungal cultures but positive for herpes simplex, showed that patients developed dendritic keratitis after undergoing CXL. There have been reports of herpetic keratitis after CXL.[19] Kymionis et al. reported a case of a young woman who underwent CXL and five days later presented with geographic keratitis and anterior uveitis. Similarly, Qarni and Harbi reported two cases of patients who underwent CXL and later presented with dendritic keratitis in the early postoperative period. Hence, past history of herpetic keratitis is a contraindication for CXL, and viral keratitis can also develop without prior history of keratitis.[20]
Equipment
The various equipment and instruments needed to perform CXL are
- Conjunctival forceps
- Diluted absolute alcohol
- Calliper
- Ink pen marker
- Hockey stick blade
- Crescent blade
- Riboflavin
- Ultraviolet light
- Hand-held slit lamp
- Normal saline
- Saline cannula
In the case of accelerated CXL, an accelerated CXL machine is needed.[21]
Personnel
For performing CXL, a combined effort of a team of Ophthalmic surgeons, an assistant OT nurse, mid-level ophthalmic personnel (MLOP), and a dedicated counselor is needed. The surgeon performs the surgery, the assistant nurse helps in patient preparation and provides instruments for the surgery, the MLOP helps in patient movement, and the counselor helps in guiding the patient pre-operative and postoperatively.[22]
Technique or Treatment
Dresden Protocol
As Wollensak et al. described, the Dresden protocol is considered the "conventional" CXL treatment protocol. This technique is named Dresden protocol because it was initially designed at the University of Dresden (Germany). Till today, this is the standard protocol followed at majority centers. Also known as the 'epi-off' protocol, this involves the removal of central 8-9 m epithelium followed by applying 0.1% riboflavin solution followed by 30 minutes of UVA radiation of wavelength 370nm and power 3mW/cm.[5] Although clinical outcomes of this technique have been reported primarily in prospective or retrospective case series, the medium-term results (6-26 months) are very favorable.[5][23] The Dresden group, in 2015, published their 10-year follow-up results of 34 eyes, demonstrating long-term stability and a good safety profile.[23][24]
Thinner corneas are more susceptible to radiation damage to the endothelium and hence are unsuitable for epi-off CXL. Additional protection can be provided to the endothelium by either retaining the epithelium or using hypo-osmolar riboflavin to increase stromal thickness during the radiation exposure stage. Riboflavin penetration through tight epithelial junctions can improve using chemical enhancers like benzalkonium chloride or EDTA. This "epi-on" protocol reduces the risk of complications associated with the "epi-off" protocol. There are conflicting reports about the efficacy of this procedure. [18]
A few modifications of the Dresden protocol, which have been used, are partial reepithelialization, with short- and medium-term results stabilization. But the effectiveness of this technique over the conventional Dresden protocol is still questionable.[18] The other modification of this protocol is the induction of mechanical compression of the tissue by applying a semi-scleral rigid contact lens with a flatter curvature (back radius of 11 mm) to the cornea after putting the riboflavin. This is followed by riboflavin application every 5 minutes beneath the contact lens using UVA irradiation with a blunt cannula. The irradiation was increased to 34 minutes since the contact lens absorbs 11 % of the UVA light. The contact is kept over the cornea for more than 1 hour after the procedure. The technique had an excellent flattening effect, but the results of this technique were inferior compared to the conventional Dresden protocol. This was probably due to the difference in precorneal riboflavin with contact lens in place.[25]
Epithelial On Collagen Cross Linking
In this technique, the corneal epithelium is not scraped, and cross-linking is performed with epithelium. In the beginning, various studies showed limited riboflavin diffusion through the tight epithelial junctions, thus reducing the effectiveness of CXL.[26] However, this technique offered the advantage of no risk for keratitis, postoperative pain, and persistent epithelial defect. This problem has been overcome by adding chemicals that cause epithelial disruption, such as surfactant benzalkonium chloride (BAC) or ethylenediaminetetraacetic acid (EDTA).[27]
Wollensak and Iomdina conducted a study using iso-osmolar riboflavin (20% dextran) and BAC, which resulted in biochemical and biomechanical changes after cross-linking in comparison to the control group. However, studies have shown that CXL without epithelial scrape and using BAC containing proparacaine eye drops causes biomechanical stiffening, approximately one-fifth of that induced by Dresden protocol.[18] Another study of porcine eyes comparing epi-off versus epi-on showed that epi-on was 70 % less effective. Although few studies have shown promising results, the efficacy of epi-on is still controversial, and few studies have shown increased keratometry values and retreatment necessity in many cases.[28]
Accelerated Collagen Cross Linking
Accelerated CXL protocols were introduced to clinical practice to shorten the procedure length and reduce the exposure time of the cornea to sources of infection. This concept works on the Bunsen-Roscoe law of photochemical reciprocity: the same photochemical effect is achieved with a shorter irradiation time by a corresponding increase in irradiation intensity. The irradiation time is shortened from 30 minutes to 3 minutes with comparable results to the standard protocol and may be safe to use in thin corneas.[18][29] Therefore it is inferred that a higher intensity will be needed with reduced exposure time to achieve the same total energy. In a landmark study by Wernii et al., they studied the effect of irradiance between 3 and 90 mW/cm2. They found that radiation intensity above 50 mW/cm^2 with a time of fewer than 2 minutes failed to increase the corneal stiffness. It is known that UVA is highly toxic to endothelial cells and has cytotoxic activity. Cytotoxic levels of UVA have been described as approximately 0.35 mW/cm2, which is twice that of protocol (0.18 mW/cm^2). Riboflavin helps in reducing the toxic effect of UVA and increases corneal stiffness.[30]
A cut-off of 400 microns is taken to avoid damaging the endothelium, although sometimes endothelial damage can occur even above 400 microns. In a comparative study of four CXL protocols by Shetty et al., in steeper corneas, they found that corneas treated with Dresden protocol (3 mW/cm^2 for 30 minutes) showed a more significant flattening effect compared to accelerated CXL protocol of 9, 18, and 30 mW/cm2. In a study by Kanellopoulos of 21 patients treated with accelerated CXL protocol in one eye and Dresden in the other, they found equal results in both eyes with no progression of keratoconus endothelial damage, and there was good improvement in visual acuity and keratometry.[31]
It is also known that a minimum oxygen concentration is needed in the stroma for CXL to occur and while applying UVA light in the presence of riboflavin. Moreover, considering the theoretical model of photochemical kinetics of CXL, the UVA radiations produce a reduction in the available oxygen in the riboflavin-applied cornea, leading to a secondary reduction in reactive oxygen species. When UV light is turned off, the oxygen level is returned to the original level from the environment within 3-4 minutes. Another alternative to increase the level of oxygen across the corneal stroma is to push the UV light during the cross-linking process to allow reoxygenation during the pause in exposure.[18]
Iontophoresis
A technique currently under investigation is iontophoresis CXL, which facilitates the penetration of riboflavin through the cornea through a low-intensity electrical current. Iontophoresis shortens riboflavin penetration time and duration of irradiation and does not require epithelium removal.[32] There are, as yet, no long-term published studies comparing this to conventional CXL, and the results of short-term follow-up show that it may also be inferior to conventional CXL.[33]
Combined Collagen Cross Linking
Combining CXL with other modalities was initially suggested by Kymionis et al. in 2011, known as 'CXL Plus.'[29] The technique involved doing PRK in KC patients to regularise the astigmatic corneal surface, followed by CXL to strengthen the corneal biomechanically, thus giving the additional benefit of improvement in visual acuity, which CXL alone cannot offer. Intracorneal ring segments (ICRS) have proven helpful in KC or post-LASIK ectasia patients for regularising corneal astigmatism, but they cannot halt the progression of the disease. According to a few studies combining CXL with ICRS does offer the combined benefit of improved visual acuity and strengthened cornea biomechanically.[29]
Complications
Common complications related to this include temporary corneal haze (10 to 90%), delayed epithelial closure, sterile infiltrates, and central stromal scars. The literature describes postoperative microbial keratitis from bacterial, herpetic, protozoal, and fungal sources.[34] The stromal haze is usually temporary and appears to be likely due to increased edema and keratocyte activation and occurs three to six months post-operatively.[34][35]
Rarer but more serious side effects include corneal melts and endothelial failure. Treatment failure is also a noted complication, defined as the progression of the condition with an increase in Kmax values of 1.0 D over the pre-operative value or greater than a 10% decrease in pachymetry readings six months post-operatively; this may occur in up to 10% of patients.[1]
Clinical Significance
More than 15 years since its introduction, conventional CXL has proven to be a safe and effective means of halting the progression of corneal ectasias, reducing the need for more invasive treatment options. Modified CXL techniques have had mixed results regarding efficacy when compared to conventional CXL, and long-term follow-up results are still needed. In addition to treating ectasias, CXL has increasing utilization as a treatment for microbial keratitis and reduction of low myopia, and further research into these domains is still underway.[36][37]
The number of CXL procedures performed in the last ten years has increased drastically. The majority of patients are seen in the first two decades of life. Hence it is highly imperative to pick up these patients early in the course of the disease, document the progression, and cross-link these eyes so that irreversible visual loss can be prevented. There are treatment options for thin corneas of less than 400 um, and vision can be safeguarded in most cases.[38]
Enhancing Healthcare Team Outcomes
An interprofessional team approach involving subspecialty-trained physicians and ophthalmic-trained nurses providing patient support and follow-up care will lead to the best outcomes. With early detection of keratoconus by healthcare members, corneal crosslinking is an effective way of slowing down and potentially halting progression.[39] [Level 3]
Nursing, Allied Health, and Interprofessional Team Interventions
Nursing, allied health staff, and interprofessional team play a key role in patient preparation and pre-operative and postoperative counseling of keratoconus patients.[40]
Nursing, Allied Health, and Interprofessional Team Monitoring
Nursing, allied health staff, and interprofessional team help monitor these patients after the surgery for BCL removal, intraocular pressure check, regular medication use, Schiempflug imaging visit, and to document any progression with the change of spectacles or contact lenses.[40]